Volume 32, Number 4—April 2026
Research
Accelerated Increase in Candida auris Bloodstream Infections during COVID-19 Pandemic, South Africa
Cite This Article
Citation for Media
Abstract
The COVID-19 pandemic coincided with rising secondary bloodstream infections (BSIs) from multidrug-resistant organisms, including Candida auris. To assess candidemia trends, we conducted a retrospective analysis of blood culture isolates from public and private laboratories in South Africa taken during January 2019–June 2022. We evaluated weekly aggregated Candida BSI counts and COVID-19 cases using segmented regression within an interrupted time-series framework. In total, 15,393 candidemia cases were identified, 70% from the private sector. C. parapsilosis accounted for 39% of cases, whereas C. auris represented 26%. The proportion of C. auris increased significantly from 17% in 2019 to 31% in 2021 (p<0.01). After the pandemic onset, Candida BSIs rose by 11 cases per week (p = 0.03), largely driven by C. auris (+5 cases/week; p<0.01); peaks coincided with COVID-19 waves. Those results highlight an accelerated shift toward C. auris in Candida BSIs and the urgent need for enhanced surveillance, diagnostics, and infection prevention.
The COVID-19 pandemic, caused by the novel respiratory virus SARS-CoV-2, created strains on the global healthcare system that were experienced mainly in the delivery of acute hospital services (1,2). Those challenges led to secondary healthcare-associated bloodstream infections (BSIs) caused by multidrug-resistant bacteria and fungi, partially related to increased background use of antimicrobial agents, steroids, and immunomodulatory agents (3–5). In addition, viral-induced immune-mediated damage by SARS-CoV-2 might have encouraged bacterial and fungal colonization (6).
Candida BSI is a growing concern worldwide. More than 90% of Candida BSIs are caused by 1 of 6 species: C. albicans, Nakaseomyces glabratus (previously C. glabrata), C. tropicalis, C. parapsilosis, Pichia kudriavzevii (previously C. krusei), and, increasingly, C. auris (7). Those 6 species are also included in the World Health Organization Fungal Priority Pathogen List (8). In the EUROBACT-2 international cohort study, fungal pathogens accounted for ≈8% (230/2,927) of all healthcare-associated BSIs from 2,600 enrolled patients in 333 intensive care units in 52 countries during June 2019–January 2020 (8). Candida species were the most common fungi; 58% (133/230) were identified as non–C. albicans Candida species (9). Candida spp. was the third most common healthcare-associated BSI pathogen (≈12% [333/2,828]) in a prepandemic multicenter study from India (10).
The emergence of C. auris in South Africa was first recognized retrospectively from a BSI isolate collected in 2009, which was initially misidentified as C. haemulonii because of limitations in standard diagnostic methods (11). National surveillance during October 2012–November 2016 identified a total of 1,692 cases of confirmed or probable C. auris infections across South Africa; cases were concentrated in Gauteng Province, where 92% of cases were reported from private-sector hospitals (11). Nationally, the number of cases rose exponentially from 18 in the baseline period (October 2012–November 2013) to 861 in the corresponding final period (October 2015–November 2016) (11). By 2016–2017, C. auris was the third most common cause of BSI nationally, surpassing most other non–C. albicans species (12). Molecular epidemiology revealed that isolates in South Africa predominantly belonged to the African clade III, which is genetically distinct and almost universally resistant to fluconazole; ≈6% had reduced susceptibility to amphotericin B (MIC >2 µg/mL) (13). Of note, 5% of isolates were resistant to both fluconazole and amphotericin B (13).
The COVID-19 pandemic coincided with a marked increase in C. auris outbreaks worldwide, particularly in intensive care settings (14). Several contributing factors were identified, including prolonged mechanical ventilation, widespread antimicrobial use, increased indwelling device exposure, and compromised infection prevention protocols (14). A systematic review (14) documented C. auris outbreaks in >10 countries, including India, Brazil, Mexico, Pakistan, the United States, and multiple countries in Europe; reported crude mortality rates among COVID-19 co-infected patients ranged from 30% to 72%. Evidence from Orange County, California, USA, further illustrated this trend (15); in long-term acute-care hospitals, the probability of new C. auris colonization during the first COVID-19 wave reached 22.5% (95% CI 18.5–26.6) within 30 days of admission. That increase coincided with widespread disruptions in infection prevention practices and shortages of protective equipment, and although incidence declined in later phases of the pandemic, the overall prevalence of colonization remained high (15). Schaefer et al. (16) reported that C. auris incidence at a major academic hospital in New York increased nearly 3-fold, from 2.6 cases/10,000 admissions in 2019 to 7.8 cases/10,000 admissions in 2022 (16). Similarly, national surveillance data from China revealed 1,846 laboratory-confirmed C. auris cases across 22 provinces; ≈80% of isolates were resistant to fluconazole and ≈7% were resistant to amphotericin B (17).
In this study, we aimed to evaluate the distribution of Candida species isolated from blood cultures from both private and public health sectors in South Africa and to describe changes during the COVID-19 pandemic. Permission to conduct this study was obtained from the South African Society of Clinical Microbiology and NHLS, and ethics approval was obtained from the Human Research Ethics Committee (Medical) of the University of the Witwatersrand (clearance no. M210752).
Study Design, Population, and Setting
We conducted a secondary analysis of blood culture data archived in pathology laboratory information systems from January 1, 2019, through June 30, 2022, in South Africa. The study population consisted of all patients who had a blood culture submitted either to the National Health Laboratory Service (NHLS) or to 1 of 3 large amalgamated private pathology practices (Ampath [https://www.ampath.co.za], Lancet Laboratories [https://www.lancet.co.za], or PathCare/Vermaak and Partners [https://www.vpath.co.za]). The NHLS provides routine diagnostic pathology services to the public health sector, serving ≈83% of the population of South Africa, and has 60 laboratories that offer microbiology testing, including automated blood culture systems and culture identification (11). The 3 large private pathology groups provide a full range of microbiology testing for almost all inpatients in South Africa with private health insurance (11). The NHLS used several platforms to identify Candida to species level at the time of this study: Vitek 2 YST, API 20C Aux, or API ID 32C (bioMérieux, https://www.biomerieux.com); Auxacolor (Bio-Rad Laboratories, https://www.bio-rad.com); and Microscan (Beckman Coulter, https://www.beckmancoulter.com) (11). The private pathology groups identified Candida primarily using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry methods (Bruker, https://www.bruker.com; or bioMérieux). The total number of blood culture specimens collected during the study period was not available as a denominator. The private health sector represents a small proportion of the general population, and laboratory data from this sector might not be directly comparable with the public sector owing to factors such as specimen-taking practices (18). We included patients with blood cultures positive with any of the 6 most common Candida species (C. albicans, C. auris, N. glabratus, P. kudriavzevii, C. parapsilosis, and C. tropicalis) in this analysis. Blood culture data from NHLS were obtained from the surveillance data warehouse at the National Institute for Communicable Diseases (NICD) (19). Blood culture data from the private sector were obtained through the South African Society of Clinical Microbiology, which has formed a public–private partnership with the NICD (19).
Definitions
We defined a case of Candida BSI was defined as illness in a person seen at a healthcare facility in South Africa who had a blood culture from which C. albicans, C. auris, N. glabratus, P. kudriavzevii, C. parapsilosis, or C. tropicalis was isolated. We regarded positive blood cultures with the same organism that had been collected within 30 days of a first positive blood culture as duplicates and excluded them.
DATCOV and SARS-CoV-2
We extracted COVID-19 hospital admission data from March 5, 2020, through June 30, 2022, from the inpatient national surveillance system, DATCOV. Established in April 2020 by the NICD, DATCOV served as an active surveillance program for COVID-19 hospital admissions for both public and private hospitals in South Africa (1). We extracted routine surveillance data for SARS-CoV-2 infections from the reengineered Notifiable Medical Conditions Surveillance System for the period March 1, 2020, through June 30, 2022. In July 2020, the NICD established the Notifiable Medical Conditions Surveillance System, which contains data for all SARS-CoV-2 tests (PCR or antigen detection) conducted in South Africa (20). January 2019–February 2020 was considered the prepandemic baseline, and March 2020–June 2022 was the COVID-19 pandemic period.
Data Management and Analysis
We reported categorical variables for blood culture data in tables as frequencies and percentages or presented them as bar charts and tables. We used the Pearson χ2 test or Fisher exact test to compare Candida species distribution among years, sectors, and provinces. We performed all analyses using Stata version 15 (StataCorp LLC, https://www.stata.com) or R Studio version 4.5.1 (https://rstudio.com/products/rstudio). We prepared and tabulated blood culture, DATCOV, and SARS-CoV-2 datasets in Microsoft Excel (https://www.microsoft.com). We used those tabulated datasets to plot graphs and apply 30-day moving averages for cases of C. auris and C. parapsilosis BSI. We conducted interrupted time series analysis to assess the effects of the COVID-19 pandemic on the incidence of Candida BSI, with a specific focus on C. auris. We defined the intervention point as the onset of the COVID-19 pandemic in South Africa (i.e., March 2020). We aggregated Candida BSI and COVID-19 case counts by week and scaled COVID-19 count data to enable visual comparison. We then plotted weekly counts to examine trends before and after the interruption. We used segmented regression to estimate changes in the level and slope of Candida BSI incidence associated with the pandemic. We annotated national lockdown levels 1–5 in increasing order of restrictiveness. We used interrupted time series models to evaluate both immediate and gradual changes in weekly Candida BSI rates.
Distribution of Cases of Candida BSI
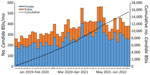
Over the 42-month period, 15,393 cases of Candida BSI were reported: 3,415 cases in 2019, 4,229 cases in 2020, 5,315 cases in 2021 and 2,434 cases during January–June 2022. The private sector accounted for 70% (10,826/15,393) of all cases. Cases of Candida BSI increased across the country, from 4,048 during the 14-month baseline period (January 2019–February 2020) to 5,413 in the first 14 months of the pandemic (March 2020–April 2021), a 34% rise; cases then increased to 5,932 in the following 14-month period (May 2021–June 2022), a further 10% increase (Figure 1). Cases in Gauteng Province accounted for 50% (7,706/15,393) of all Candida BSI cases, followed by KwaZulu-Natal Province (14% [2,163/15,393]) and the Western Cape Province (12% [1,809/15,393]). Similar distribution patterns were observed for those provinces in both private and public sectors (Appendix Table).
C. parapsilosis accounted for 39% (6,080/15,393) of all Candida BSI cases in South Africa, followed by C. auris (26% [3,928/15,393]). Nationally, there were notable differences in the distribution of Candida species. The relative frequencies of cases of C. auris BSI increased from 17% (597/3,415) in 2019 to 31% (1,626/5,315) in 2021 (p<0.01); a corresponding decrease was noted in the percentage of C. parapsilosis, from 46% (1,555/3,415) in 2019 to 37% (1,955/5,315) in 2021 (p<0.01), as well as a percentage decrease for N. glabratus from 10% (332/3,415) in 2019 to 7% (359/5,315) in 2021 (p<0.01). The relative frequencies of C. albicans, P. kudriavzevii, and C. tropicalis were relatively unchanged (Table). C. parapsilosis was the most common species in 8 of the 9 provinces in South Africa. C. auris was the dominant species in the North West Province and accounted for 42% (312/749) of cases (Appendix Figure). In the private sector, C. parapsilosis was the dominant species, accounting for 44% (4,742/10,826) of cases, followed by C. auris, which accounted for 32% (3,507/10,826) of cases. We observed a more marked increase in the percentage of cases of C. auris BSI in the private sector, from 24% (580/2,393) in 2019 to 37% (1,439/3,880) in 2021 (p<0.01); we observed a corresponding decrease in the percentage of C. parapsilosis, from 51% (1,217/2,393) in 2019 to 40% (1,567/3,880) in 2021, with a further decrease to 37% (629/1,684) in 2022 (p<0.01). In the public sector, C. albicans was the dominant species, accounting for 41% (1,883/4,567) of cases, followed by C. parapsilosis, which accounted for 29% (1,338/4,567) of cases. Similar to the private sector, the percentage of cases of C. auris BSI in the public sector increased substantially, from 2% (17/1,022) in 2019 to 13% (187/1,435) in 2021 (p<0.01); we noted a corresponding decrease in the percentage of C. parapsilosis, from 33% (338/1,022) in 2019 to 27% (388/1,435) in 2021 (p<0.01), as well as a decrease in N. glabratus BSIs, from 16% (160/1,022) in 2019 to 12% (169/1,435) in 2021 (p = 0.01) (Table).
Of the 15,393 total cases, 54% (8,333) of patients were male and 46% (7,060) female. The highest number of cases of Candida BSI were seen in persons <1 year of age (17% [2,561]), 51–60 years of age (16% [2,480]) and 61–70 years of age (15% [2,348]). When stratified by health sector, 38% (1,745/4,567) of cases in the public sector were in persons <1 year of age, whereas 40% (4,316/10,826) of cases from the private sector were in persons 51–70 years of age (Figure 2, panels A, B). With the exception of 2 age categories (21–30 years of age and 51–60 years of age) in the private sector, we observed distinct differences among the other age categories across all 4 years. We observed that 74% (201/273) of persons <1 year of age with a C. auris BSI were from the public sector. In contrast, almost all C. auris BSI cases in persons 51–60 years of age (98% [821/834]) and in persons 61–70 years of age (99% [769/775]) were reported from the private sector (Figure 2, panels C, D).
Distribution of Cases of Candida BSI before and during the COVID-19 Pandemic
The first laboratory-confirmed case of SARS-CoV-2 infection was documented in South Africa on March 5, 2020. During the study period, 3,995,881 cases of COVID-19 were reported; 486,789 of those resulted in hospital admission. More than half of COVID-19 hospital admissions (52% [254,975/486,789]) occurred in the public sector.
During the pandemic period (March 2020–June 2022), the number of COVID-19 cases and admissions increased together; the admission peak lagged behind the case peak for each major COVID-19 wave. We observed a temporally related increase in the 30-day moving average of Candida BSI cases after each of the 4 waves caused by the ancestral SARS-CoV-2 and 3 variants, Beta, Delta, and Omicron BA.1, in South Africa during the study period. The monthly average number of Candida BSI cases was 419 in August 2020, 487 in February 2021, 563 in August 2021, and 438 in April 2022. We also observed fluctuations of the 30-day moving average in Candida BSI in the prepandemic period. In the public sector, an increase in the Candida BSI 30-day moving average was only noted after the second COVID-19 wave, caused by the Beta variant; a monthly average of 138 cases was reported in February 2021 (Figure 3).
Using C. parapsilosis (the most common species in the private sector and second most common species in the public sector) as a comparator, we found that, as the COVID-19 pandemic progressed from March 2020 to June 2022, the average number of cases of C. auris BSI increased to the point where C. parapsilosis and C. auris had similar 30-day moving averages. That phenomenon occurred in both the private and public health sectors, although in the private sector, by the end of the study period, C. auris increased from an average of 2 cases/day in February 2019 to 3 cases/day in June 2022 (Figure 4).
Interrupted Time Series Analysis
Before the COVID-19 pandemic (January 2019–February 2020), the weekly trend in Candida BSI was gradually increasing, and the slope before the pandemic was significantly positive (cases increasing by 0.038/week before March 2020; p = 0.02). The onset of the pandemic in March 2020 was associated with a significant increase in Candida BSI cases (immediate jump of 11 cases/week; p = 0.03). However, the postintervention slope for Candida BSIs did not differ significantly from the prepandemic slope (cases decreasing by 0.023/week; p = 0.20). Peaks in Candida BSI were temporally aligned with each of the 4 major COVID-19 waves in South Africa (Figure 5). C. auris alone showed a different pattern. Before the COVID-19 pandemic, cases of C. auris were increasing, but not significantly (cases increasing by 0.010/week before March 2020; p = 0.37). After the pandemic began, C. auris exhibited a sharp immediate increase (C. auris cases jumped by 5/week; p<0.01) and continued to increase after March 2020 (cases increasing by 0.032/week; p<0.01) (Figure 6).
After the pandemic, C. auris cases of candidemia immediately increased sharply and continued to increase. One quarter of all cases of BSI in South Africa were caused by C. auris; more cases of C. auris infection in infants were diagnosed in the public sector, and more cases in older age groups were diagnosed in the private sector.
We observed a substantial increase in cases of Candida BSI after the peak of COVID-19 hospital admissions. Overall, this trend was similar across all 4 COVID-19 waves and most pronounced for the Delta wave in the Southern Hemisphere in winter of 2021. Fungal infections are a substantial concern in managing patients infected with SARS-CoV-2 (21). Our study showed that the number of cases of C. auris increased and were similar in monthly averages to cases of C. parapsilosis, particularly in the private sector. C. parapsilosis and C. auris are both species known to cause invasive infections because of their ability to contaminate the hospital environment, form biofilms on medical devices, colonize the hands of healthcare workers, and contribute to healthcare-associated outbreaks (14,21). Outbreaks are more likely to occur when hospital services are strained, as occurred during the height of the pandemic.
Although C. albicans is a common colonizer of the skin and mucous membrane, a virulent species, and causes a high proportion of invasive infections, non–C. albicans Candida species have greater antifungal drug resistance (22,23). In South Africa, the species causing infection might be used as a proxy for azole resistance, particularly for C. auris, N. glabratus, P. kudriavzevii, and C. parapsilosis. For instance, C. albicans isolated from patients with BSI have the lowest incidence of azole resistance, whereas N. glabratus has decreased susceptibility across the azole class (24). On the basis of national laboratory-based Candida BSI surveillance data in South Africa from 2009–2010, almost 25% of isolates were resistant to fluconazole and 10% were resistant to voriconazole (25). At a tertiary care hospital in Johannesburg during 2016–2020, a total of 47% (201/426) of non–C. albicans Candida isolates were azole-resistant and 0.9% (4/491) were resistant to amphotericin B (26).
Overall, we demonstrated a change in the distribution of BSI Candida species isolated during 2019–2022; C. auris was the second most common species. An earlier national survey conducted during 2016–2017 found that C. parapsilosis was the most common species causing BSI and accounted for 44% (2,600/5,876) of cases, followed by C. albicans (23% [1,353/5,876]) and C. auris (14% [794/5,876]) (27).
Fifty percent of Candida BSI cases reported in our study were from the Gauteng Province, the most densely populated region of South Africa and the region with the most healthcare facilities and hospital beds (25,28). That province accounted for almost one third of C. auris cases. We found that C. auris caused more than one third of Candida BSI cases from the North West province. The Eastern Cape, KwaZulu-Natal, Limpopo, and Mpumalanga Provinces each accounted for >20% of C. auris cases in South Africa.
The observed changes in C. auris case numbers during the study period might have been caused by the patterns of COVID-19 through the pandemic waves (29). Alternatively, C. auris has spread out of Gauteng Province to cause outbreaks in multiple new facilities. Of concern, the incidence of C. auris in Northern Cape Province might be underestimated in the public sector. That finding might reflect differences in the extent of laboratory investigation of BSI between the public and private sectors in the Northern Cape Province.
The first limitation of our study is that overall, more than two thirds of all cases of Candida BSI were reported from the private sector in South Africa and half were reported from the Gauteng Province. Fewer cases of Candida BSI were reported from the public sector. Because we excluded Candida species other than the 6 most common species, we might have undercounted cases in the public sector because some laboratories either did not identify Candida to species level or misclassified the common Candida species as rarer species (22). Not all the data from the private laboratories were included in this study, and therefore these data do not necessarily represent the overall epidemiology of Candida BSI in the private sector. The prevalence and incidence of Candida BSI in South Africa could not be determined because denominator data were not available. Furthermore, we could not determine whether any cases were co-infections with Candida and SARS-CoV-2.
In conclusion, we identified changes in the distribution of Candida species in both the private and public heath sectors and in different provinces in South Africa during 2019–2021, particularly for C. auris. From 2017 to 2019, C. auris increased from 14% to 17%. However, large percentage increases occurred during the COVID-19 pandemic in 2020 (24%), 2021 (31%), and 2022 (28%). Taken together with temporal increases after each wave, our findings suggest that the COVID-19 pandemic might have driven the major increase in C. auris and thus accelerated the epidemiologic shift of C. auris in South Africa.
Dr. Ismail is a PhD medical scientist and trained field epidemiologist at the National Institute for Communicable Diseases. Her primary research interests are public health research and disease surveillance.
Acknowledgments
We thank the team from the Surveillance Information Management Unit at the National Institute for Communicable Diseases for making National Health Laboratory Service data available and the South African Society of Clinical Microbiology editorial committee for comments and suggestions. In addition, we thank Andronica Moipone Shonhiwa, Genevie Ntshoe, Joy Ebonwu, Lactatia Motsuku, Liliwe Shuping, Mazvita Muchengeti, Jackie Kleynhans, Gillian Hunt, Victor Odhiambo Olago, Husna Ismail, Nevashan Govender, Ann Mathews, Vivien Essel, Veerle Msimang, Tendesayi Kufa-Chakezha, Nkengafac Villyen Motaze, Natalie Mayet, Tebogo Mmaborwa Matjokotja, Mzimasi Neti, Tracy Arendse, Teresa Lamola, Itumeleng Matiea, Darren Muganhiri, Babongile Ndlovu, Khuliso Ravhuhali, Emelda Ramutshila, Salaminah Mhlanga, Akhona Mzoneli, Nimesh Naran, Trisha Whitbread, Mpho Moeti, Chidozie Iwu, Eva Mathatha, Fhatuwani Gavhi, Masingita Makamu, Matimba Makhubele, Simbulele Mdleleni, Bracha Chiger, Jackie Kleynhans, and the information technology team, Tsumbedzo Mukange, Trevor Bell, Lincoln Darwin, Fazil McKenna, Ndivhuwo Munava, Muzammil Raza Bano, Themba Ngobeni, Vusumzi Tshetu, and Gloseijie Bazolana, for making the COVID-19 data available. We also thank Lucille Blumberg, Maureen Masha, Caroline Vika, Caroline Mudara, Tracy Arendse, Lovelyn Ozougwu, and Rebone Kai for making the DATCOV data available.
This study was supported by the National Institute for Communicable Diseases. N.P.G. was supported by the National Institute for Health and Care Research (NIHR) (grant nos. NIHR134342 and NIHR303140) with UK international development funding from the UK government to support global health research. The views expressed in this article are those of the authors and not necessarily those of the NIHR or the UK government.
All authors contributed significantly to this paper. H.I. contributed to conceptualization, methodology, data cleaning, data analysis, writing, review, and editing. O.P. and R.M. contributed writing, review, and editing. W.L., C.G., P.E., W.J., and R.W. performed data curation, writing, review, and editing. N.P.G. contributed to conceptualization, methodology, data cleaning, data analysis, writing, review, editing, and supervision of the study.
References
- Jassat W, Cohen C, Tempia S, Masha M, Goldstein S, Kufa T, et al. DATCOV author group. Risk factors for COVID-19-related in-hospital mortality in a high HIV and tuberculosis prevalence setting in South Africa: a cohort study. Lancet HIV. 2021;8:e554–67. DOIPubMedGoogle Scholar
- Edoka I, Fraser H, Jamieson L, Meyer-Rath G, Mdewa W. Inpatient care costs of COVID-19 in South Africa’s public healthcare system. Int J Health Policy Manag. 2022;11:1354–61. DOIPubMedGoogle Scholar
- Gomez-Simmonds A, Annavajhala MK, McConville TH, Dietz DE, Shoucri SM, Laracy JC, et al. Carbapenemase-producing Enterobacterales causing secondary infections during the COVID-19 crisis at a New York City hospital. J Antimicrob Chemother. 2021;76:380–4. DOIPubMedGoogle Scholar
- Nori P, Cowman K, Chen V, Bartash R, Szymczak W, Madaline T, et al. Bacterial and fungal coinfections in COVID-19 patients hospitalized during the New York City pandemic surge. Infect Control Hosp Epidemiol. 2021;42:84–8. DOIPubMedGoogle Scholar
- Machado M, Estévez A, Sánchez-Carrillo C, Guinea J, Escribano P, Alonso R, et al. Incidence of candidemia Is higher in COVID-19 versus non-COVID-19 patients, but not driven by intrahospital transmission. J Fungi (Basel). 2022;8:305. DOIPubMedGoogle Scholar
- Shafran N, Shafran I, Ben-Zvi H, Sofer S, Sheena L, Krause I, et al. Secondary bacterial infection in COVID-19 patients is a stronger predictor for death compared to influenza patients. Sci Rep. 2021;11:12703. DOIPubMedGoogle Scholar
- Reda NM, Hassan RM, Salem ST, Yousef RHA. Prevalence and species distribution of Candida bloodstream infection in children and adults in two teaching university hospitals in Egypt: first report of Candida kefyr. Infection. 2023;51:389–95. DOIPubMedGoogle Scholar
- World Health Organization. WHO fungal priority pathogens list to guide research, development and public health action [cited 2025 Dec 8]. https://www.who.int/publications/i/item/9789240060241
- Tabah A, Buetti N, Staiquly Q, Ruckly S, Akova M, Aslan AT, et al.; EUROBACT-2 Study Group, ESICM, ESCMID ESGCIP and the OUTCOMEREA Network. Epidemiology and outcomes of hospital-acquired bloodstream infections in intensive care unit patients: the EUROBACT-2 international cohort study. Intensive Care Med. 2023;49:178–90. DOIPubMedGoogle Scholar
- Mathur P, Malpiedi P, Walia K, Srikantiah P, Gupta S, Lohiya A, et al.; Indian Healthcare Associated Infection Surveillance Network collaborators. Health-care-associated bloodstream and urinary tract infections in a network of hospitals in India: a multicentre, hospital-based, prospective surveillance study. Lancet Glob Health. 2022;10:e1317–25. DOIPubMedGoogle Scholar
- Govender NP, Magobo RE, Mpembe R, Mhlanga M, Matlapeng P, Corcoran C, et al. Candida auris in South Africa, 2012–2016. Emerg Infect Dis. 2018;24:2036–40. DOIPubMedGoogle Scholar
- Naicker SD, Maphanga TG, Chow NA, Allam M, Kwenda S, Ismail A, et al. Clade distribution of Candida auris in South Africa using whole genome sequencing of clinical and environmental isolates. Emerg Microbes Infect. 2021;10:1300–8. DOIPubMedGoogle Scholar
- Maphanga TG, Mpembe RS, Naicker SD, Govender NP. for GERMS-SA. In vitro antifungal activity of manogepix and other antifungal agents against South African Candida auris isolates from bloodstream infections. Microbiol Spectr. 2022;10:
e0171721 . DOIPubMedGoogle Scholar - Najeeb H, Siddiqui SA, Anas Z, Ali SH, Usmani SUR, Jawed F, et al. The menace of Candida auris epidemic amidst the COVID-19 pandemic: a systematic review. Diseases. 2022;10:58. DOIPubMedGoogle Scholar
- Dratch AH, Le M, Zahn M. Increased incidence of Candida auris colonization in early COVID-19 pandemic, Orange County, California, USA. Emerg Infect Dis. 2025;31:1747–54. DOIPubMedGoogle Scholar
- Schaefer S, Walits E, Thaler K, Patel G. Impact of the COVID-19 pandemic on Candida auris infections: a retrospective analysis in an academic medical center in New York City. Open Forum Infect Dis. 2024;11:
ofae148 . DOIPubMedGoogle Scholar - Bing J, Huang Y, Du H, Guo P, Cao J, Kang M, et al. Rapid spread of Candida auris in China after COVID-19. J Infect. 2025;90:
106476 . DOIPubMedGoogle Scholar - Ismail H, Lowman W, Bch MB, Path FC, Microbiol SA, Microbiol M, et al. Surveillance and comparison of antimicrobial susceptibility patterns of ESKAPE organisms isolated from patients with bacteraemia in South Africa, 2016–2017. S Afr Med J. 2019;109:934–40. PMID:PubMedGoogle Scholar
- National Department of Health. Surveillance for antimicrobial resistance and consumption of antimicrobials in South Africa, 2021 [cited 2025 Sep 8]. https://knowledgehub.health.gov.za/system/files/elibdownloads/2023-04/AMR%2520and%2520AMC%2520report%2520for%25202021%2520in%2520South%2520African_June2022.pdf
- Pulliam JRC, van Schalkwyk C, Govender N, von Gottberg A, Cohen C, Groome MJ, et al. Increased risk of SARS-CoV-2 reinfection associated with emergence of Omicron in South Africa. Science. 2022;376:
eabn4947 . DOIPubMedGoogle Scholar - Ramos-Martínez A, Pintos-Pascual I, Guinea J, Gutiérrez-Villanueva A, Gutiérrez-Abreu E, Díaz-García J, et al. Impact of the COVID-19 pandemic on the clinical profile of candidemia and the incidence of fungemia due to fluconazole-resistant Candida parapsilosis. J Fungi (Basel). 2022;8:1–12. DOIPubMedGoogle Scholar
- Lockhart SR, Jackson BR, Vallabhaneni S, Ostrosky-Zeichner L, Pappas PG, Chiller T. Thinking beyond the common Candida species: need for species-level identification of Candida due to the emergence of multidrug-resistant Candida auris. J Clin Microbiol. 2017;55:3324–7. DOIPubMedGoogle Scholar
- Mroczyńska M, Brillowska-Dąbrowska A. Virulence of clinical Candida isolates. Pathogens. 2021;10:466. DOIPubMedGoogle Scholar
- Whaley SG, Berkow EL, Rybak JM, Nishimoto AT, Barker KS, Rogers PD. Azole antifungal resistance in Candida albicans and emerging non-albicans Candida species. Front Microbiol. 2017;7:2173. DOIPubMedGoogle Scholar
- Govender NP, Patel J, Magobo RE, Naicker S, Wadula J, Whitelaw A, et al.; TRAC-South Africa group. Emergence of azole-resistant Candida parapsilosis causing bloodstream infection: results from laboratory-based sentinel surveillance in South Africa. J Antimicrob Chemother. 2016;71:1994–2004. DOIPubMedGoogle Scholar
- Chibabhai V. Incidence of candidemia and prevalence of azole-resistant candidemia at a tertiary South African hospital—a retrospective laboratory analysis 2016–2020. S Afr J Infect Dis. 2022;37:326. DOIPubMedGoogle Scholar
- van Schalkwyk E, Mpembe RS, Thomas J, Shuping L, Ismail H, Lowman W, et al. GERMS-SA. Epidemiologic shift in candidemia driven by Candida auris, South Africa, 2016–2017. Emerg Infect Dis. 2019;25:1698–707. DOIPubMedGoogle Scholar
- Alavinejad M, Mellado B, Asgary A, Mbada M, Mathaha T, Lieberman B, et al. Management of hospital beds and ventilators in the Gauteng province, South Africa, during the COVID-19 pandemic. PLOS Glob Public Health. 2022;2:
e0001113 . DOIPubMedGoogle Scholar - Yang W, Shaman JL. COVID-19 pandemic dynamics in South Africa and epidemiological characteristics of three variants of concern (Beta, Delta, and Omicron). eLife. 2022;11:1–50. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: April 02, 2026
Table of Contents – Volume 32, Number 4—April 2026
| EID Search Options |
|---|
|
|
|
|
|
|





Please use the form below to submit correspondence to the authors or contact them at the following address:
Nelesh P. Govender, Wits Mycology Division, 1 Modderfontein Rd, Sandringham 2131, South Africa
Top