Volume 3, Number 3—September 1997
Synopsis
Borna Disease Virus Infection in Animals and Humans
Cite This Article
Citation for Media
Abstract
The geographic distribution and host range of Borna disease (BD), a fatal neurologic disease of horses and sheep, are larger than previously thought. The etiologic agent, Borna disease virus (BDV), has been identified as an enveloped nonsegmented negative-strand RNA virus with unique properties of replication. Data indicate a high degree of genetic stability of BDV in its natural host, the horse. Studies in the Lewis rat have shown that BDV replication does not directly influence vital functions; rather, the disease is caused by a virus-induced T-cell--mediated immune reaction. Because antibodies reactive with BDV have been found in the sera of patients with neuropsychiatric disorders, this review examines the possible link between BDV and such disorders. Seroepidemiologic and cerebrospinal fluid investigations of psychiatric patients suggest a causal role of BDV infection in human psychiatric disorders. In diagnostically unselected psychiatric patients, the distribution of psychiatric disorders was found to be similar in BDV seropositive and seronegative patients. In addition, BDV-seropositive neurologic patients became ill with lymphocytic meningoencephalitis. In contrast to others, we found no evidence is reported for BDV RNA, BDV antigens, or infectious BDV in peripheral blood cells of psychiatric patients.
Borna disease (BD), first described more than 200 years ago in southern Germany as a fatal neurologic disease of horses and sheep, owes its name to the town Borna in Saxony, Germany, where a large number of horses died during an epidemic in 1885. The virus etiology of BD was proven in the early 1900s when Zwick and coworkers (1) in Giessen, Germany, successfully transmitted brain homogenates from infected horses to experimental animals. Other milestones in BD-related research were the demonstration of virus growth in cell cultures (2-4); the finding that the pathogenesis of BD is caused by a T-cell--dependent immune mechanism (5-8); and most recently, the molecular characterization of the etiologic agent of BD, the highly neurotropic Borna disease virus (BDV) (9-17).
BD is characterized by a disseminated nonpurulent meningoencephalomyelitis with infiltration of mononuclear cells (1,8,18,19) and a predilection for the gray matter of the cerebral hemispheres and the brain stem (8,19). In neurons, sometimes in glia cells, acidophilic intranuclear inclusion bodies, called Joest-Degen inclusion bodies, are occasionally found. BD occurs sporadically in Germany and Switzerland; its presence in other countries has not yet been substantiated. Natural infections in other Equidae, ruminants, rabbits, cats, and ostriches have also been described (19-21).
This review discusses the etiology of Borna disease, the natural and experimental infection in various animal species, the pathogenesis of the disease in the experimental rat model, the genetic stability of BDV, and the possible link between BDV or a similar agent and human neuropsychiatric disorders.
The etiologic agent of BD, BDV, has been recently characterized as an enveloped, nonsegmented, negative-stranded RNA virus with a genomic size of approximately 9 kb and a nuclear site for replication and transcription (14-17). The genomic organization is similar to that of members of the Mononegavirales order; therefore, BDV is the prototype of the new family Bornaviridae within this order. The Mononegavirales also include Filoviridae (e.g., Marburg and Ebola viruses), Paramyxoviridae (e.g., mumps, measles virus), and Rhabdoviridae (e.g., rabies, vesicular stomatitis virus). Six major open reading frames (ORFs)(I,II,III, IV,V,x1) are predicted in the genome sequence (16,17). Only five ORFs correspond to previously identified proteins with molecular weights of 10 (ORF x1: p10 BDV gene; 22), 18 (ORF III: gp18 BDV gene; 23), 24 (ORF II: p24 BDV gene; 24), 38/39 (ORF I: p38 BDV gene; 25), and 94 (ORF IV: p57 BDV gene; 26) or 84 (ORF IV: p57 BDV gene; 27, Richt et al., unpub. obs.) kDa.
Natural Infection
Extensive epizootiologic studies in horses have shown that BD is rare but occurs all over Germany, extending beyond the classic disease-endemic regions (28-30). Furthermore, BDV-specific antibodies were detected in horses in several European countries, Israel (28,29), Japan (31), Iran (32), and the United States (33). Since BDV-specific antibodies are frequently found in clinically healthy horses (20,28,29), natural infections in horses seem to remain subclinical in most cases. Unknown exogenous and endogenous factors might influence the genesis of the disease (20).
In addition to its predominant natural host, the horse, other Equidae, sheep, cattle, rabbits, goats, deer, alpacas, llamas, cats, pigmy hippopotamus, sloth, vari monkeys (memur variegatus), and ostriches have become naturally infected with BDV (8,19,20,34,35). In sheep flocks, clinical BD can affect large numbers of animals; however, in horse stables, usually only a few animals show clinical signs. The virus is assumed to be transmitted through salival, nasal, or conjunctival secretions because BDV-specific RNA has been found in these secretions (20,28,29,36). Animals become infected by direct contact with these secretions or by exposure to contaminated food or water. A minimum incubation period of 4 weeks is estimated for horses and sheep with nonspecific signs such as hyperthermia, anorexia, colic, and constipation in the initial phase of the disease. During the acute phase of disease, neurologic signs such as ataxia, depression, circular movement, standing in awkward positions, collapsing, running into obstacles, and paralysis, result from nonpurulent meningoencephalomyelitis. Clinical illness usually lasts 1 to 3 weeks, and death rates for diseased horses are 80% to 100% (18-20,37). In surviving animals, recurrent episodes are possible, especially after stress (18). Clinical manifestations, however, may vary among individual animals and various species.
BD tends to occur in spring and early summer and is more frequent in some years than in others; therefore, arthropods have been discussed as a potential vector. BDV, however, has never been isolated from insects in Europe. In the Near East, ticks have been associated with transmission of an equine encephalomyelitis similar to BD (38). A definite virus reservoir for BDV has not been found; various rodents most likely represent such a reservoir. In addition, since many seropositive horses with subclinical infections have infectious virus or BDV-specific RNA in various secretions, they can be potential sources of infection for other animals and humans (29,36).
Experimental Infection
BDV can be experimentally transmitted to a wide variety of animal species, from chickens to nonhuman primates (1,8,19,20,37). Incubation periods, clinical signs, and severity of the disease depend on the animal species and the virus variants. Rabbits, Lewis rats, and guinea pigs are highly susceptible, whereas chickens, monkeys, cattle, and tree shrews (Tupaia glis; 39) are less susceptible (8,19). Some animals (hamsters, black-hooded rats, mice, ferrets, pigeons, and dogs), however, do not develop disease despite being persistently infected with BDV (1,8,18,19). The pathohistologic picture after experimental infections resembles that of natural infections. Most experimentally infected animals develop perivascular and parenchymal central nervous system (CNS) infiltrations and have infectious virus in the brain tissue. Frequently, the clinical picture in experimentally infected animals does not differ from that of spontaneously infected natural hosts.
The result of BDV infection in rats depends on the inbred rat strain and the virus isolate used for infection. Black-hooded rats do not show clinical signs after BDV infection with BDV isolates despite persistent virus infection and mononuclear infiltration of the CNS (40). Lewis rats, however, are highly susceptible and are therefore used extensively for studies of BD pathogenesis (see below; 5,8,19). When infected with various BDV variants, Lewis rats exhibited clinical manifestations such as behavioral disorders, paralytic disease, or obesity, in addition to fertility problems (8,19), which indicates that BDV can form virus variants with different biologic properties.
BDV is a highly neurotropic agent that gains access to the CNS, probably by intraaxonal migration through the olfactory nerve or nerve endings in the oropharyngeal and intestinal regions (18,41). Virus spreads throughout the CNS by intraaxonal transport and centrifugally into the peripheral nerves. Antibody titers in naturally infected animals, such as in horses with clinical BD, are relatively low and are found in sera and cerebrospinal fluid (CSF) (19,20,28,29). Whether BDV-specific antibodies are neutralizing is not certain (5,42); the coexistence of BDV-specific antibodies and infectious virus in the CSF contradicts this assumption (43).
Extensive studies in the Lewis rat showed that the immune response to viral antigens after BDV infection does not elicit protective immunity but rather an immunopathologic reaction in which T cells play an important role (5,44). After adult rats are infected intracerebrally or intranasally, productive virus replication is found in the entire CNS. Once introduced into the rat's CNS, BDV usually causes a persistent infection with continuous productive replication in the brain and spinal cord (5,8). In immunocompetent animals, no infectivity was found in extraneural tissues at any stage of infection (5,45). In newborn animals, in contrast, the virus spreads throughout the whole organism; BDV-specific antigen was found in parenchymal cells of numerous organs, and infectious virus was found in excretions (45). The age of rats at the time of infection affects the capacity of host cells to support virus replication and, therefore, to control the spread of BDV.
The reason for the variation in virus replication is still unclear, and differences in the host's immunocompetence cannot be the only factor, since in adult immunosuppressed rats the virus is strictly neurotropic (45). All immunocompetent animals develop antibodies to BDV-specific antigens, and these antibodies coexist with infectious virus in the CNS (5). The outcome of BDV infections in rats depends on the rat strain and the passage history of the virus (see above; 19,40).
Despite productive virus replication in the CNS, immunocompromised animals (immunologically incompetent newborn and athymic rats) and immunosuppressed (by cyclophosphamide or cyclosporine A) animals do not become ill with BD or encephalitis, as do immunocompetent animals (5-8,29,44,46,47). The hypothesis of a virus-induced cell-mediated immunopathologic basis of BD was confirmed in adoptive transfer experiments in which spleen cells from infected rats were transferred into immunosuppressed virus carriers (5). Recipient rats developed BDV-specific antibodies, encephalitis and disease (5). The critical role of T cells in the pathogenesis of BD was demonstrated in adoptive transfer experiments: transfer of BDV-specific major histocompatibility complex class II-restricted TH1 cell lines into drug-tolerized virus carriers resulted in acute meningoencephalitis and disease (Table 1; 6-8,29,47).
Consequently, conventional immunization with inactivated virus preparations or purified virus-specific antigens did not confer protective immunity against clinical BD, despite detectable levels of virus-specific antibodies after immunization (25; Richt, unpub. data). However, when viable BDV-specific TH1 cells were passively transferred into naive rats before virus infection, animals were protected against clinical BD after challenge (Table 1; 29,47,48, Schmeel et al., unpub. obs.). In contrast, injection of the same viable BDV-specific TH1 cells after BDV-infection induced clinical disease faster in BDV-uninfected animals than in BDV-infected control animals (29,47,48, Schmeel et al., unpub. obs.). Thus, in BDV infections of the CNS with immunopathologic lesions, the same virus-specific immune cells that cause disease can also prevent disease, depending on the dynamics of virus replication at the time of the T-cell response.
Several reports of the pathogenesis of BD in BDV-infected animals indicate that the pathogenesis of BD in naturally and experimentally infected animals is also immunopathologic since the pathohistologic picture, including CNS-infiltrating cells in naturally BDV-infected animals, resembles that of experimentally infected rats (19); and immunosuppression or splenectomy of experimentally BDV-infected rabbits (49) and splenectomy of BDV-infected monkeys (Macaca mulatta; 50) affected the severity of clinical signs or prevented disease.
To study the genetic variability of BDV, virus isolates were derived from three clinically ill horses in Germany (Lower Saxony, Rhineland-Palatina, Bavaria), euthanized between 1989 and 1995. BDV-specific antibodies were detected in the serum, and infectious virus, viral proteins, and viral RNA were detected in the CNS of all three horses (Pfeuffer et al., unpub. obs., 51). Molecular analysis of five different ORFs was performed. The coding regions of ORFs I, II, III, IV, and x1 were amplified by the reverse transcriptase-polymerase chain reaction (RT-PCR) technique with total RNA isolated from the CNS (36); the amplified cDNA was cloned and sequenced. The sequences of the respective ORFs were aligned and compared with viral sequences derived from a cell-culture_adapted virus strain, BDV strain V (14), which was isolated in 1929 from a horse with clinical BD. In the analyzed clones, two deletions resembling post-transcriptionally spliced mRNA species were identified (Pfeuffer et al., unpub. obs., 51). Similarly, two introns were recently identified when cell-culture--adapted BDV isolates were molecularly analyzed (16,17).
The degree of homology between the analyzed BDV isolates and the reference BDV strain V isolated more than 60 years ago (14) for the p24 BDV gene is summarized in Table 2. There is a maximal divergence of 2.0% at the nucleotide level and 2.5% at the amino acid level (Table 2). These data agree with recently published observations showing a high degree of homology within the p24 BDV gene of wild-type and cell-adapted viruses (52,53). When the other four ORFs were similarly analyzed, a homology of >95% at the nucleotide level and >97% at the amino acid level was found (Pfeuffer et al., unpub. obs., 51), indicating that in addition to the previously analyzed p24 (ORF II; 52,53) and p38 (ORF I; 53) genes, the other BDV-ORFs (ORFs III, IV, x1) also exhibit high genetic stability within one host species, the horse (Pfeuffer et al., unpub. obs., 51).
Nevertheless, antigenic variations of BDV wild-type isolates were found when brain material from approximately 50 horses with BDV infections were screened by immunocytochemistry and immunoblot assays with BDV-specific monoclonal and monospecific antibodies (51). Two isolates (horse #215, horse #2,300) showed a remarkable difference in antigenicity (Figure 1A,B). These isolates were not recognized by the p38 specific monoclonal antibody (mAb) Bo18 (25,51; Figure 1A). Polyclonal reconvalescent (51) and monospecific p38 antiserum or monospecific p24 antiserum (Figure 1B), however, reacted with BDV-specific proteins from both isolates in immunocytochemistry assays (Figure 1B) and immunoblots (51), indicating that the p38 and p24 BDV-proteins were synthesized. The recognition site for the mAb Bo18 within the p38 BDV-protein, however, was altered. To obtain detailed information on the location of these changes, we cloned and analyzed the p38 BDV gene of two horse isolates. Sequence analysis showed several nucleotide mutations within the respective p38 BDV genes; only one, however, led to an amino acid replacement (51). The amino acid replacements were adjacent to each other within the N-terminal part of the p38 protein, indicating that they are within the epitope of mAb Bo18 (51).
BDV's wide host range and its association with behavioral abnormalities in many species such as rhesus macaques (1,50), tree shrews (Tupaia glis; 39), and rats (5,8) suggest that BDV may be involved in human neuropsychiatric illnesses. Seroepidemiologic studies have demonstrated BDV-specific antibodies in sera of psychiatric and neurologic patients in Germany, Japan, and the United States (13,51,54-71). Recent studies with many patients showed a significantly higher prevalence of BDV serum antibodies among hospitalized psychiatric patients and a moderately higher seroprevalence among neurologic patients than among controls (13,56,60,68). Investigations of CSFs of BDV-seropositive patients acutely ill with psychiatric disorders (mainly schizophrenia and affective psychoses) showed intrathecally synthesized BDV-specific immunoglobulin (Ig) G in 25% of the patients (59).
A slightly higher level of seroprevalence was found among psychiatric patients in an area with high incidence of natural BD in horses (the BD-endemic area around Günzburg in Southern Germany) than among psychiatric patients in an area without endemic BD in the United States (Philadelphia area) (6.8% vs. 4.5%). The incidence in healthy controls was considerably lower in the United States (0% to 1%) than in the Günzburg area (3% to 3.5%; 55). These studies indicated a high number of clinically inconspicuous BDV-seropositive persons in the BD-endemic area. Therefore, as is the case in horses, inapparent BDV infections may be very common in humans (28,29,55).
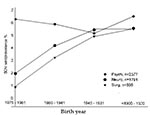
An epidemiologic investigation of BDV seroprevalence in a large number (n = 4,673) of surgical (control), neurologic, and psychiatric patients showed continuous age-dependent increase in seroprevalence in controls and neurologic patients (Figure 2; 55); the dynamics of seropositivity were different in psychiatric patients. A twofold higher seroprevalence in young neurologic patients paralleled a higher prevalence in patients with acute or chronic meningoencephalitis and psychiatric disorders (56,71). Seroprevalence among the youngest quartile (aged 17-30 years) of psychiatric patients was 600% higher than among surgical patients (Figure 2; 55), which suggests a role of BDV infections in the pathogenesis of psychiatric disorders (psychiatric disorders frequently begin in younger persons).
Initial investigations indicated a correlation between BDV-specific antibodies and affective psychoses (54,67). In a random group of psychiatric patients, however, we found a similar distribution of psychiatric disorders among BDV-seropositive and BDV-seronegative patients (multiple psychiatric disorders in one patient and brain atrophy were higher among the BDV- seropositive patients [71]). An unequivocal classification to a definite psychiatric disease is not possible at the moment.
If a causal relationship exists between BDV infections in naturally infected animals and BDV seropositivity in humans, identifying patients with symptoms and disorder progression similar to those of BDV-infected animals (paresis, weakness of legs, excitability, or depression) would be important. Recently, we identified a BDV-seropositive neurologic patient who exhibited abnormal body postures, depressive and apathetic behavior, and paresis (57). The patient worked at his father's farm, where one of four horses and four of 10 sheep had antibodies specific for BDV (57). The patient initially had a cerebral seizure and later neurologic and psychiatric symptoms resembling those of BD in animals and not typical of other known human encephalitic disorders, possibly implicating an acute, mild BDV encephalitis. Although isolation and molecular analysis of the respective viruses did not show an infectious chain, BDV transmission from animals to humans seems possible.
In addition, to study possible horizontal transmission of BDV within families, we reviewed the medical history of approximately 200 BDV-seropositive neuropsychiatric patients for coincident onset of mental or neurologic disorders during 1 year within each family. Of four families meeting the criteria, two were studied in detail; both had neuropsychiatric illness and BDV seropositivity in the primary patient and also in a second family member in the household (58). In two additional families, only the primary patients, not the other psychiatrically ill family members, showed BDV seropositivity (58). In summary, humans can be infected by BDV or a similar agent; a correlation between BDV infection in animals and humans exists; and BDV infections seem to contribute to or initiate various psychiatric syndromes.
To verify the etiologic role of BDV in human neuropsychiatric disorders, we tested specimens from the CSF of more than 40 BDV-seropositive neuropsychiatric patients for isolates of BDV or a related agent. The CSFs were spread onto cell cultures or injected intracerebrally into rabbits. BDV or a related agent was detected in the CSF of three neuropsychiatric patients (two patients with acute lymphocytic meningoencephalitis, one patient with acute schizophrenic psychosis; 67). The CSF of the schizophrenic patient showed high amounts of BDV-specific IgG, which became normal 5 years later when the CSF of this patient was again analyzed during myelography before lumbar surgery; at that time the patient was mentally healthy (60). Injection of the respective CSFs onto cell cultures showed expression of BDV-specific antigens (by BDV-specific staining pattern in an indirect immunofluorescence assay). Expression of BDV antigen, however, was lost, when these cells were passaged (13,67). In addition, all three cerebrospinal fluids induced BDV-specific antibodies in inoculated animals; however, neither BDV replication nor BDV-specific antigen was detected in the CNS of any of these animals (13,67).
Most recently, de la Torre and coworkers (72) detected both BDV-specific antigen and BDV-RNA in four autopsied human brains with hippocampal sclerosis and astrocytosis, which confirms previous studies that BDV can infect human brain tissue; whether BDV infection contributes to the pathophysiology of human neuropsychiatric disorders, however, warrants further investigation.
Before this study, BDV-specific proteins (73), BDV-specific RNA (63,64,69,74,75), and infectious BDV (76) were found in peripheral blood mononuclear cells of psychiatric patients. In addition, the presence of BDV-RNA in these cells in 4% to 5% of Japanese blood donors was also reported (63). Since these results have important implications for BD pathogenesis, the intra vitam diagnosis, and the safety of blood and blood products, we conducted a double-blind study of BDV-specific markers in the blood of psychiatric patients. Samples were aliquoted before distribution to two different laboratories and tested independently. Forty-two seropositive psychiatric patients and four seronegative controls from a BD-endemic area in Germany were analyzed. Consecutive samples obtained over a 9-month period were included in the study. The nested RT-PCR method was used with primers specific for the p24 BDV-gene, the internal primers identical to the one used by Kishi and coworkers (63,64). Positive results for BDV-specific RNA were obtained only in the laboratory where BDV is routinely studied. Simultaneous analysis of the samples in another laboratory yielded negative results (62). In addition, cocultivation of peripheral blood mononuclear cells from 47 samples of seropositive patients over two passages and from eight samples over 8 to 10 passages with rabbit embryonic brain cells did not provide evidence of BDV or BDV-specific antigens (62). We thus concluded that contamination can have a serious influence on the outcome of studies using nested RT-PCR methods to probe for BDV-specific sequences in the peripheral blood mononuclear cells of psychiatric patients.
Viral infections of the CNS are often difficult to diagnose because conventional viral and serologic laboratory methods are still unsatisfactory. Existing standard techniques are either insensitive and slow (virus culture) or highly invasive and rare (brain biopsy) and, therefore, are unsatisfactory as a measure for the accuracy of a new technique such as PCR or RT-PCR (77). PCR techniques (PCR, RT-PCR, nested PCR), with their speed and high molecular sensitivity possess great potential for the diagnosis of viral infections of the CNS.
To overcome the discrepancies of recent RT-PCR studies about whether BDV or BDV-RNA can be detected in the peripheral blood of humans and animals, a multicenter study needs to be conducted with an independent center responsible for collecting and distributing samples to prevent potential contamination by BDV-RNA. These laboratories need to use similar RNA isolation and RT-PCR techniques. Furthermore, the methods used by the laboratories conducting sera or CSF analysis for BDV-specific antibodies in animals and humans urgently need further standardization.
Studies of the epizootiology and epidemiology of BDV infections in the last decade underscore the importance of BD as an emerging zoonosis. Data clearly indicate that BDV or an analogous virus can also infect humans. In addition to the seroepidemiologic results, data from preliminary studies on the isolation of BDV from the CSF and the detection of BDV-specific antigen and BDV-RNA in human brain confirm this conclusion. Therefore, humans belong to the wide spectrum of animal species, from birds to mammals, that are susceptible to BDV infections.
The question of whether BDV infections in humans contribute to human neuropsychiatric disorders remains unsolved. A convincing argument might be the significantly increased BDV-seroprevalence in psychiatric patients compared with healthy controls. That some mentally or neurologically healthy persons are BDV seropositive does not contradict this hypothesis, since the natural infection in horses remains subclinical in most cases and experimental intracerebral application of BDV into highly susceptible animals does not always result in clinical disease (Rott, pers. comm.). In addition, epizootiologic studies suggest that BDV is not highly contagious since only individual horses within a stable succumb to clinical disease. Similarly, analysis of a possible horizontal transmission of BDV within family members and spouses of approximately 200 BDV-seropositive neuropsychiatric patients showed only few cases of coincident onset of mental or neurologic disorders within 1 year. Whether BDV or BDV-RNA is present in human peripheral blood mononuclear cells remains questionable. Depending on the virus strain and the host, BDV can induce in animals a variety of clinical manifestations, from inapparent infections, behavioral abnormalities, obesity, fertility problems to fatal neurologic disease. Animals frequently show behavioral abnormalities during the course of the disease. This was very obvious when tree shrews (Tupaia glis), classified phylogenetically at the root of primates, were infected with BDV. Here, mainly disturbances in social behavior and severe apathy were observed. In contrast, an unequivocal classification of a definite psychiatric disease with BDV infections in humans is at present not possible.
To establish the etiologic role of BDV infections in human psychiatric symptoms, the following findings must be considered: symptomatology of BD in animals is highly variable depending upon many factors such as age, immune status, and strain of the animal used for infection, in addition to the genetic makeup of the virus; psychiatric symptomatology is nonspecific (78) even for known genetic causes, which has represented a major problem for research investigating known neuropsychiatric disorders caused by infectious agents. Despite the similarities in BDV infections of animals and humans, additional future molecular, seroepidemiologic, and clinical studies will be required to substantiate the possible contribution of BDV to the pathophysiology of neuropsychiatric disorders.
Dr. Richt is a research scientist and group leader at the Institute of Virology, Justus-Liebig-Universität Giessen, Germany. His current research topic is molecular pathogenesis of Borna disease virus infections. His interests include the epidemiology/epizootiology and immunopathology of Borna disease virus infections and the replication strategy of Borna disease virus.
Acknowledgment
This paper is dedicated to Prof. Hermann Becht on the occasion of his 65th birthday.
References
- Zwick W. Bornasche Krankheit und Encephalomyelitis der Tiere. In: Gildenmeister E, Haagen E, Waldmann O, editors. Handbuch der Viruserkrankungen.Gustav Fischer Verlag, Jena; 1939. p. 252-354.
- Mayr A, Danner K. Production of Borna virus in tissue culture. Proc Soc Exp Biol Med. 1972;140:511–5.PubMedGoogle Scholar
- Ludwig L, Becht H, Groh L. Borna disease (BD), a slow virus infection--biological properties of the virus. Med Microbiol Immunol (Berl). 1973;158:275–89.
- Herzog S, Rott R. Replication of Borna disease virus in cell cultures. Med Microbiol Immunol (Berl). 1980;173:153–8.
- Narayan O, Herzog S, Frese K, Scheefers H, Rott R. Behavioral disease in rats caused by immunopathological responses to persistent Borna virus in the brain. Science. 1983;220:1401–3.PubMedGoogle Scholar
- Richt JA, Stitz L, Wekerle H, Rott R. Borna disease, a progressive encephalomyelitis as a model for CD4+ T cell-mediated immunopathology in the brain. J Exp Med. 1989;170:1045–50.PubMedGoogle Scholar
- Richt JA, Stitz L, Deschl U, Frese K, Rott R. Borna disease virus-induced meningoencephalomyelitis caused by a virus-specific CD4+ T cell-mediated immune reaction. J Gen Virol. 1990;71:2565–73.PubMedGoogle Scholar
- Richt JA, VandeWoude S, Zink MC, Clements JE, Herzog S, Stitz L, Infection with Borna disease virus: molecular and immunobiological characterization of the agent. Clin Infect Dis. 1992;14:1240–50.PubMedGoogle Scholar
- Lipkin WI, Travis GH, Carbone KM, Wilson MC. Isolation and characterization of Borna disease agent cDNA clones. Proc Natl Acad Sci U S A. 1990;87:4184–8.PubMedGoogle Scholar
- VandeWoude S, Richt JA, Zink MC, Rott R, Narayan O, Clements JE. A Borna virus cDNA encoding a protein recognized by antibodies in humans with behavioral diseases. Science. 1990;250:1278–81.PubMedGoogle Scholar
- Richt JA, VandeWoude S, Zink MC, Narayan O, Clements JE. Analysis of Borna disease virus-specific RNAs in infected cells and tissues. J Gen Virol. 1990;72:2251–5.
- Richt JA, Clements JE, Herzog S, Pyper J, Wahn K, Becht H. Analysis of virus-specific RNA species and proteins in Freon-113 preparations of the Borna disease virus. Med Microbiol Immunol (Berl). 1993;182:271–80.
- Richt JA, Herzog S, Pyper JM, Clements JE, Narayan O, Bechter K, Borna disease virus: nature of the etiologic agent and significance of infection in man. Arch Virol. 1993;7(Suppl):101–9.
- Briese T, Schneemann A, Lewis AJ, Park YS, Kim S, Ludwig H, Genomic organization of Borna disease virus. Proc Natl Acad Sci U S A. 1994;91:4362–6.PubMedGoogle Scholar
- Cubitt B, Oldstone C, de la Torre JC. Sequence and genome organization of Borna disease virus. J Virol. 1994;68:1382–96.PubMedGoogle Scholar
- de la Torre JC. Molecular biology of Borna disease virus: prototype of a new group of animal viruses. J Virol. 1994;68:7669–75.PubMedGoogle Scholar
- Schneemann A, Schneider PA, Lamb RA, Lipkin WI. The remarkable coding strategy of Borna disease virus: a new member of the nonsegmented negative strand RNA viruses. Virology. 1995;210:1–8.PubMedGoogle Scholar
- Heinig A. Die Bornasche Krankheit der Pferde und Schafe. In: Roehrer H, editor. Handbuch der Virusin-fektionen bei Tieren. VEB Fischer Verlag: Jena 1969;4:83-148.
- Rott R, Becht H. Natural and experimental Borna disease in animals. Borna disease. Curr Top Microbiol Immunol. 1995;190:17–30.PubMedGoogle Scholar
- Becht H, Richt JA. Borna disease. In: Studdert MJ, editor. Virus diseases of equines. Amsterdam: Elsevier Science Publishers BV; 1996. p. 235-44.
- Malkinson M, Weisman Y, Perl S, Asash EA. Borna-like disease of ostriches in Israel. Borna Disease. Curr Top Microbiol Immunol. 1995;190:31–8.PubMedGoogle Scholar
- Wehner T, Ruppert A, Frese K, Herden C, Becht B, Richt JA. Detection of a novel Borna disease virus encoded protein of 10 kilodalton in infected cells and tissues. J Gen Virol. 1997;78. In press.PubMedGoogle Scholar
- Kliche S, Briese T, Henschen AH, Stitz L, Lipkin WI. Characterization of a Borna disease virus glycoprotein, gp18. J Virol. 1994;68:6918–23.PubMedGoogle Scholar
- Thiedemann N, Presek P, Rott R, Stitz L. Antigenic relationship and further characterization of two major Borna disease virus proteins. J Gen Virol. 1992;73:1057–64.PubMedGoogle Scholar
- Haas B, Becht H, Rott R. Purification and properties of an intranuclear virus-specific antigen from tissues infected with Borna disease virus. J Gen Virol. 1986;67:235–41.PubMedGoogle Scholar
- Schneider PA, Hatalski CG, Lewis AJ, Lipkin WI. Biochemical and functional analysis of the borna disease virus G protein. J Virol. 1997;71:331–6.PubMedGoogle Scholar
- Gonzalez-Dunia D, Cubitt B, Grässer FA, de la Torre JC. Characterization of Borna disease virus p56 protein, a surface glycoprotein involved in virus entry. J Virol. 1997;71:3208–18.PubMedGoogle Scholar
- Herzog S, Frese K, Richt JA, Rott R. Ein Beitrag zur Epizootiologie der Bornaschen Krankheit des Pferdes. Wien Tierärztl Wschr. 1994;81:374–9.
- Richt JA, Herzog S, Schmeel A, Frese K, Rott R. Current knowledge about Borna disease. In: Nakajima H, Plowright W, editors. Equine infectious diseases VII. Newmarket: R & W Publications; 1994. p. 55-60.
- Dürrwald R. Die natürliche Borna-Virus-Infektion der Einhufer und Schafe: Untersuchungen zur Epidemiologie, zu neueren diagnostischen Methoden (ELISA, PCR) und zur Antikörperkinetik bei Pferden nach Vakzination mit Lebendimpfstoff. Inaugural-Dissertation, Freie Universität Berlin; 1993.
- Nakamura Y, Kishi M, Nakaya T, Asahi S, Tanaka H, Sentsui H, Demonstration of Borna disease virus RNA in peripheral blood mononuclear cells from healthy horses in Japan. Vaccine. 1995;13:1076–9.PubMedGoogle Scholar
- Bahmani MK, Nowrouzian I, Nakaya Y, Nakamure Y, Hagiwara K, Takahashi H, Varied prevalence of Borna disease virus infection in Arabic, thoroughbred and their cross-bred horses in Iran. Virus Res. 1996;45:1–13.PubMedGoogle Scholar
- Kao M, Hamir AN, Rupprecht CE, Fu ZF, Shankar V, Koprowski H, Detection of antibodies against Borna disease virus in sera and CSF of horses in the USA. Vet Rec. 1993;132:241–4.PubMedGoogle Scholar
- Schüppel KF, Kinne J, Reinacher M. Bornavirus-Antigennachweis bei Alpakas (Lama pacos) sowie bei einem Faultier (Choleopus didiactylus) und einem Zwergflu§pferd. Verh Erkr Zootiere. 1994;36:189–93.
- Schüppel KF, Kinne J, Lebelt J, Reinacher M. Zwei Fälle von Bornascher Krankheit bei Varis (Memur variegatus). 38. Tagung der Fachguppe "Pathologie der DVG", Hamburg; 1995.
- Richt JA, Herzog S, Haberzettl K, Rott R. Demonstration of Borna disease virus-specific RNA in secretions of naturally infected horses by the polymerase chain reaction. Med Microbiol Immunol (Berl). 1993;182:293–304.
- Ludwig H, Kraft W, Kao M, Gosztonyi G, Dahme E, Krey HF. Die Borna-Krankheit bei natürlich und experimentell infizierten Tieren: Ihre Bedeutung für Forschung und Praxis. Tierarztl Prax. 1985;13:421–53.PubMedGoogle Scholar
- Daubney R, Mahlau EA. Viral encephalomyelitis of equines and domestic ruminants in the Near East. Res Vet Sci. 1967;8:375–97.PubMedGoogle Scholar
- Sprankel H, Richarz K, Ludwig H, Rott R. Behavior alterations in tree shrews (Tupaia glis, Diard 1820) induced by Borna disease virus. Med Microbiol Immunol (Berl). 1978;165:1–18.
- Herzog S, Frese K, Rott R. Studies on the genetic control of resistance of black hooded rats to Borna disease. J Gen Virol. 1991;72:535–40.PubMedGoogle Scholar
- Morales JA, Herzog S, Kompter C, Frese K, Rott R. Axonal transport of Borna disease virus along olfactory pathways in spontaneously and experimentally infected rats. Med Microbiol Immunol (Berl). 1988;177:51–68.
- Ludwig H, Furuya K, Bode L, Klein N, Dürrwald R, Lee DS. Biology and neurobiology of Borna disease viruses (BDV), defined by antibodies, neutralizability and their pathogenic potential. Arch Virol. 1993;7(Suppl):111–33.
- Ludwig H, Koester V, Pauli G, Rott R. The cerebrospinal fluid of rabbits infected with Borna disease virus. Arch Virol. 1977;55:209–23.PubMedGoogle Scholar
- Stitz L, Dietzschold B, Carbone KM. Immunopathogenesis of Borna disease. Curr Top Microbiol Immunol. 1995;190:75–92.PubMedGoogle Scholar
- Herzog S, Kompter C, Frese K, Rott R. Replication of Borna disease virus in rats: age-dependent differences in tissue distribution. Med Microbiol Immunol (Berl). 1984;173:171–7.
- Stitz L, Soeder D, Deschl U, Frese K, Rott R. Inhibition of immune-mediated meningoencephalitis in persistently Borna disease virus-infected rats by Cyclosporine A. J Immunol. 1989;143:4250–6.PubMedGoogle Scholar
- Richt JA, Schmeel A, Frese K, Carbone KM, Narayan O, Rott R. Borna disease virus-specific T cells protect against or cause immunopathological Borna disease. J Exp Med. 1994;179:1467–73.PubMedGoogle Scholar
- Schmeel A, Frese K, Richt JA. Immunopathogenesis of Borna disease virus infections in rats: role of BDV-specific TH1 cells in induction and prevention of Borna disease. In: Schwyzer M, Ackermann M, Bertoni G, Kocherhans R, McCullough K, Engels M, Wittek R, Zanoni R, editors. Immunobiology of viral infections. Proceedings of the 3rd Congress ESVV Interlaken 1995 Sep; 1995. p. 182-7.
- Gierend M, Ludwig H. Influence of immunosuppressive treatment on Borna disease in rabbits. Arch Virol. 1981;67:217–28.PubMedGoogle Scholar
- Stitz L, Krey HF, Ludwig H. Borna disease in rhesus monkeys as a model for uveo-cerebral symptoms. J Med Virol. 1980;6:333–40.
- Herzog S, Pfeuffer I, Haberzettl K, Feldmann H, Frese K, Bechter K, Molecular characterization of Borna disease virus from naturally infected animals and possible links to human disorders. [Suppl.]. Arch Virol. 1997;▪▪▪:13.
- Binz T, Lebelt J, Niemann H, Hagenau K. Sequence analysis of the p24 gene of Borna disease virus in naturally infected horse, donkey and sheep. Virus Res. 1994;34:281–9.PubMedGoogle Scholar
- Schneider PA, Briese T, Zimmermann W, Ludwig H, Lipkin WI. Sequence conservation in field and experimental isolates of Borna disease virus. J Virol. 1994;68:63–8.PubMedGoogle Scholar
- Amsterdam JD, Winokur A, Dyson W, Herzog S, Gonzalez F, Rott R, Borna disease virus: a possible etiological factor in human affective disorders? Arch Gen Psychiatry. 1985;42:1093–6.PubMedGoogle Scholar
- Bechter K. Borna disease virus--mögliche Ursache neurologischer und psychiatrischer Störungen des Menschen. Habilitationsschrift der Fakultät für Klinische Medizin der Universität Ulm. 1995.
- Bechter K, Herzog S, Schüttler R. Possible significance of Borna disease for humans. Neurology Psychiatry Brain Research. 1992;1:23–9.
- Bechter K, Herzog S, Schüttler R. Case of neurological and behavioral abnormalities: due to Borna disease virus encephalitis? Psychiatry Res. 1992;42:193–6.PubMedGoogle Scholar
- Bechter K, Herzog S, Schüttler R. Borna disease virus: possible causal agent in psychiatric and neurological disorders in two families. Psychiatry Res. 1992;42:291–4.PubMedGoogle Scholar
- Bechter K, Herzog S, Behr W, Schüttler R. Investigations of cerebrospinal fluid in Borna disease virus seropositive psychiatric patients. Eur Psychiatry. 1995;10:250–8.PubMedGoogle Scholar
- Bechter K, Herzog S, Schüttler R. Borna disease-possible cause of human neuropsychiatric disorders. Neurology Psychiatry Brain Research. 1996;4:45–52.
- Bode L, Riegel S, Lange W, Ludwig H. Human infections with Borna disease virus: seroprevalence in patients with chronic diseases and healthy individuals. J Med Virol. 1992;36:309–15.PubMedGoogle Scholar
- Richt JA, Alexander RC, Herzog S, Hooper DC, Kean R, Spitsin S, Failure to associate human psychiatric disorders with Borna disease virus infection. J Neurovirol. 1997;3:174–8.PubMedGoogle Scholar
- Kishi M, Nakaya T, Nakamura Y, Kakinuma M, Takahashi TA, Sekiguchi S, Prevalence of Borna disease virus RNA in periopheral blood mononuclear cells from blood donors. Med Microbiol Immunol (Berl). 1995;184:135–8.
- Kishi M, Nakaya T, Nakamura Y, Zhong Q, Ikeda K, Senjo M, Demonstration of human Borna disease virus RNA in human peripheral blood mononuclear cells. FEBS Lett. 1995;3645:293–7.
- Kitze B, Herzog S, Rieckmann P, Poser S, Richt JA. No evidence of Borna disease virus-specific antibodies in multiple sclerosis patients in Germany. J Neurol. 1996;234:660–1.
- Kubo K, Fujijoshi T, Yokoyama MM, Kamei K, Richt JA, Kitze B, Lack of association of Borna disease virus and human T-cell leukemia virus type I infections with psychiatric disorders among Japanese patients. Clin Diagn Lab Immunol. 1997;4:189–94.PubMedGoogle Scholar
- Rott R, Herzog S, Fleischer B, Winokur A, Amsterdam J, Dyson W. Detection of serum antibodies to Borna disease virus in patients with psychiatric disorders. Science. 1985;228:755–6.PubMedGoogle Scholar
- Rott R, Herzog S, Bechter K, Frese K. Borna disease, a possible hazard for man? Arch Virol. 1991;118:143–9.PubMedGoogle Scholar
- Sauder C, Müller A, Cubitt B, Mayer J, Steinmetz J, Trabert W, Detection of Borna disease virus (BDV) antibodies and BDV RNA in psychiatric patients: evidence for high sequence conservation and human blood-derived BDV RNA. J Virol. 1997;70:7713–24.
- Waltrip RW II, Buchanan RW, Summerfelt A, Breier A, Carpenter WT, Bryant NL, Borna disease virus and schizophrenia. Psychiatry Res. 1995;56:33–44.PubMedGoogle Scholar
- Bechter K, Herzog S, Estler HC, Schüttler R. Increased psychiatric comorbidity in Borna disease virus seropositive psychiatric patients. Acta Psychiatr Belg. 1997. In press.
- de la Torre JC, Gonzalez-Dunia D, Cubitt B, Mallory M, Mueller-Lantzsch N, Grässer FA, Detection of Borna disease virus antigen and RNA in human autopsy brain samples from neuropsychiatric patients. Virology. 1996;223:272–82.PubMedGoogle Scholar
- Bode L, Steinbach F, Ludwig H. A novel marker for Borna disease virus infection. Lancet. 1994;343:297–8.PubMedGoogle Scholar
- Bode L, Zimmermann W, Ferszt R, Steinbach F, Ludwig H. Borna disease virus genome transcribed and expressed in psychiatric patients. Nat Med. 1995;1:232–6.PubMedGoogle Scholar
- Bode L. Human infections with Borna disease virus and potential pathogenic implications. Borna Disease. Curr Top Microbiol Immunol. 1995;190:103–30.PubMedGoogle Scholar
- Bode L, Dürrwald R, Rantam FA, Ferszt R, Ludwig H. First isolates of infectious human Borna disease virus from patients with mood disorders. Mol Psychiatry. 1996;1:200–12.PubMedGoogle Scholar
- Jeffrey KJM, Read SJ, Peto TEA, Mayon-White RT, Bangham CRM. Diagnosis of viral infections of the central nervous system: clinical interpretation of PCR results. Lancet. 1997;349:313–7.PubMedGoogle Scholar
- Buchsbaum MS, Rieder R. Biologic heterogeneity and psychiatric research. Arch Gen Psychiatry. 1979;36:1163–9.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 3, Number 3—September 1997
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Jürgen A. Richt, Institut für Virologie, Frankfurterstrasse 107, D-35392 Giessen, Germany; fax: 49-641-99-38359
Top