Volume 3, Number 4—December 1997
THEME ISSUE
Foodborne
Factors that Influence the Emergence or Reemergence and Dissemination of Microbial Foodborne Pathogens and Human Disease
Produce Handling and Processing Practices
Cite This Article
Citation for Media
Abstract
In the past decade, outbreaks of human illness associated with the consumption of raw vegetables and fruits (or unpasteurized products produced from them) have increased in the United States. Changes in agronomic, harvesting, distribution, processing, and consumption patterns and practices have undoubtedly contributed to this increase. Pathogens such as Listeria monocytogenes, Clostridium botulinum, and Bacillus cereus are naturally present in some soil, and their presence on fresh produce is not rare. Salmonella, Escherichia coli O157:H7, Campylobacter jejuni, Vibrio cholerae, parasites, and viruses are more likely to contaminate fresh produce through vehicles such as raw or improperly composted manure, irrigation water containing untreated sewage, or contaminated wash water. Contact with mammals, reptiles, fowl, insects, and unpasteurized products of animal origin offers another avenue through which pathogens can access produce. Surfaces, including human hands, which come in contact with whole or cut produce represent potential points of contamination throughout the total system of growing, harvesting, packing, processing, shipping, and preparing produce for consumption. Treatment of produce with chlorinated water reduces populations of pathogenic and other microorganisms on fresh produce but cannot eliminate them. Reduction of risk for human illness associated with raw produce can be better achieved through controlling points of potential contamination in the field; during harvesting; during processing or distribution; or in retail markets, food-service facilities, or the home.
Advances in agronomic, processing, preservation, packaging, shipping, and marketing technologies on a global scale have enabled the fresh fruit and vegetable industry to supply consumers with a wide range of high-quality produce year round. Some of the same technologies and practices have also introduced an increased risk for human illness associated with pathogenic bacteria, mycotoxigenic molds, viruses, and parasites. The use of manure rather than chemical fertilizer, as well as the use of untreated sewage or irrigation water containing pathogens, viruses, or parasites, undoubtedly contributes to this increased risk. Changes in the produce industry, social demographics, food consumption patterns, and awareness of fresh fruits and vegetables as potential vehicles of infection may also be contributing to an increase in documented produce-associated outbreaks of human illness.
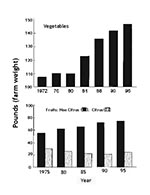
Changing factors that contribute to the epidemiology of diseases that may be associated with fresh fruits and vegetables were discussed by Hedberg et al. (1). Increases in foodborne illness during the summer are not fully understood, although fresh produce is likely to play a role since it is consumed in higher quantities during the summer. The per capita consumption of fresh produce has increased in the United States in recent years (Figure 1), not only in the summer but also in other seasons, partly because of increased importation. Knowledge of the presence and numbers of specific pathogens on produce imported to the United States from countries that may have lower sanitation standards is minimal. However, produce from a single grower, packinghouse, or shipper, whether located outside or within the United States, may be routinely distributed throughout the country, thus facilitating widespread dissemination of potential pathogens. The epidemiology of foodborne diseases is greatly influenced by these global changes. Control or elimination of pathogenic microorganisms from fresh fruits and vegetables can be achieved only by addressing the entire system, from the field, orchard, or vineyard to the point of consumption.
We reviewed some of the practices (particularly preharvest practices) used by the fresh fruit and vegetable industry that may promote contamination of produce with pathogenic microorganisms.
The presence of pathogenic bacteria, viruses, and parasites on fresh fruits and vegetables has been extensively documented (3). Contamination of produce can occur in the field or orchard; during harvesting, postharvest handling, processing, shipping, or marketing; or in the home (Table).
Preharvest Sources
Spores of Clostridium species, including C. botulinum and C. perfringens, as well as spores of enterotoxigenic Bacillus cereus, are commonly found in soil, so their occasional presence on fruits and vegetables should not be unexpected. Numbers of clostridial spores on some types of vegetables appear to increase during the summer (4). Perhaps the most prevalent disease-causing microorganism in soil is Listeria monocytogenes (5,6). Twenty-seven strains were isolated from soil and vegetation taken from 19 sites in the Netherlands (7). Plant materials from which the organism was isolated included dead and decayed corn and soybean plants and wild grasses, indicating its preference to exist in nature as a saprophyte. A study of soil and domestic animal feces has shown that Listeria is more often present during July to September than other months (8). L. monocytogenes and L. innocua were predominant in feces, whereas L. ivanovi and L. seeligeri were most common in soil.
Vegetation in a rural area in Virginia where clinical listeriosis is rare was analyzed for L. monocytogenes (9). Dead soybean plant material and stalks, leaves, and tassels of corn were collected in April following the previous planting year. Eight of twelve sampling sites yielded plant materials positive for L. monocytogenes. Only 25% of the strains were pathogenic for mice, a low frequency compared with the percentage of pathogenic strains isolated from Listeria-positive humans and animals in Virginia and the United States as a whole. These observations suggest that the predominance of certain serotypes of L. monocytogenes may be influenced by the environment and that some strains indigenous to decaying plant vegetation are incapable of causing human illness.
Weiss and Seeliger (10) isolated 154 strains of L. monocytogenes in Germany from soil and plants, 16 from feces of deer and stag, nine from moldy fodder and wildlife feeding grounds, and eight from birds. Corn, wheat, oats, barley, and potato plants and soils from the fields in which they were growing were among the materials analyzed. Nearly 10% of the corn plants and 13% of the grain plants were infected with L. monocytogenes. Plants from cultivated fields had a lower incidence (12.5%) than plants from uncultivated fields (44%). Twenty-three percent of samples collected from wildlife feeding grounds were positive for L. monocytogenes. It was suggested that L. monocytogenes is a saprophyte that lives in a plant-soil environment and could therefore be contracted by humans and animals through many possible routes from many sources. Birds and animals are unlikely to be the only sources responsible for the distribution of L. monocytogenes in nature and its presence on fruits and vegetables.
The presence of other pathogenic bacteria, viruses, and parasites in soil likely results largely from application of feces or untreated sewage, either by chance or design. Whatever the case, soil on the surface of fruits and vegetables may harbor pathogenic microorganisms that remain viable through subsequent handling to the point of consumption unless effective sanitizing procedures are administered.
Irrigation and surface run-off waters can be sources of pathogenic microorganisms that contaminate fruits and vegetables in the field. Irrigation water containing raw sewage or improperly treated effluents from sewage treatment plants may contain hepatitis A, Norwalk viruses, or enteroviruses (poliomyelitis, echoviruses, and Coxsackie viruses) (11). Rotaviruses are known to retain viability on the surface of vegetables held at 4°C for up to 30 days (12).
Listeria and other potentially pathogenic bacteria have been reported in sewage. Watkins and Sleath (13) analyzed 52 sewage, river water, and industrial effluents for pathogens. Effluents were from abattoirs, cattle markets, and poultry packing plants. L. monocytogenes was isolated from all samples. In many instances, populations of L. monocytogenes were higher than those for salmonellae and, in some instances, L. monocytogenes was isolated when no salmonellae were detected. Application of sludge containing L. monocytogenes and salmonellae to soil showed that L. monocytogenes could survive longer. Populations of L. monocytogenes in soil remained essentially unchanged during 7 weeks after application.
Treatment of sewage does not always yield a sewage sludge cake or a final discharge free of Listeria (14). The use of sewage as a fertilizer could contaminate vegetation destined for human consumption. MacGowan et al. (8) examined sewage at 2-month intervals in 1991 to 1992 and found 84% to 100% contained L. monocytogenes or L. innocua.
Application of sewage sludge or irrigation water to soil is one avenue through which parasites can contaminate fruits and vegetables. Ascaris ova sprayed onto tomatoes and lettuce remain viable for up to 1 month, while Endamoeba histolytica could not be recovered 1 week after spraying (15,16). If sewage irrigation or night soil application is stopped 1 month before harvest, the produce would not likely be vectors for transmission of diseases caused by these parasites.
Wang and Dunlop (17,18) recovered Salmonella, Ascaris ova, and Endamoeba coli cysts from more than half of irrigation water samples contaminated with either raw sewage or primary-treated, chlorinated effluents. Only one of 97 samples of vegetables irrigated with this water yielded Salmonella, but Ascaris ova were recovered from two of 34 vegetable samples. Barbier et al. (19) concluded that application of sewage sludge containing Taenia saginata eggs offers a serious risk for cattle even after a 3-week no-grazing period.
Feces have been suspected as sources of pathogens on contaminated fruits, vegetables, or minimally processed produce that have subsequently been associated or confirmed as causes of human disease outbreaks (3). Among the more recent outbreaks are those linking unpasteurized apple juice to Escherichia coli O157:H7 infections. This pathogen can remain viable in bovine feces for up to 70 days, depending on inoculum level and temperature (17). Cryptosporidium infection linked to consumption of unpasteurized apple juice was hypothesized to have been caused by contamination of apples by calf feces (20). Contact of fruits and vegetables by pickers and handlers at the time of harvest also offers a mechanism by which pathogens in feces can contaminate raw produce.
Wild birds are known to disseminate Campylobacter (21,22), Salmonella (22,23), Vibrio cholerae (24), and Listeria species (25). More recently, E. coli O157:H7 has been isolated from wild bird feces. In a survey of wild birds (mainly gulls), 0.9% of the bacterial isolates from fecal samples at an urban landfill and 2.9% of bacterial isolates from fecal samples on intertidal sediments were Vero cytotoxin-producing E. coli O157:H7 (26). Pathogenic bacteria are apparently picked up as a result of birds feeding on garbage, sewage, fish, or lands that are grazed with cattle or have had applications of fresh manure. Control of preharvest contamination of fruits and vegetables with pathogenic bacteria by wild birds would be exceptionally difficult.
Postharvest Sources
Some of the possible preharvest sources of pathogenic microorganisms may also be postharvest sources (Table). The fecal-oral route of transmission of pathogens broadens to include workers handling fruits and vegetables from the point of removal from the plant through all stages of handling, including preparation at the retail and food service levels and in the home. Changes in eating habits, particularly the increased consumption of meals away from home, must be considered when attempting to provide reasons for increased frequency of outbreaks associated with fresh produce. Proper training of food-service workers in hygienic practices is essential. One cannot assume that newly hired personnel have even rudimentary knowledge of food microbiology. This is particularly critical among teenagers who, partly because they and their parents are eating more meals away from home, have had minimal or no exposure to proper food-handling practices. Instruction in elementary principles of food hygiene at the high school or middle school levels has diminished greatly in the past two decades.
Traditionally recognized postharvest control points for access of pathogens to whole or cut produce include transport containers and vehicles and sorting, packing, cutting, and further processing equipment. The development of new processing equipment and technologies should include a team of experts in food microbiology as well as engineering. Too often, aspects of sanitizing equipment are not considered or are an afterthought and can increase the risk for contaminated end products. Temperature control is absolutely critical at every stage of postharvest handling if any success is to be achieved in minimizing the growth of pathogens.
Sanitizers that can be used to wash or to assist in lye peeling of fruits and vegetables are regulated by the U.S. Food and Drug Administration in accordance with the Federal Food, Drug and Cosmetic Act as outlined in the Code of Federal Regulations, Title 21, Ch. 1, Section 173.315. As noted by Barmore (27), no chlorine substitute effective for washing fruits and vegetables is available. Numerous alternatives for sanitizing equipment (28) can be used in a total sanitation program, but none has as broad a spectrum of activity as chlorine.
Chlorine is routinely used as a sanitizer in wash, spray, and flume waters used in the fresh fruit and vegetable industry. Antimicrobial activity depends on the amount of free available chlorine (as hypochlorous acid) in water that comes in contact with microbial cells. The efficacy of chlorine in killing pathogenic microorganisms has been extensively studied. Possible uses in packinghouses and during washing, cooling, and transport to control postharvest diseases of whole produce have been reviewed by Eckert and Ogawa (29). The effect of chlorine concentration on aerobic microorganisms and fecal coliforms on leafy salad greens was studied by Mazollier (30). Total counts were markedly reduced with increased concentrations of chlorine up to 50 ppm, but a further increase in concentration up to 200 ppm did not have an additional substantial effect. A standard procedure for washing lettuce leaves in tap water was reported to remove 92.4% of the microflora (31). Including 100 ppm available free chlorine in wash water reduced the count by 97.8%. Adjusting the pH from 9 to 4.5 to 5.0 with inorganic and organic acids resulted in a 1.5- to 4.0-fold increase in microbicidal effect. Increasing the washing time in hypochlorite solution from 5 to 30 minutes did not decrease microbial levels further, whereas extended washing in tap water produced a reduction comparable to hypochlorite. The addition of 100 ppm of a surfactant (Tween 80) to a hypochlorite washing solution enhanced lethality but adversely affected sensory qualities of lettuce.
Dipping Brussels sprouts into chlorine solution (200 ppm) for 10 seconds decreased the number of viable L. monocytogenes cells by about 2 log10 CFU/g (32). The maximum log10 reduction of L. monocytogenes on shredded lettuce and cabbage treated with 200 ppm chlorine for 10 minutes was 1.3 to 1.7 log10 CFU/g and 0.9 to 1.2 log10 CFU/g, respectively (12). Numbers decreased only marginally with increased exposure time from 1 to 10 minutes, which agrees with observations made by Brackett (32) that the action of chlorine against L. monocytogenes occurs primarily during the first 30 seconds of exposure. Nguyen-the and Carlin (33) concluded that the elimination of L. monocytogenes from the surface of vegetables by chlorine is unpredictable and limited.
Populations of Salmonella Montevideo on the surface and in the stem core tissue of tomatoes can be substantially reduced by dipping fruits 2 minutes in a solution containing 60 or 110 ppm chlorine, respectively; however, treatment in a solution containing 320 ppm chlorine does not result in complete inactivation (34). The ineffectiveness of 100 ppm chlorine against S. Montevideo injected into cracks in the skin of mature green tomatoes was demonstrated by Wei et al. (35). Treatment of alfalfa seeds injected with Salmonella Stanley (102 to 103 CFU/g) in 100 ppm chlorine solution for 10 minutes has been reported to cause a substantial reduction in population, and treatment in 290 ppm chlorine solution resulted in a substantial reduction compared with treatment with 100 ppm chlorine (36). Initial free chlorine concentrations up to 1,000 ppm, however, did not result in further reductions. Treatment of seeds containing 101 to 102 CFU/g of S. Stanley for 5 minutes in a solution containing 2,040 ppm chlorine reduced the population to less than 1 CFU/g.
We have studied the efficacy of chlorine, hydrogen peroxide, and ethanol in removing Salmonella from injected alfalfa sprouts. Sprouts were dipped in solutions containing 200, 500, or 2,000 ppm chlorine for 2 minutes. The pathogen was reduced by about 2 log10 CFU/g after treatment with 500 ppm chlorine, compared with the control, and to an undetectable level (<1 CFU/g) after treatment with 2,000 ppm chlorine (Figure 2). Chlorine treatment (2,000 ppm) of cantaloupe cubes injected with the same five-serotype cocktail of Salmonella resulted in less than 1 log10 reduction in viable cells (Figure 2). The very high level of organic matter in the juice released from cut cantaloupe tissue apparently neutralizes the chlorine before its lethality can be manifested.
As noted by Lund (37), the inaccessibility of chlorine to microbial cells in crevices, creases, pockets, and natural openings in the skin also undoubtedly contributes to the overall lack of effectiveness of chlorine in killing pathogens. The hydrophobic nature of the waxy cuticle on tissue surfaces protects surface contaminants from exposure to chlorine and other produce sanitizers that do not penetrate or dissolve these waxes. Surface-active agents lessen the hydrophobicity of fruit and vegetable skins as well as the surfaces of edible leaves, stems, and flowers, but they may also cause deterioration of sensory qualities (31,37). Sanitizers that contain a solvent that would remove the waxy cuticle layer, and with it enmeshed contaminants, without adversely affecting sensory characteristics would hold greater potential than chlorinated water in reducing microbial populations on whole raw produce. Such sanitizers may be limited to use on produce that will be further processed into juice or cut products, or on whole fruits, vegetables, or plant parts destined for immediate consumption, since their application could adversely affect visual appearance. Clearly, chlorine, at concentrations currently permitted for use by the industry to wash fresh fruits and vegetables, cannot be relied upon to eliminate pathogens.
References
- Hedberg CW, MacDonald KL, Osterholm MT. Changing epidemiology of food-borne disease: a Minnesota perspective. Clin Infect Dis. 1994;18:671–82.PubMedGoogle Scholar
- United States Department of Agriculture. Vegetables and specialties/VGS-269/July. Fruits and tree nuts/FTS-278/October. Washington (DC): USDA Economic Research Service; 1996. p. 22,81.
- Beuchat LR. Pathogenic microorganisms associated with fresh produce. J Food Prot. 1996;59:204–6.
- Ercolani GL. Occurrence and persistence of culturable clostridial spores on the leaves of horticultural plants. J Appl Microbiol. 1997;82:137–40. DOIGoogle Scholar
- Beuchat LR. Listeria monocytogenes: incidence in vegetables. Food Contr. 1996;7:223–8. DOIGoogle Scholar
- Welshimer HJ. Survival of Listeria monocytogenes in soil. J Bacteriol. 1960;80:316–20.PubMedGoogle Scholar
- Welshimer HJ, Donker-Voet J. Listeria monocytogenes in nature. Appl Environ Microbiol. 1971;21:516–9.
- MacGowan AP, Bowker K, McLauchlin J, Bennett PM, Reeves DS. The occurrence and seasonal changes in the isolation of Listeria spp. in shop bought food stuffs, human feces, sewage and soil from urban sources. Int J Food Microbiol. 1994;21:325–34. DOIPubMedGoogle Scholar
- Welshimer HJ. Isolation of Listeria monocytogenes from vegetation. J Bacteriol. 1968;95:300–3.PubMedGoogle Scholar
- Weiss J, Seeliger HPR. Incidence of Listeria monocytogenes in nature. Appl Microbiol. 1975;29:29–32.
- Bagdasargan GA. Survival of viruses of the enterovirus group (poliomyelitis, ECHO, Coxsackie) in soil and on vegetables. J Hyg Epidemiol Microbiol Immunol. 1964;8:497–505.
- Badaway AS, Gerba CP, Kelly LM. Survival of rotavirus SA-11 on vegetables. Food Microbiol. 1985;2:199–205. DOIGoogle Scholar
- Watkins J, Sleath KP. Isolation and enumeration of Listeria monocytogenes from sewage sludge and river water. J Appl Bacteriol. 1981;50:1–9.PubMedGoogle Scholar
- Al-Ghazali MR, Al-Azawi SK. Detection and enumeration of Listeria monocytogenes in a sewage treatment plant in Iraq. J Appl Bacteriol. 1986;60:251–4.PubMedGoogle Scholar
- Rudolfs W, Falk LL, Ragotzkie RA. Contamination of vegetables grown in polluted soil. III. Field studies on Ascaris eggs. Sewage Ind Waste. 1951;23:656–60.
- Rudolfs W, Falk LL, Ragotzkie RA. Contamination of vegetables grown in polluted soil. II. Field and water studies on Endamoeba cysts. Sewage Ind Waste. 1951;23:478–85.
- Wang G, Zhao R, Doyle MP. Fate of enterohemorrhagic Escherichia coli O157:H7 in bovine feces. Appl Environ Microbiol. 1996;62:2567–70.PubMedGoogle Scholar
- Dunlop SG, Wang W-LL. Studies on the use of sewage effluent for irrigation of truck crops. J Food Prot. 1961;24:44–7.
- Barbier D, Perrine D, Duhamel C, Doublet R, Georges P. Parasitic hazard with sewage sludge applied to land. Appl Environ Microbiol. 1990;56:1420–2.PubMedGoogle Scholar
- Millard PG, Gensheimer KF, Addiss DG, Sosin DM, Beckett GA, Houck-Jankoski A, An outbreak of cryptosporidiosis from fresh-pressed apple cider. JAMA. 1994;272:1592–6. DOIPubMedGoogle Scholar
- Luechtefeld N, Blaser M, Reller L, Wang W. Isolation of Campylobacter fetus subsp. jejuni from migratory wildfowl. J Clin Microbiol 1980;12:406-8. Quessy S, Messier S. Campylobacter spp. and Listeria spp. in ring-billed gulls (Larus delawarensis). J Wildl Dis. 1992;28:526–1.PubMedGoogle Scholar
- Jones F, Smith P, Watson DC. Pollution of a water supply catchment by breeding gulls and the potential environmental health implications. J Inst Water Eng Sci. 1978;32:469–82.
- Lee JV, Basford D, Donovan T, Furniss A, West D. The incidence of Vibrio cholerae in water, animals and birds in Kent, England. J Appl Bacteriol. 1982;52:281–91.PubMedGoogle Scholar
- Fenlow DR. Wild birds and silage as reservoirs of Listeria in the agricultural environment. J Appl Bacteriol. 1985;59:537–44.PubMedGoogle Scholar
- Wallace JS, Cheasty T, Jones K. Isolation of Vero cytotoxin-producing Escherichia coli O157:H7 from wild birds. J Appl Microbiol. 1997;82:399–404. DOIPubMedGoogle Scholar
- Barmore CR. Chlorineare there alternatives? Cutting Edge 1995; Spr 1995; 4-5.
- Cords BR, Dychdala GR. Sanitizers: halogens, surface-active agents and peroxides. In: Davidson PM, Branen AL, editors. Antimicrobials in Foods. 2nd ed. New York: Marcel Dekker, Inc.; 1993. p. 469-537.
- Eckert JW, Ogawa JM. The chemical control of postharvest diseases: deciduous fruits, berries, vegetables and root/tubers crops. Annu Rev Phytopathol. 1988;26:433–63. DOIGoogle Scholar
- Mazollier J. Ivè gamme. Lavage-desinfection des salades. Infros-Crifl. 1988;41:19.
- Adams MR, Hartley AD, Cox LJ. Factors affecting the efficiency of washing procedures used in the production of prepared salads. Food Microbiol. 1989;6:69–77. DOIGoogle Scholar
- Brackett RE. Antimicrobial effect of chlorine on Listeria monocytogenes. J Food Prot. 1987;50:999–1003.
- Nguyen-the C, Carlin F. The microbiology of minimally processed fresh fruits and vegetables. Crit Rev Food Sci Nutr. 1994;34:371–401. DOIPubMedGoogle Scholar
- Zhuang R-Y, Beuchat LR, Angulo FJ. Fate of Salmonella montevideo on and in raw tomatoes as affected by temperature and treatment with chlorine. Appl Environ Microbiol. 1995;61:2127–31.PubMedGoogle Scholar
- Wei CI, Huang TS, Kim JM, Lin WF, Tamplin ML, Bartz JA. Growth and survival of Salmonella montevideo on tomatoes and disinfection with chlorinated water. J Food Prot. 1995;58:829–36.
- Jaquette CB, Beuchat LR, Mahon BE. Efficacy of chlorine and heat treatment in killing Salmonella stanley inoculated onto alfalfa seeds and growth and survival of the pathogen during sprouting and storage. Appl Environ Microbiol. 1996;62:2212–5.PubMedGoogle Scholar
- Lund BM. Bacterial spoilage. In: Dennis C, editor. Post-harvest pathology of fruits and vegetables. London: Academic Press; 1983. p. 219.
- Zhang S, Farber JM. The effects of various disinfectants against Listeria monocytogenes on fresh-cut vegetables. Food Microbiol. 1996;13:311–21. DOIGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 3, Number 4—December 1997
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Larry R. Beuchat, Center for Food Safety and Quality Enhancement, University of Georgia, Griffin, Georgia 30223 USA; fax: 770-229-3216
Top