Volume 4, Number 2—June 1998
Perspective
Could Myocarditis, Insulin-Dependent Diabetes Mellitus, and Guillain-Barré Syndrome Be Caused by One or More Infectious Agents Carried by Rodents?
Cite This Article
Citation for Media
Abstract
The numbers of small rodents in northern Sweden fluctuate heavily, peaking every 3 or 4 years. We found that the incidence of Guillain-Barré syndrome and insulin-dependent diabetes mellitus, as well as the number of deaths caused by myocarditis, followed the fluctuations in numbers of bank voles, although with different time lags. An environmental factor, such as an infectious agent, has been suggested for all three diseases. We hypothesize that Guillain-Barré syndrome, myocarditis, and insulin-dependent diabetes mellitus in humans in Sweden are caused by one or more infectious agents carried by small rodents. Also, a group of novel picornaviruses recently isolated from these small rodents is being investigated as the possible etiologic agent(s).
Nephropathia epidemica (NE) is a disease caused by Puumala virus (genus Hantavirus, family Bunyaviridae). The vector and natural reservoir of Puumala virus is a small rodent, the bank vole (Clethrionomys glareolus) (1). In most parts of southern Sweden, bank vole populations are noncyclic, whereas in the north, populations fluctuate on a 3- or 4-year cycle of abundance (2-4). NE is endemic only in the northern part of the country, and the number of human cases cycles with the bank vole population (5-6).
We found the incidence of myocarditis, Guillain-Barré syndrome (GBS), and insulin-dependent-diabetes mellitus (IDDM) to lag behind the population density fluctuations of bank voles, although with different time delays. We hypothesize that in Sweden the bank vole is a vector for one or more infectious agents, which are pathogenic and cause these diseases in humans.
Density of Bank Voles
Long-term records (>10 years) on small rodent abundance are scarce. However, in Sweden such data are available from the area with cyclic rodent populations (2, 4-8). We used data from Grimsö (59°40' N, 15°25' E), where small rodents have been collected every autumn since 1973 (6-8). Snap-trapping was performed where possible in six adjacent 5x5-km areas, each with four 1-ha plots. Of 24 permanent 1-ha plots, 20 were trapped. At each trapping, approximately 940 traps were set during three consecutive days, amounting to approximately 2,800 "trap-nights."
Species, date, and location of trapped animals were recorded (4, 6-8). As an index of bank vole abundance, we calculated the number of animals trapped per 100 trap nights (i.e., density). Bank vole abundance fluctuated in cycles with 3- or 4-year intervals between density peaks (Figure 1).
Incidence of Disease
We used the national census to calculate (according to counties and age groups) annual disease incidences per 100,000 population. Because of the lack of long-term records on noncyclic small rodent populations, we analyzed data from the counties with cyclic rodent populations: Norrbotten, Västerbotten, Västernorrland, Jämtland, Gävleborg, Kopparberg, Värmland, Örebro, Västmanland, Uppsala, Stockholm, Södermanland, Göteborg, Älvsborg, Jönköping, and Kronoberg (2-3). We excluded Stockholm and Uppsala counties as they are predominantly urban and on the border between counties with cyclic and noncyclic rodents.
Myocarditis
Myocarditis is a clinical condition in which the heart is infiltrated by inflammatory cells. Diagnosis is very difficult during the acute stage and is often made by postmortem microscopic investigation of the myocardium. All causes of death in Sweden are registered by the Swedish Cause-of-Death Statistics, Statistics Sweden, S-115 81 Stockholm, Sweden. Our study included all deaths between 1970 and 1986, in the age group 11 to 46 years, caused by myocarditis (ICD 8 code 422), if the diagnosis was given as either the direct (primary) cause of death or as the first out of six recorded contributing causes of death. The age group 11 to 46 years was chosen because congenital or arteriosclerotic cardiovascular disease is less common in this age group. In 201 (92%) of 218 cases, the diagnoses were based on clinical or forensic autopsies. The remaining diagnoses were based on clinical examination before death. We did not use data collected after 1986, as the disease classification changed from ICD 8 to ICD 9 in 1987, making comparison with the period after 1986 difficult.
Guillain-Barré Syndrome
In different parts of Europe (9), reported incidence of GBS varies considerably depending on study method. Our data, from the hospital discharge registry, Swedish National Board of Health and Welfare, S-106 30 Stockholm, Sweden, are considered adequate for epidemiologic surveillance of GBS (9). We selected patients hospitalized with the diagnosis GBS (ICD 8 code 354) and gathered information on age, sex, county of residence, county of hospitalization, diagnosis, date of admission and date of discharge. We only analyzed data of GBS patients 46 years of age and younger, as the diagnoses in older patients frequently are obscured by nonspecific illnesses and other problems (10).
Insulin-Dependent Diabetes Mellitus
IDDM was studied in Medelpad (part of Västernorrland County), an area with cyclic rodent populations. The patients were identified through registries of patients with diabetes at the only hospital and at 16 out of the 17 health-care centers, as well as through common registries of diagnosis at one health-care center. The World Health Organization (WHO) diagnostic criteria were applied. We analyzed case data in all age groups because precision in the diagnosis of IDDM is not age related.
Statistical Analysis
A cross-correlation function (CCF) (11) can indicate any direct or delayed dependence between two different time series. A CCF graph is a plot of positive and negative correlation coefficients between pair-wise values from the two series. The correlations have been calculated between the two series for different time shifts (lags), with lag 0 meaning no time shift, lag -1 meaning the first series has been shifted backwards one time unit, and lag 1 meaning the first series has been shifted forward by one time unit.
We used CCFs, calculated in Statistical Package for the Social Sciences (SPSS) (12), to indicate any temporal association between the incidence in cyclic versus noncyclic areas of GBS and of deaths from myocarditis, respectively. We also used CCFs to indicate any direct (at lag 0) or delayed dependence (at negative lags) of the different disease incidences on vole density. At negative lags, the vole time series is the leading indicator that is shifted "backwards" along the time scale, relative to the disease series. Thus, the correlation coefficients at negative lags are in focus, as they indicate any disease incidence dependence on past vole densities, i.e., on densities 1, 2, or 3 years ago. At positive lags, the disease time series is shifted "backwards" relative to the vole time series. The correlation coefficients at positive lags may be large because of the similarities between the two series, but we see no causal relationship between vole density and past disease incidence.
Log transformation of the time series was used because of the variable amplitudes of the fluctuations in the disease and vole series. In the case of IDDM, we also ran one CCF with the disease and vole time series difference-transformed by one year.
Myocarditis
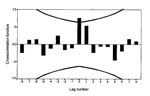
A total of 218 patients, ages 11 to 46 years, died of acute myocarditis in 1970 to 1986; 148 in counties with cyclic rodent density and 70 in counties with noncyclic rodent density. Sex ratio was 2:1 (146 male, 72 female patients). Myocarditis death incidences in cyclic and noncyclic areas (Figure 2) were not correlated as revealed by cross-correlation of the disease series (CCF not shown; n = 17 computable 0-order correlations). Cross-correlation of the annual incidence of myocarditis deaths in 1970 to 1986 in the cyclic area with bank vole abundance (Figure 3) showed that the incidence of myocarditis was most highly correlated with vole abundance in the previous year, according to the high positive and significant correlation at lag -1 in the CCF (Figure 4, 5) (r = 0.635, p < 0.05, n = 13).
Guillain-Barré Syndrome
In five cyclic counties and two noncyclic counties, where complete data were available, 258 GBS patients (< 46 years of age) were hospitalized in 1973 to 1982; the sex ratio was 1.1:1 (135 male, 123 female patients), 104 from the cyclic and 154 from the noncyclic area.
No correlation was found between GBS in the cyclic and noncyclic areas by cross-correlation of the disease series (CCF not shown; n = 10 computable 0-order correlations). Cross-correlation of the annual incidence of GBS in cyclic areas in 1973 to 1982 with bank vole abundance (Figure 6) suggested that the incidence of GBS co-varied with current vole abundance, as shown by the high positive and significant correlation at lag 0 in the CCF (Figures 7, 8) (r = 0.757, p < 0.05, n = 10).
Insulin-Dependent Diabetes Mellitus
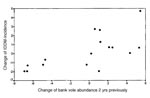
A total of 318 cases of IDDM were recorded in 1971 to 1991, representing a sex ratio of 1.3:1 (179 male, 139 female patients). No cross-correlation was found between the annual incidence of IDDM in the cyclic area and bank vole abundance (Figure 9)(CCF not shown; n = 19 computable 0-order correlations), as was found for myocarditis and GBS with vole abundance. On the other hand, after the alternative transformation (to log transformation) by differencing the IDDM and vole time series by 1 year, changes of the IDDM incidence from one year to the next covaried with the changes of vole abundance 2 years previously. This was suggested by the high positive and significant correlation at lag -2 in the CCF (Figures 10, 11) (r = 0.595, p < 0.05, n = 16).
The confidence limits (+2 SE) for the most important cross correlations, at lag 0 for GBS (Figure 7), lag -1 for myocarditis (Figure 4), and at lag -2 for IDDM (Figure 10), were calculated in an alternative way to the default calculation in SPSS (12) according to formula 11.1.11, p. 377 (11). The alternative calculations were made according to formula 11.1.7, p. 376 (11). These calculations were made assuming that 1) the three disease time series represented white noise (i.e. contain no autocorrelation); 2) the vole time series contained autocorrelation, but it was zero after lag 5; 3) for the CCF of GBS with the vole time series, correlation existed at lag 0 and +1 but was zero at other lags; 4) for the CCF of myocarditis with the vole series, correlation existed at lag -1 but was zero at other lags; and 5) for the CCF of the differenced IDDM with the differenced vole time series, correlation existed at lag 0 to lag -4 but was zero at other lags and the differenced vole time series contained autocorrelation up to, but not after, lag 7. The calculations of the alternative confidence limits showed that the most important cross-correlations were significant at p < 0.05. The confidence limits for GBS at lag 0 were 0.757 + 0.542; for myocarditis at lag -1, 0.635 + 0.450; and for the differenced IDDM at lag -2, 0.595 + 0.478.
Zoonotic disease reservoir or vector population density data can indicate a link between the etiologic agent and its reservoir/vector and between the agent and the disease. This type of data linked rodents to NE in Sweden in the 1930s and to Korean hemorrhagic fever in the 1950s (13-14). In a previous study, we found that the Puumala virus infection rate in bank voles in the spring tracked the vole density in the previous autumn and that infection rate peaked as vole population declined (5). We believe that this explains the approximately 9-month time lag in the incidence of NE in the spring relative to vole abundance in the previous autumn, although the incubation period in humans is only 2-6 weeks.
Our present data show significant temporal correlations of bank vole population density with incidence of death from myocarditis (Figures 3-5) and incidence of GBS (Figures 6- 8). Our data also show a positive and significant correlation between changes in IDDM incidence and changes in bank vole density (Figures 9- 11). We hypothesize that these three diseases are caused or triggered by one or more infectious agents carried by bank voles in Sweden. In this context, several sudden deaths among Swedish orienteers were recognized during 1989 to 1992. Six of approximately 200 elite orienteers died of acute myocarditis (15). Orienteering involves finding the fastest/shortest way between several checkpoints, often in forested areas. Because of extensive exposure to nature, it has been speculated that the sudden deaths of the orienteers were caused by a vector-borne (rodent or arthropod) infectious agent.
If our hypothesis is correct, the different time lags of different diseases relative to vole abundance, as seen in the present study, may depend in part on the specific infection rate dynamics of any agent in the bank vole population as seen in the case of NE (5). The time elapsed from infection to disease onset or death may also be important in IDDM and myocarditis.
The disease time series presented here were not very long, and the results should therefore be treated with caution. The present findings do not prove any direct link between the diseases investigated and the bank vole. Many infectious diseases and other biological phenomena have a cyclic variation, and the relationships presented here may be spurious or may involve another reservoir or vector population (e.g., small rodents and small game species that fluctuate synchronously with the bank vole at these latitudes) (4-5,7-8,16-17).
The identification of the etiologic agent or agents is critical to "prove" our hypothesis. However, as seen in several zoonotic diseases, humans are often dead-end hosts, and it may be easier to isolate the etiologic agent from the reservoir or vector than from the patient. Also, because of the time elapsed between primary infection and onset of disease, the infectious agent may be present only in very small amounts or absent. It is also possible that several human diseases with no etiologic agent identified are caused by agents that are difficult to cultivate, at least by standard techniques.
Thus, because we need to identify the etiologic agent(s) and it is difficult to isolate any virus from humans, we have initially attempted to isolate new viruses from small rodents, the primary suspects. The bank vole is the most common wild-living Swedish mammal. Its abundance varying by >300 times (4), this rodent is likely to have a high and highly variable potential over time to transmit any etiologic agent to humans. In addition, bank voles are known to enter buildings, thus transporting disease risk closer to humans.
Three novel virus isolates from bank voles, resembling picornaviruses in size and morphology, have been found. The first isolate was named Ljungan after the Ljungan river in Medelpad County, Sweden, where the animals were trapped. The second and third isolate originated from animals trapped in Västerbotten County. The amino acid sequences of predicted Ljungan virus capsid proteins were closely related (approximately 70% similarity) to the human pathogen echovirus 22. Partial 5' noncoding region sequence of Ljungan virus was most closely related to cardioviruses. Two additional isolates were serologically and molecularly related to the prototype (Niklasson et al., unpub. observation). Echovirus 22 is a known human pathogen, and other known cardioviruses can induce not only myocarditis but also neurologic diseases and IDDM in several species of animals.
We hope to elucidate the role of Ljungan virus as a human pathogen by serologic assays for Ljungan virus, diagnostic polymerase chain reaction based on generated sequence data, and specific antisera for viral antigen detection. Studies are also under way to identify and determine the role of other viruses of these small rodents in inducing myocarditis, GBS, and IDDM in humans.
Acknowledgment
We thank Sara Sjöstedt for statistical help and Stiftelsen Olle Engkvist, Byggmästare and by the Swedish Environment Protection Agency (via the National Environmental Monitoring Programme) for financial support for the vole trapping.
References
- Niklasson B, Le Duc J. Epidemiology of nephropathia epidemica in Sweden. J Infect Dis. 1987;155:269–76.PubMedGoogle Scholar
- Hansson L, Henttonen H. Gradients in density variations of small rodents: the importance of latitude and snow cover. Oecologia. 1985;67:394–402. DOIGoogle Scholar
- Hansson L, Henttonen H. Rodent dynamics as community processes. Trends Ecol Evol. 1988;3:195–200. DOIGoogle Scholar
- Hörnfeldt B. Delayed density dependence as a determinant of vole cycles. Ecology. 1994;75:791–806. DOIGoogle Scholar
- Niklasson B, Hörnfeldt B, Lundkvist Å, Björsten S, LeDuc J. Temporal dynamics of Puumala virus antibody prevalence in voles and of nephropathia epidemica incidence in humans. Am J Trop Med Hyg. 1995;53:134–40.PubMedGoogle Scholar
- Hörnfeldt B. Smådäggdjursinventering i PMK:s referensområden - rapport från verksamheten 1992. Solna, Sweden: Swedish Environmental Protection Agency; 1994. Report 4294.
- Lindström ER, Andrén H, Angelstam P, Cederlund G, Hörnfeldt B, Jäderberg L, Disease reveals the predator: sarcoptic mange, red fox predation and prey populations. Ecology. 1994;75:1042–9. DOIGoogle Scholar
- Lindström ER, Hörnfeldt B. Vole cycles, snow depth and fox predation. Oikos. 1994;70:156–60. DOIGoogle Scholar
- Jiang GX, de Pedro-Cuesta J, Fredriksson G. Guillain-Barré syndrome in South-West Stockholm, 1973-1991. Quality of registered hospital diagnosis and incidence. Acta Neurol Scand. 1995;91:109–17.PubMedGoogle Scholar
- Winner SJ, Evans JG. Age-specific incidence of Guillain-Barré syndrome in Oxfordshire. Quarterly Journal of Medicine. New Series. 1990;77:1297–304.
- Box GEP, Jenkins GM. Time series analysis, forecasting and control. San Francisco: Holden Day; 1970.
- SPSS Base System Syntax Reference Guide. Release 6.0. Carey (NC): SPSS Inc.; 1993.
- Myhrman G. En njursjukdom med egenartad symptombild. [In Swedish]. Nord Med Tidskr. 1934;7:793–4.
- Gajdusek DC. Virus haemorrhagic fevers-special reference to haemorrhagic fevers with renal syndrome (epidemic haemorrhagic fever). J Pediatr. 1962;60:841–57. DOIPubMedGoogle Scholar
- Wesslén L, Påhlson C, Friman G, Fohlman J, Lindquist O, Johansson C. Myocarditis caused by Chlamydia pneumonie (TWAR) and sudden unexpected death in a Swedish elite orienteer. Lancet. 1992;340:427–8. DOIPubMedGoogle Scholar
- Hörnfeldt B. Synchronous population fluctuations in voles, small game, owls and tularemia in northern Sweden. Oecologia. 1978;32:141–52. DOIGoogle Scholar
- Hörnfeldt B, Löfgren O, Carlsson B-G. Cycles in voles and small game in relation to variations in plant production indices in northern Sweden. Oecologia. 1986;68:496–502. DOIGoogle Scholar
Figures
Cite This ArticleTable of Contents – Volume 4, Number 2—June 1998
| EID Search Options |
|---|
|
|
|
|
|
|









Please use the form below to submit correspondence to the authors or contact them at the following address:
Bo Niklasson, Swedish Institute for Infectious Disease Control, S-105 21 Stockholm, Sweden; fax: 46-8-735-66-15
Top