Volume 6, Number 6—December 2000
Dispatch
Mass Die-Off of Caspian Seals Caused by Canine Distemper Virus
Cite This Article
Citation for Media
Abstract
Thousands of Caspian seals (Phoca caspica) died in the Caspian Sea from April to August 2000. Lesions characteristic of morbillivirus infection were found in tissue specimens from dead seals. Canine distemper virus infection was identified by serologic examination, reverse transcriptase-polymerase chain reaction, and sequencing of selected P gene fragments. These results implicate canine distemper virus infection as the primary cause of death.
During the spring of 2000, high death rates were reported in Caspian seals (Phoca caspica) (1), which live only in the Caspian Sea and are listed as a vulnerable species by the International Union for the Conservation of Nature (2). The die-off was first reported near the mouth of the Ural River, Kazakhstan, in late April; it subsequently spread south to the Mangistau region (Figure 1). More than 10,000 seals are estimated to have died during April and May along the Kazakhstan coast. High death rates were also reported in May and June along the Apsheron peninsula of Azerbaijan and the Turkmenistan coast. We present evidence that canine distemper virus infection was the primary cause of these deaths.
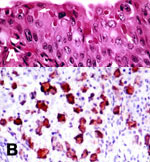
Clinical signs in infected seals included debilitation, muscle spasms, ocular and nasal exudation, and sneezing. In necropsies performed in June on eight seals from Azerbaijan (Table), no consistent gross lesions were found. However, microscopic lesions, including broncho-interstitial pneumonia, encephalitis, pancreatitis, and lymphocytic depletion in lymphoid tissues, were seen in these and four seals found in Kazakhstan in May. Multiple intracytoplasmic and rare intranuclear acidophilic inclusions, characteristic of morbillivirus infection (3), were observed in many epithelial tissue specimens (Figure 2A). Paraffin-embedded tissue sections were examined for morbillivirus antigen by an immunohistochemical technique (4). A monoclonal antibody against the nucleoprotein of phocine distemper virus, known to cross-react with canine distemper virus and cetacean morbilliviruses, was used as primary antibody. Morbillivirus antigen was detected in multiple tissues, including lung, lymph nodes (Figure 2B), spleen, brain, pancreas, liver, and epithelial tissue of the reproductive, urinary, and gastrointestinal tracts. These multisystemic tissue lesions are characteristic of distemper in terrestrial and aquatic mammals (3).
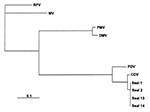
Tissues from 12 seal carcasses found on the coasts of Kazakhstan, Azerbaijan, and Turkmenistan (Table) were examined for morbillivirus nucleic acid by reverse-transcriptase polymerase chain reaction (RT-PCR). One set of universal morbillivirus primers, based on conserved sequences in the phosphoprotein (P) gene, and a second set specific for the canine distemper virus fusion (F) gene, were used in this technique (5). Tissues from nine seals were positive with both P and F primers, yielding the expected products of 429 bp and 372 bp, respectively. Selected P gene fragments were sequenced for phylogenetic comparison (Figure 3). The resulting sequences matched those of canine distemper virus and were clearly distinct from those of other members of the genus Morbillivirus, including phocine distemper virus. Except for one nucleotide change in the P gene fragment from seal 14, the sequences from Kazakhstan and Azerbaijan were identical, indicating that seals from widely separated regions of the Caspian Sea were infected by the same virus. This finding establishes spatial and temporal links between the seal deaths in these regions.
These sequences were identical to that of canine distemper virus detected in 1997 in brain tissue from a single Caspian seal in which no evidence of morbillivirus lesions was found (6). These results suggest either persistence of canine distemper virus in the Caspian seal population over a period of several years or repeated spillover from the same terrestrial reservoir.
Serum specimens from 13 seals (Table) were tested for canine distemper virus-specific immunoglobulin (Ig)M and IgG antibodies by an antibody-capture enzyme-linked immunosorbent assay (ELISA) and an indirect ELISA, respectively (7). Eight of these seals had serum IgM antibodies, and 12 had IgG antibody titers ranging from 40 to 640. These serologic data confirm recent and geographically widespread canine distemper virus infection in the Caspian seal population.
In recent years, several morbillivirus epizootics have occurred in pinniped and cetacean populations in the northern hemisphere (3). Canine distemper virus infection, the primary cause of high death rates in Baikal seals (Phoca siberica) in 1987-88 (8), was associated with a die-off in crab-eating seals (Lobodon carcinophagus) in Antarctica in 1955 (9). In both these pinniped populations, viral infection was thought to have been transmitted through contact with domestic dogs. The origin of the canine distemper virus that infected the Caspian seals is unknown, but there are anecdotal reports of contact between seals and terrestrial carnivores in this region (6). Further studies are required to determine if the latter species are infected with a canine distemper virus genetically similar to that found in the seals. The epidemiology of canine distemper virus infection, including its effects on the Caspian seal population, also remains to be investigated.
High levels of chemical contaminants have been recently identified in tissues of Caspian seals (10). As some of these substances have been shown to have immunotoxic effects in seals at the reported concentrations (11), further work is under way to determine whether pollutants contributed to these deaths.
Dr. Kennedy is head of the Diagnostic Unit of the Veterinary Science Division, Department of Agriculture and Rural Development, Belfast, Northern Ireland. His research interests include morbilliviruses of aquatic mammmals and mammalian circoviruses.
Acknowledgment
This work was partially funded by the World Bank through a donation by the Japanese Consultant Trust Fund, as well as by the Offshore Kazakhstan International Operating Company.
References
- International Society for Infectious Diseases. Reports on diseases in seals. Promed-mail program for monitoring emerging infectious diseases. Available from: URL: http://www.promedmail.org [from home page click "Search the Archives," then enter "seals" in "Search Terms'" then click "Search in - Subject (Title)]
- Baillie J, Groombridge B, eds. 1996 IUCN Red List of Threatened Animals. Gland, Switzerland: International Union for the Conservation of Nature; 1996.
- Kennedy S. Morbillivirus infections in marine mammals.J Comp Pathol. 1998;119:201–25. DOIPubMedGoogle Scholar
- Kennedy S, Smyth J, Cush PF, Duignan P, Platten M, McCullough SJ, Histopathologic and immunocytochemical studies of distemper in seals.Vet Pathol. 1989;26:97–103. DOIPubMedGoogle Scholar
- Barrett T, Visser IKG, Mamaev L, Goatley L, Van Bressem MF, Osterhaus ADME. Dolphin and porpoise morbilliviruses are genetically distinct from phocine distemper virus.Virology. 1993;193:1010–2. DOIPubMedGoogle Scholar
- Forsyth MA, Kennedy S, Wilson S, Eybatov T, Barrett T. Canine distemper virus in a Caspian seal (Phoca caspica).Vet Rec. 1998;143:662–4.PubMedGoogle Scholar
- Osterhaus ADME, Rimmelzwaan GF, Martina BEE, Bestebroer TM, Fouchier RAM. Influenza B virus in seals.Science. 2000;288:1051. DOIPubMedGoogle Scholar
- Osterhaus ADME, Groen J, UytdeHaag FGCM, Visser IKG, van de Bildt MGW, Bergman A, . Distemper virus in Baikal seals.Nature. 1989;338:209–10. DOIPubMedGoogle Scholar
- Bengston JL, Boveng P, Franzen U, Have P, Heide-Jorgensen M-P, Harkonen TL. Antibodies to canine distemper virus in Antarctic seals.Mar Mamm Sci. 1991;7:85–7. DOIGoogle Scholar
- Hall AJ, Duck CD, Law RJ, Allchin CR, Wilson S, Eybatov T. Environ Pollut. 1999;106:203–12. DOIPubMedGoogle Scholar
- De Swart RL, Ross PS, Vedder LJ, Timmerman HH, Heisterkamp SH, Van Louveren H, Impairment of immune function in harbor seals (Phoca vitulina) feeding on fish from polluted waters.Ambio. 1994;23:155–9.
Figures
Table
Cite This ArticleTable of Contents – Volume 6, Number 6—December 2000
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Seamus Kennedy, Veterinary Sciences Division, Department of Agriculture and Rural Development, Stormont, Belfast BT4 3SD, Northern Ireland; fax: +44 28 90525767
Top