Volume 4, Number 4—December 1998
Perspective
Ecologic Studies of Rodent Reservoirs: Their Relevance for Human Health
Abstract
Within the past few years, the number of "new" human diseases associated with small-mammal reservoirs has increased dramatically, stimulating renewed interest in reservoir ecology research. A consistent, integrative approach to such research allows direct comparisons between studies, contributes to the efficient use of resources and data, and increases investigator safety. We outline steps directed toward understanding vertebrate host ecology as it relates to human disease and illustrate the relevance of each step by using examples from studies of hosts associated with rodent-borne hemorrhagic fever viruses.
The practical importance of understanding host and vector ecology has been recognized at least since the early 1900s. Knowledge of the container-breeding habits of Aedes aegypti enabled early successes in the control of yellow fever virus transmission and, ultimately, the completion of the Panama Canal in 1914 (1). Diverse applications of vector/reservoir ecology-based measures to prevent zoonotic disease include the prediction of Lyme disease risk by monitoring acorn mast production and its impact on the vertebrate hosts of the tick vectors (2), control of vector populations for Borrelia burgdorferi and Yersinia pestis through the application of acaricides and insecticides to rodents and deer at feeding stations (3,4), dissemination of bait containing vaccines to control rabies in foxes (5), and use of satellite imagery to predict the activity of Rift Valley fever in East Africa (6,7).
The rodent-borne hemorrhagic fevers, among the most dramatic of recently emerging infectious diseases, are caused by two distinct groups of negative-stranded RNA viruses: the arenaviruses (family Arenaviridae) and the hantaviruses (genus Hantavirus, family Bunyaviridae). With few exceptions, each virus in these two groups is primarily associated with a single species of rodent host of the family Muridae. In the specific host, the virus establishes a prolonged infection, which rarely causes disease in the animal. The infected host sheds virus into the environment (in urine, feces, and saliva) for extended periods (8-10). These characteristics are key to the transmission of the viruses to humans (by the inhalation of aerosolized virus) and to other rodents (by horizontal and sometimes vertical mechanisms).
Arenaviruses cause the South American hemorrhagic fevers, which produce hundreds of cases annually, with a case-fatality ratio as high as 33%. The best studied of these agents is Junín virus, which is carried by the corn mouse (Calomys musculinus) and causes Argentine hemorrhagic fever (AHF). AHF was first recognized in 1955 on the central pampas of Argentina (11), where before the deployment of a new vaccine in 1992, hundreds of cases occurred each year. Although arenaviral diseases of humans (other than lymphocytic choriomeningitis associated with the introduced Old World rodent Mus musculus) have not been recognized in North America, Tamiami virus has been recognized in association with cotton rats (Sigmodon hispidus) since 1969 (12), and Whitewater Arroyo virus was identified from wood rats (Neotoma species) in the southwestern United States in 1995 (13). The potential of Whitewater Arroyo virus for causing human disease is under investigation.
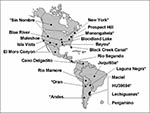
Hantaviruses cause hundreds of thousands of cases of hemorrhagic fever with renal syndrome (HFRS) in Europe and Asia each year. Hantaviral disease was thought to be rare or absent in the United States, although three cases of mild HFRS associated with rat-borne Seoul virus had been described (14). In 1993, the discovery of hantavirus pulmonary syndrome (HPS), which rapidly kills approximately half of those infected, surprised public health officials and virologists in the United States. The causative agent, Sin Nombre virus (SNV), is hosted by the deer mouse, Peromyscus maniculatus, and the disease syndrome markedly differs from HFRS. Since 1993, at least 20 additional hantaviruses have been isolated from rodents throughout the Americas; about half are known human pathogens (Figure 1).
Recognition of new rodent-borne diseases has renewed interest in reservoir host ecology in the United States, and recognition of HPS in South America has prompted field studies in Paraguay, Argentina, Chile, and Brazil. In 1997, representatives from 13 American countries, meeting in Lima, identified surveillance in humans and rodents as a priority for combating emerging hantaviral disease. Reservoir studies are being designed and conducted in many parts of the world by mammalogists, ecologists, and vector-control personnel. However, sanitarians and others with no specific training in such areas are being asked to conduct reservoir studies in the course of investigating disease outbreaks. Rarely are these professionals trained in the safe handling of animals potentially infected with viruses capable of causing fatal disease.
To promote discussion of study design, data collection, and appropriate and safe methods for reservoir studies, we review prior efforts and lessons learned. Although specific examples are restricted to the rodent-borne hemorrhagic fever viruses, many of the generalizations can be applied to other pathogens maintained by small-mammal reservoirs.
Assuming that the primary reservoir host has already been implicated, the following consecutive but overlapping steps can promote an understanding of rodent reservoir ecology as it relates to human disease: 1) determination of the geographic distribution of the host; 2) determination of the geographic range of the pathogen within the host range; 3) determination of the regional distribution of the host and pathogen among the distinct biomes or habitat types; 4) determination of the relative prevalence of infection among demographic subpopulations of the host (e.g., males versus females and adults versus juveniles); 5) elucidation of the temporal and fine-scale spatial patterns of host-pathogen dynamics through prospective, longitudinal studies; and 6) development of an integrative time- and place-specific predictive model.
Examples from studies of hantaviruses and arenaviruses illustrate how each step is relevant to human health.
Geographic Distribution of the Host
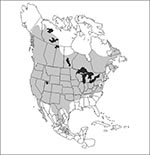
The geographic distribution of the host(s) defines the maximum area in which the disease can be endemic. For most small-mammal species in North America, these distributions are known relatively precisely (15). For example, the deer mouse is one of the best-studied small-mammal species in the world, and its distribution throughout North America is well defined (Figure 2). Fewer data are available on the distribution of Central and South American species. The range of the cotton rat, S. hispidus, which is the reservoir of both Black Creek Canal virus (a hantavirus that causes HPS) and Tamiami virus (an arenavirus not associated with human disease) is well defined in the United States. However, the southern limits of the species' distribution are unclear (16).
Problems in defining species' distributions are exacerbated by imprecise taxonomy. The multimammate rat, Mastomys natalensis, was originally described as the reservoir for Lassa fever in West Africa. Subsequent taxonomic studies have shown that several morphologically similar animals in sub-Saharan Africa actually form a species complex (17). The phylogeny of this complex is being studied by cytologic and molecular techniques (18), but it will be years before the geographic distributions of each species can be mapped and used to interpret the restricted distribution of Lassa fever.
Andes virus, associated with Oligoryzomys longicaudatus, causes HPS in the northern Patagonian regions of Argentina and Chile (19). Another hantavirus, Oran virus, also causes HPS and is apparently associated with the same rodent species in extreme northern Argentina (19). However, the population of O. longicaudatus in northern Argentina appears to be disjunct from the population that follows the Andean cordillera and includes northwestern Patagonia (20). Careful taxonomic and distributional studies are required to determine if these disjunct rodent populations represent distinct species and to define the potential HPS-endemic areas associated with each virus.
Geographic Distribution of the Pathogen within the Host Range
The extent of the geographic area in which the infected host can be found indicates the area in which human disease can be endemic. In some cases, host and pathogen distributions are largely coincident. SNV infection has been found in deer mice throughout North America north of Mexico, with the exception of Alaska (Ksiazek et al., unpub. obs.; 21,22). Rattus norvegicus is found throughout most of the world, and infection with Seoul virus has been reported on every continent except Antarctica (23).
However, for many arenaviruses, including Junín, Guanarito, and Tamiami, and for some hantaviruses, the area in which the disease is endemic apparently includes only a small portion of the range of individual host species (Figure 3). The reservoir for Laguna Negra virus, which causes HPS in Paraguay, is Calomys laucha, and approximately 12% of the C. laucha tested from the Chaco region of western Paraguay are infected with Laguna Negra virus (24). C. laucha is also one of the most common rodents in central Argentina, yet testing of thousands of samples has provided no evidence of Laguna Negra virus in central Argentina (25; Levis et al., unpub. obs.). However, the distributional data of C. laucha indicate that populations in Paraguay are disjunct from those in Argentina and Uruguay. Additional distributional, taxonomic, and laboratory studies are required to determine whether genetic differences in host populations are influencing susceptibility to infection or other factors are influencing virus distribution in a uniformly susceptible population.
The geographic range of infection by the pathogen within reservoir populations may be dynamic, not static. In 1955, the AHF-endemic area included approximately 10,000 km2 centered around the town of Junín in Buenos Aires province. By 1985, the AHF-endemic area encompassed more than 150,000 km2 in four provinces (26). Prevalence of infection in reservoir populations may be very high in some localities and very low, or absent, at nearby sites, for both arenaviruses (27) and hantaviruses (28,29). It is likely that pathogens periodically become extinct in local reservoir populations, only to be reintroduced later from neighboring populations. The dynamics and factors controlling these processes are unknown but indicate that, although a single cross-sectional survey approximates the range of infection, repeated surveys or longitudinal studies are necessary to elucidate temporal and spatial patterns.
Finally, in many instances, reservoir surveillance provides a clearer definition of potential disease-endemic areas than human disease surveillance, even when the incidence of human disease is extremely low (e.g., in HPS-endemic areas), and cases where the use of a vaccine may mask human exposure (e.g., in the AHF-endemic area).
Distribution of the Pathogen among Habitats
The distribution of the host and the pathogen among different biotic communities and habitat types indicates the risk for human infection in various habitats within a geographic region. The deer mouse, a habitat generalist, was found in all eight biomes studied in the four-state region of the initial HPS outbreak in the United States (28). Prevalence of SNV infection, however, varied among habitat types, being lowest at the climatic and altitudinal extremes (desert and alpine tundra) and highest at middle-altitude habitats such as piñon-juniper woodland, where most HPS cases occurred during the 1993 outbreak.
The corn mouse, reservoir for Junín virus, was described as a crop field specialist (30), and virus transmission was said to occur when farmers working in their fields inhaled aerosols of virus shed by infected rodents (31). However, during detailed studies of habitat preferences in the AHF-endemic area (Figure 4), the corn mouse was rarely found in crop fields. Instead, it inhabited the more stable, weedy fence lines and roadsides bordering crop fields. These results suggest that humans might become infected with Junín virus in border habitats and that cutting or burning vegetation in these habitats might help decrease the incidence of AHF.
The relative frequency of infection in different demographic subgroups within the host population suggests potential mechanisms of pathogen transmission within host populations. An association of antibody prevalence with age, suggesting horizontal (rather than vertical) transmission, has been documented for Norway rats infected with Seoul virus (33); deer mice, western harvest mice, and brush mice infected with SNV or SNV-like viruses (28); cotton rats infected with Black Creek Canal virus (29); and corn mice infected with Junín virus (33). For Norway rats, deer mice, and cotton rats, a J-shaped curve of antibody prevalence with age was apparent; very young animals had prevalence rates similar to those of some adults. This pattern may be explained by the presence of maternal antibody in the offspring of infected females; as this apparently protective antibody wanes, animals become infected.
Fighting and biting among adult male rodents may be a common mechanism of virus transmission. Scars or wounds on rodents can indicate intraspecific aggressive encounters. A significant association between the acquisition of scars and the development of antibody to Seoul virus was demonstrated for rats in Baltimore (34), implying that virus was transmitted by biting during fights. In Argentina, male C. musculinus had more scars than females, and adult males with scars were more often infected with Junín virus than adult males without scars. In North America, the prevalence of antibody to SNV among male deer mice, brush mice, and western harvest mice is at least twice that among females (28), and scarring appears associated with antibody in some of these species. However, infection patterns vary. Male-to-female infection ratios are approximately 2:1 for deer mice and SNV, but two studies of harvest mice found infection of 40% to 50% in males (21,28), whereas no females were infected. Although scars and antibody to Seoul virus were highly correlated for Norway rats in Baltimore, antibody prevalence did not differ between male and female rats (34). These species-specific differences are presumably related to differences in behavior and, consequently, to differences in specific mechanisms of viral transmission. Further study of these mechanisms may lead to an understanding of the timing and conditions under which increased transmission in reservoir populations takes place, which, in turn, would lead to improved prediction of how and when humans become infected.
Temporal Dynamics of Host and Pathogen Populations
Longitudinal studies of reservoir populations are needed to identify 1) the effects of infection on individuals and populations of host species, 2) the seasonal and year-to-year fluctuations in incidence and prevalence as well as the sequence and duration of infection in the host, and 3) the environmental variables associated with changes in host density or rates of transmission. The mark-release-recapture method, important for longitudinal studies of small-mammal populations, establishes permanent trapping plots operated for several nights at standard intervals (usually monthly for disease studies) and maintained for several years. Captured animals are anesthetized, identified, weighed, measured, sampled (e.g., blood and oral swab), marked with a unique number, and released at the exact site of capture. Animals may be recaptured in subsequent months so that growth rates, movement, reproductive condition, and infection status can be monitored over time. Environmental variables, such as temperature, rainfall, and vegetation, relate to reservoir population dynamics. Mark-release-recapture studies should continue long enough to monitor the effects of unusual events, such as the El Niño southern oscillation (ENSO).
The mark-release-recapture method was used in a 3-year study of corn mouse populations on the Argentine pampas (33). AHF epidemics are highly seasonal, with the peak number of cases and peak rodent populations occurring in May when autumn crops are harvested. During the first 2 years, corn mouse population densities were relatively low, and the magnitude of the annual AHF epidemics was also low (Figure 5). During the third year, unusually mild weather resulted in reproduction throughout the winter, high overwinter survival (35), and a very high population base at the beginning of the spring reproductive season. The following summer had abundant rainfall, vegetation remained green, and the usual midsummer decline in rodent populations did not occur. Consequently, corn mouse populations and numbers of infected corn mice reached extremely high levels by early autumn; an unusually severe epidemic of AHF cases followed (Figure 5; 33).
Longitudinal field studies also show the effects of infection on host populations. Contrary to indications from laboratory studies (10), Junín virus infection had no effect on the growth, movement, or longevity of corn mice (33), which implies that laboratory infection may not mimic natural transmission and illustrates the importance of investigating transmission mechanisms under field conditions.
Because the animals are released, mark-release-recapture studies do not provide data on diet, reproduction, precise population age structure (using molar wear patterns or eye lens weights), and variation in genetics of host and pathogen populations. Complementary investigations in which animals are collected for species identification and harvesting of tissues are therefore essential.
Predictive Models
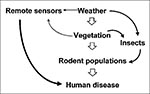
The goal of reservoir ecology is to integrate and apply the data toward the development or refinement of predictive models (Figure 6) that help public health authorities identify specific times and locations that may pose a threat to public health. In addition to early warning capability, these models may suggest when, where, and how to intervene (e.g., in the form of reservoir control or habitat modification) to break the transmission cycle or otherwise decrease the incidence of human infection.
Figure 6 is a simplified schematic for such a model. We assume that the risk for human disease is directly related to reservoir population density because increased density increases the probability of human contact with infected rodents. Data on the incidence of HFRS due to Puumala virus in Sweden and studies of rodent population densities at HPS case and control households in the United States support this assumption (36,37). We also assume that reservoir population density is influenced by characteristics of the biotic environment (e.g., habitat quality and food supply), which in turn are influenced by characteristics of the physical environment (e.g., climate, weather, and edaphic conditions). Accurate predictive models are not yet available, but recent developments are promising. The clear correlation between reservoir population density and the risk for AHF indicates that monitoring of rodent populations might give public health professionals a 2- to 3-month predictive capability—enough time to make advisory warnings but not enough to intervene with a vaccination program or integrated pest management. The Argentine model also provided clues about factors contributing to the rodent irruption. The proper model might have allowed prediction of the rodent irruption and increased risk to humans a year before the epidemic.
Continuous measurement of environmental variables wherever rodent-borne zoonoses are endemic may not be necessary. Remote sensing and geographic information systems have measured vegetation indexes that help predict changing risk for zoonotic diseases (6,38). Integration of remote sensing with long-term studies measuring environmental conditions on the ground may provide the key to predicting future risk for virus transmission. A more complete understanding of generalized climatic patterns, such as ENSO, and their effects on local environmental variables may lead to even earlier predictive capabilities.
An otherwise complete and carefully executed study can be rendered useless if the species from which each sample was taken cannot be confirmed. Except in the case of mark-release-recapture studies, voucher specimens for all captured small mammals should be archived, and museum accession numbers should be cited in reports of these studies. Even if field identifications are accurate according to existing nomenclature, taxonomic revisions may leave the source of a pathogen in question unless the voucher is available for reexamination. For example, the recent search for the reservoir of Ebola virus near Kikwit, Republic of the Congo, resulted in the identification of at least seven taxa of rodents and insectivores that are preliminarily considered new species (39).
Where resources are available, a field study is enhanced by the maintenance of specimens in a museum frozen tissue collection. These specimens can be used by future investigators for genetic, taxonomic, and microbiologic studies. A listing of museums (in the Western Hemisphere) maintaining mammal collections, including frozen tissue collections, is available (40), as are instructions for preparation and maintenance of voucher and tissue specimens (41-43).
Although the precise circumstances of their transmission to humans are often unclear, zoonotic pathogens are associated with direct or indirect exposure to infected hosts. Modes of human infection include inhalation of infectious aerosols, direct contact of contaminated fomites with broken skin or mucous membranes, animal bites, or arthropod vectors. Mammalogists and others who work with reservoir populations are likely at increased risk. Safe methods and proper protective equipment must be used in handling potentially infected rodents. In handling reservoir species for known human pathogens such as hantaviruses and arenaviruses, protective clothing, latex gloves, and respirators with high-efficiency particulate air filters should be used. Animals should be anesthetized to minimize the risk for injuries, and the use of sharp instruments and needles for collection should be minimized. Safety guidelines are available in English (41,44) and Spanish (45).
Investigators studying relatively benign or less easily transmitted agents (e.g., Borrelia) should be alert to the potential presence of more virulent agents associated with any wild animals they may be handling. Certainly not all arenavirus- and hantavirus-host associations have been described, and some species, such as the white-footed mouse and the cotton rat, are known hosts for multiple agents.
Reservoir studies are an essential component of any integrated public health response to established or emerging zoonotic diseases. The proposed algorithm facilitates study of reservoir population ecology as it relates to human disease. Each step improves understanding of the ecology and epizootiology of hantaviruses and arenaviruses. The approach is offered as one model to stimulate discussion of appropriate methods and as a conceptual framework for other investigators to critique and improve. Other techniques, such as radio-tracking (46) and the use of injectable passive integrative transponders (PIT tags) (47), may better identify and track movements of individual animals. Methods and approaches will evolve as our knowledge of the subject matter increases. All the steps may not be appropriate in all cases, and many are beyond the capability of any single investigator or institution. For example, the delineation of the geographic range of any mammal species requires intensive taxonomic, genetic, and ecologic studies and could require the collaboration of investigators from several countries. These studies will be greatly facilitated by properly archived specimens from previous reservoir studies.
The phylogenetic relationship and distribution of pathogens may help elucidate the taxonomic relationships between closely related reservoir species. Although we have specifically addressed field studies, parallel laboratory studies of hosts and pathogens are needed for maximum benefit from these field studies. For example, studies of host and virus genetics, host specificity, and pathogenesis and viral shedding may elucidate 1) the roles of cospeciation and coevolution in the associations of hantaviruses and arenaviruses with their hosts, 2) mechanisms of viral persistence, and 3) the fundamental question of defining viral species.
Dr. Mills is chief of the Medical Ecology Unit, Special Pathogens Branch, Division of Viral and Rickettsial Diseases, CDC. His research interests include zoonotic diseases, specifically host-pathogen evolution and interactions.
Dr. Childs is the epidemiology section chief in the Viral and Rickettsial Zoonoses Branch, CDC. His major areas of expertise and research interests are the means by which zoonotic agents are maintained in animal populations and transmitted to humans.
Acknowledgment
We thank Barbara Ellis for helpful discussions, encouragement, graphics, and a critique of the manuscript. C.J. Peters provided stimulating discussion, ideas, and suggestions.
References
- Marquardt WC. Introduction to arthropods and vectors. In: Beaty BJ, Marquardt WC, editors. The biology of disease vectors. Niwot (CO): University Press of Colorado; 1996. p. 1-24.
- Ostfeld RS. The ecology of Lyme-disease risk. Am Sci. 1997;85:338–46.
- Lane RS, Casher LE, Peavey CA, Piesman J. Modified bait tube controls disease carrying ticks and fleas. Calif Agric. 1998. In press.
- Sonenshine DE, Allan SA, Andrew I, Norval RA, Burridge MJ. A self-medicating applicator for control of ticks on deer. Med Vet Entomol. 1996;10:149–54. DOIPubMedGoogle Scholar
- Aubert M. Control of rabies in foxes: what are the appropriate measures? Vet Rec. 1994;134:55–9.PubMedGoogle Scholar
- Linthicum KJ, Bailey CL, Davies FG, Tucker CJ. Detection of Rift Valley fever viral activity in Kenya by satellite remote sensing imagery. Science. 1987;235:1656–9. DOIPubMedGoogle Scholar
- Peters CJ. Emergence of Rift Valley fever. In: Saluzzo JF, Dodet B, editors. Factors in the emergence of arbovirus diseases. Paris: Elsevier; 1997. p. 253-64.
- Lee HW, Lee PW, Baek LJ, Song CK, Seong IW. Intraspecific transmission of Hantaan virus, etiologic agent of Korean hemorrhagic fever, in the rodent Apodemus agrarius. Am J Trop Med Hyg. 1981;30:1106–12.PubMedGoogle Scholar
- Hutchinson KL, Rollin PE, Peters CJ. Pathogenesis of North American hantavirus, Black Creek Canal virus, in experimentally infected Sigmodon hispidus. Am J Trop Med Hyg. 1998;59:58–65.PubMedGoogle Scholar
- Vitullo AD, Hodara VL, Merani MS. Effect of persistent infection with Junín virus on growth and reproduction of its natural reservoir, Calomys musculinus. Am J Trop Med Hyg. 1987;37:663–9.PubMedGoogle Scholar
- Arribalzaga RA. Una nueva enfermedad epidémica a germen desconocido: hipertermia nefrotóxica, leucopénica y enantemática. Dia Med. 1955;27:1204–10.PubMedGoogle Scholar
- Calisher CH, Tzianabos T, Lord RD, Coleman PH. Tamiami virus, a new member of the Tacaribe group. Am J Trop Med Hyg. 1970;19:520–6.PubMedGoogle Scholar
- Fulhorst CF, Bowen MD, Ksiazek TG, Rollin PE, Nichol ST, Kosoy MY, Isolation and characterization of Whitewater Arroyo virus, a novel North American arenavirus. Virology. 1996;224:114–20. DOIPubMedGoogle Scholar
- Glass GE, Watson AJ, LeDuc JW, Childs JE. Domestic cases of hemorrhagic fever with renal syndrome in the United States. Nephron. 1994;68:48–51. DOIPubMedGoogle Scholar
- Hall ER, Kelson KR. The mammals of North America. New York: Ronald Press; 1959. p.1083.
- Eisenberg JF. Mammals of the neotropics, the northern neotropics. Chicago: University of Chicago Press; 1989. p. 449.
- Green CA, Gordon DH, Lyons NF. Biological species in Praomys (Mastomys) natalensis (Smith), a rodent carrier of Lassa virus and bubonic plague in Africa. Am J Trop Med Hyg. 1978;27:627–9.PubMedGoogle Scholar
- Britton-Davidian J, Catalan J, Granjon L, Duplantier JM. Chromosomal phylogeny and evolution in the genus Mastomys (Mammalia, Rodentia). J Mammal. 1995;76:248–62. DOIGoogle Scholar
- Levis S, Morzunov S, Rowe J, Enria D, Pini N, Calderón G, Genetic diversity and epidemiology of hantaviruses in Argentina. J Infect Dis. 1998;177:529–38. DOIPubMedGoogle Scholar
- Redford KH, Eisenberg JF. Mammals of the neotropics, the southern cone. Chicago: The University of Chicago Press; 1992. p. 430.
- Mills JN, Johnson JM, Ksiazek TG, Ellis BA, Rollin PE, Yates TL, A survey of hantavirus antibody in small-mammal populations in selected U.S. National Parks. Am J Trop Med Hyg. 1998;58:525–32.PubMedGoogle Scholar
- Drebot MA, Artsob H, Dick D, Lindsay R, Surgeoner G, Johnson A, Hantavirus surveillance in Canadian rodents. Am J Trop Med Hyg. 1997;57:143.
- LeDuc JW, Smith GA, Childs JE, Pinheiro FP, Maiztegui JI, Niklasson B, Global survey of antibody to Hantaan-related viruses among peridomestic rodents. Bull World Health Organ. 1986;64:139–44.PubMedGoogle Scholar
- Yahnke CJ, Meserve PL, Ksiazek TG, Mills JN. Prevalence of hantavirus antibody in wild populations of Calomys laucha in the central Paraguayan Chaco. In: Proceedings of the 4th International Conference on HFRS and Hantaviruses; 1998 Mar 5-7; Atlanta, Georgia. Atlanta (GA): Centers for Disease Control and Prevention; 1998.
- Schmidt K, Ksiazek TG, Mills JN. Ecology and biologic characteristics of Argentine rodents with antibody to hantavirus. In: Proceedings of the 4th International Conference on HFRS and Hantaviruses; 1998 Mar 5-7; Atlanta, Georgia. Atlanta (GA): Centers for Disease Control and Prevention; 1998.
- Maiztegui JI, Briggiler A, Enria D, Feuillade MR. Progressive extension of the endemic area and changing incidence of Argentine hemorrhagic fever. Med Microbiol Immunol (Berl). 1986;175:149–52. DOIGoogle Scholar
- Mills JN, Ellis BA, McKee KT, Ksiazek TG, Oro JG, Maiztegui JI, Junín virus activity in rodents from endemic and nonendemic loci in central Argentina. Am J Trop Med Hyg. 1991;44:589–97.PubMedGoogle Scholar
- Mills JN, Ksiazek TG, Ellis BA, Rollin PE, Nichol ST, Yates TL, Patterns of association with host and habitat: antibody reactive with Sin Nombre virus in small mammals in the major biotic communities of the southwestern United States. Am J Trop Med Hyg. 1997;56:273–84.PubMedGoogle Scholar
- Glass GE, Livingstone W, Mills JN, Hlady WJ, Fine JB, Rollin PE, Black Creek Canal virus infection in Sigmodon hispidus in southern Florida. Am J Trop Med Hyg. 1998. In press.
- de Villafañe G, Kravetz FO, Donadio O, Percich R, Knecher L, Torres MP, Dinámica de las comunidades de roedores en agroecosistemas pampásicos. Medicina (B Aires). 1977;37(Suppl 3):128–40.
- Carballal G, Videla CM, Merani MS. Epidemiology of Argentine hemorrhagic fever. Eur J Epidemiol. 1988;4:259–74. DOIPubMedGoogle Scholar
- Mills JN, Ellis BA, McKee KT, Calderón GE, Maiztegui JI, Nelson GO, A longitudinal study of Junín virus activity in the rodent reservoir of Argentine hemorrhagic fever. Am J Trop Med Hyg. 1992;47:749–63.PubMedGoogle Scholar
- Childs JE, Korch GW, Glass GE, LeDuc JW, Shah KV. Epizootiology of hantavirus infections in Baltimore: isolation of a virus from Norway rats, and characteristics of infected rat populations. Am J Epidemiol. 1987;126:55–68.PubMedGoogle Scholar
- Glass GE, Childs JE, Korch GW, LeDuc JW. Association of intraspecific wounding with hantaviral infection in wild rats (Rattus norvegicus). Epidemiol Infect. 1988;101:459–72. DOIPubMedGoogle Scholar
- Mills JN, Ellis BA, McKee KT Jr, Maiztegui JI, Childs JE. Reproductive characteristics of rodent assemblages in cultivated regions of central Argentina. J Mammal. 1992;73:515–26. DOIGoogle Scholar
- Niklasson B, Hornfeldt B, Lundkvist A, Bjorsten S, LeDuc J. Temporal dynamics of Puumala virus antibody prevalence in voles and of nephropathia epidemica incidence in humans. Am J Trop Med Hyg. 1995;53:134–40.PubMedGoogle Scholar
- Childs JE, Krebs JW, Ksiazek TG, Maupin GO, Gage KL, Rollin PE, A household-based, case-control study of environmental factors associated with hantavirus pulmonary syndrome in the southwestern United States. Am J Trop Med Hyg. 1995;52:393–7.PubMedGoogle Scholar
- Cheek J, Bryan R, Glass G. Geographic distribution of high-risk, HPS areas in the U.S. Southwest. In: Proceedings of the 4th International Conference on HFRS and Hantaviruses; 1998 Mar 5-7; Atlanta, Georgia. Atlanta (GA): Centers for Disease Control and Prevention; 1998.
- Leirs H, Mills JN, Krebs JW, Childs JE, Akaibe D, Woollen N, Search for the Ebola reservoir in Kikwit: reflections on the vertebrate collection. J Infect Dis. 1998. In press.
- Hafner MS, Gannon WL, Salazar-Bravo J, Alvarez-Castaneda ST. Mammal collections in the Western Hemisphere. Lawrence (KS): American Society of Mammalogists; 1997.
- Mills JN, Childs JE, Ksiazek TG, Peters CJ, Velleca WM. Methods for trapping and sampling small mammals for virologic testing. Atlanta: U.S. Department of Health and Human Services; 1995. p. 61.
- Yates TL. Tissues, cell suspensions, and chromosomes. In: Wilson DE, Cole FR, Nichols JD, Rudran R, Foster MS, editors. Measuring and monitoring biological diversity: standard methods for mammals. Washington: Smithsonian Institution Press; 1996. p. 275-8.
- Yates TL, Jones C, Cook JA. Preservation of voucher specimens. In: Wilson DE, Cole FR, Nichols JD, Rudran R, Foster MS, editors. Measuring and monitoring biological diversity: standard methods for mammals. Washington: Smithsonian Institution Press; 1996. p. 265-74.
- Mills JN, Yates TL, Childs JE, Parmenter RR, Ksiazek TG, Rollin PE, Guidelines for working with rodents potentially infected with hantavirus. J Mammal. 1995;76:716–22. DOIGoogle Scholar
- Mills JN, Childs JE, Ksiazek TG, Peters CJ, Velleca WM. Métodos para trampeo y muestreo de pequeños mamíferos para estudios virológicos. Washington: Organización Panamericana de la Salud; 1998. Report No. OPS/HPS/HCT98.104.
- Greenwood RJ, Newton WE, Pearson GL, Schamber GJ. Population and movement characteristics of radio-collared striped skunks in North Dakota during an epizootic of rabies. J Wildl Dis. 1997;33:226–41.PubMedGoogle Scholar
- Braude S, Ciszek D. Survival of naked mole-rats marked by implantable transponders and toe-clipping. J Mammal. 1998;79:360–3. DOIGoogle Scholar
- Carleton MD. Systematics and evolution. In: Kirkland GL, Layne JN, editors. Advances in the study of Peromyscus (Rodentia). Lubbock: Texas Tech University Press; 1989. p. 7-141.
Figures
Cite This ArticleTable of Contents – Volume 4, Number 4—December 1998
| EID Search Options |
|---|
|
|
|
|
|
|



Please use the form below to submit correspondence to the authors or contact them at the following address:
James N. Mills, Centers for Disease Control and Prevention, 1600 Clifton Road, Mail Stop G14, Atlanta, GA 30333, USA; fax: 404-639-4436
Top