Volume 30, Number 1—January 2024
Dispatch
Macacine alphaherpesvirus 1 (B Virus) Infection in Humans, Japan, 2019
Cite This Article
Citation for Media
Abstract
Two human patients with Macacine alphaherpesvirus 1 infection were identified in Japan in 2019. Both patients had worked at the same company, which had a macaque facility. The rhesus-genotype B virus genome was detected in cerebrospinal fluid samples from both patients.
The herpesvirus Macacine alphaherpesvirus 1 (herpes B virus, or B virus) is ubiquitous in macaque monkeys. Although macaque monkeys do not usually show symptoms when infected with B virus, humans show severe disease, including encephalitis and encephalomyelitis, and death frequently results from infection with B virus from monkeys (1). However, B virus infection of humans is rare. Infection can occur after being bitten or scratched by a macaque monkey that is actively shedding the virus or through direct contact with bodily fluids or contaminated laboratory materials. Since B virus infection was first reported in 1934, more than 50 cases have been reported, mainly in North America, and 29 cases have been confirmed, including a recent case in China (2,3). In most cases in which a specific macaque species was identified, patients had been exposed to rhesus macaques, rather than other species of monkey (e.g., cynomolgus macaques, African green monkeys, Vervet monkeys, or Sykes monkeys) (2).
In this study, 2 patients in Japan with chronic and long-term neurologic diseases were tested for B virus infection; the B virus genome was detected in cerebrospinal fluid (CSF). Both patients had worked at a macaque facility in Japan. To maintain confidentiality and privacy, we report no personal information, or information about the clinical course or the working environment. All protocols and procedures were approved by the research ethics committee of the National Institute of Infectious Diseases for the use of human subjects (approval no. 1314). We confirmed B virus infection in both patients by using molecular assay and, in one patient, by also using immunohistochemical analysis. We describe molecular and immunohistochemical findings in the 2 patients.
Patient 1 worked in the macaque facility at a pharmaceutical research company. In 2019, the patient was hospitalized for headache, fever, and deterioration of consciousness. CSF samples were collected at the time of hospitalization and sent to the National Institute of Infectious Diseases (Tokyo, Japan), where we tested them for B virus infection. We extracted total DNA from the CSF samples and tested the samples by using real-time PCR with primers and a fluorescent probe targeting the B virus gB gene: forward primer, 5′- CGTGGCCAGGTAGTACTGCAC-3′; reverse primer, 5′- CTCGTTCCGCTTCTCCTCGTC-3′); AND fluorescent-labeled probe, 5′- FAM-TAGCGCCGGAGGAA-MGB-3′. The reaction mixture (total volume 25 µL) contained 12.5 µL TaqMan Universal PCR Master Mix (ThermoFisher, https://www.thermofisher.com), 2.0 µg/mL of sonicated salmon sperm DNA, 0.2 µmol/L of each primer and fluorescein amidite–labeled probe, and 4.0 µL of extracted DNA. We subjected the reaction mixture to real-time PCR by using an ABI-7500 Fast Real-Time PCR System (ThermoFisher). The reaction conditions were as follows: 50°C for 2 min and 95°C for 10 min, followed by 40 cycles of 95°C for 15 s and 60°C for 60 s. We performed real-time PCR targeting the B virus gG gene as described previously (4). We performed conventional PCR by using primers targeting a gB gene region conserved among primate herpes viruses (herpes-PCR) as described previously (5). We detected the gB and gG genes in the CSF samples at a concentration of 5.1 × 105 copies/mL for gB and 7.6 × 105 copies/mL for gG. Sequencing of the 364-bp herpes-PCR product revealed 100% identity with a B virus sequence from GenBank (accession no. LC637778).
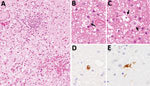
Patient 2 had been diagnosed with chronic neurologic disease. The patient was tested for B virus infection because they worked at the same facility as patient 1 and had been working with macaques. In 2014, patient 2 was admitted to hospital with fever, headache, and neurologic symptoms. Brain biopsy was performed, but no pathogen was detected at that time. In 2019, after identification of patient 1, we collected a CSF sample and tested it for B virus infection in addition to testing a paraffin-embedded section of the brain biopsy collected in 2014. Real-time PCR of the CSF detected the gB (3.5 × 105 copies/mL) and gG genes (2.0 × 106 copies/mL) of B virus. Nucleotide sequencing of the 364-bp herpes-PCR product confirmed that it had 100% identity with the sequence detected in patient 1. Furthermore, real-time PCR of the DNA extracted from the paraffin-embedded section of brain tissue biopsied in 2014 was positive for the B virus gG gene. Real-time PCR did not detect herpes simplex virus 1, herpes simplex virus 2, or varicella zoster virus. Histologic analysis of the brain biopsy revealed inflammatory cell infiltration and hemorrhage in the white matter of the cerebellum (Figure 1). Careful observation revealed the presence of inclusion bodies in the nuclei. Immunohistochemical analysis using B virus rabbit polyclonal and gB mouse monoclonal antibodies generated positive signals in cells with inclusion bodies (6).
From our results, we concluded that patients 1 and 2 had been infected with B virus. Although the 2 patients worked in the facility managing imported macaques, there was no direct evidence that they were infected from imported macaques. Given that no epidemiologic link between the 2 patients had been recorded, it seems that they were infected independently. Nucleotide sequence analysis identified B virus genotypes known to be carried by the macaque species (7,8). Recently, a case of human B virus infection was identified in China; however, the genotype was not identified (3). Phylogenetic analysis of the gB gene indicated that B virus from the 2 patients in Japan clustered with the genotype in rhesus macaques (Figure 2). The full-length nucleotide sequences of the gG, gB, and gJ genes also were obtained from the CSF of patient 2 and also were classified by phylogenetic analysis as the rhesus B virus genotype (data not shown). Although there is no direct epidemiologic evidence of zoonotic transmission of rhesus B virus in the monkey facility, the results indicated that workers in such facilities are at risk for infection with rhesus B virus infection, which is pathogenic in humans.
Dr. S. Yamada is a senior scientist in the Laboratory of Herpesviruses, Department of Virology 1, National Institute of Infectious Diseases, Japan. His research interests include virology of herpes simplex virus, human cytomegalovirus, and B virus.
Acknowledgment
This research was supported by AMED (grant nod. JP19fk0108097 and JP23fk0108634) and the Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (grant nos. 20K06404 and 21K05967).
References
- Huff JL, Barry PA. B-virus (Cercopithecine herpesvirus 1) infection in humans and macaques: potential for zoonotic disease. Emerg Infect Dis. 2003;9:246–50. DOIPubMedGoogle Scholar
- Jones-Engel E. Low incidence, high lethality or higher incidence, lower lethality: what we know and don’t know about zoonotic Macacine alphaherpesvirus 1 (monkey B virus) [Chapter 8]. In: Knauf S, Jones-Engel L, editors. Neglected diseases in monkeys. Cham (Switzerland): Springer Nature Switzerland; 2020. p. 171–204.
- Wang W, Qi W, Liu J, Du H, Zhao L, Zheng Y, et al. First human infection case of monkey B virus identified in China, 2021. China CDC Wkly. 2021;3:632–3. DOIPubMedGoogle Scholar
- Perelygina L, Patrusheva I, Manes N, Wildes MJ, Krug P, Hilliard JK. Quantitative real-time PCR for detection of monkey B virus (Cercopithecine herpesvirus 1) in clinical samples. J Virol Methods. 2003;109:245–51. DOIPubMedGoogle Scholar
- Black DH, Eberle R. Detection and differentiation of primate alpha-herpesviruses by PCR. J Vet Diagn Invest. 1997;9:225–31. DOIPubMedGoogle Scholar
- Blewett EL, Black D, Eberle R. Characterization of virus-specific and cross-reactive monoclonal antibodies to Herpesvirus simiae (B virus). J Gen Virol. 1996;77:2787–93. DOIPubMedGoogle Scholar
- Smith AL, Black DH, Eberle R. Molecular evidence for distinct genotypes of monkey B virus (herpesvirus simiae) which are related to the macaque host species. J Virol. 1998;72:9224–32. DOIPubMedGoogle Scholar
- Ohsawa K, Black DH, Torii R, Sato H, Eberle R. Detection of a unique genotype of monkey B virus (Cercopithecine herpesvirus 1) indigenous to native Japanese macaques (Macaca fuscata). Comp Med. 2002;52:555–9.PubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: December 12, 2023
Table of Contents – Volume 30, Number 1—January 2024
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Shuetsu Fukushi, Department of Virology 1, National Institute of Infectious Diseases, 1-23-1, Shinjuku, Tokyo 162-8640, Japan; e-mail:fukushi@niid.go.jp
Top