Volume 7, Number 4—August 2001
THEME ISSUE
West Nile Virus
West Nile Virus
Mosquito Surveillance and Polymerase Chain Reaction Detection of West Nile Virus, New York State
Cite This Article
Citation for Media
Abstract
West Nile (WN) virus was detected in the metropolitan New York City (NYC) area during the summer and fall of 1999. Sixty-two human cases, 7 fatal, were documented. The New York State Department of Health initiated a departmental effort to implement a statewide mosquito and virus surveillance system. During the 2000 arbovirus surveillance season, we collected 317,676 mosquitoes, submitted 9,952 pools for virus testing, and detected 363 WN virus-positive pools by polymerase chain reaction (PCR). Eight species of mosquitoes were found infected. Our mosquito surveillance system complemented other surveillance systems in the state to identify relative risk for human exposure to WN virus. PCR WN virus-positive mosquitoes were detected in NYC and six counties in the lower Hudson River Valley and metropolitan NYC area. Collective surveillance activities suggest that WN virus can disperse throughout the state and may impact local health jurisdictions in the state in future years.
During the summer and fall of 1999, the metropolitan area of New York City witnessed an outbreak of human encephalitis caused by West Nile (WN) virus (1-3). This outbreak was the first evidence of WN virus infection in the Western Hemisphere (4-8) and resulted in 62 laboratory-confirmed human cases, 7 fatal (8). Evidence of WN virus infection was documented in mosquitoes, birds, horses, and humans, primarily in the New York, Connecticut, and New Jersey area of the northeastern United States, with the southernmost distribution identified in a dead WN virus-positive crow in Baltimore, MD (9-11). Laboratory investigation into the likely geographic source of the WN virus strain identified in this outbreak indicated 99% homology with a strain found in Israel in 1998 (5).
Guidelines for WN virus surveillance developed in consultation with national experts were distributed by the Centers for Disease Control and Prevention (CDC) and included recommendations for disease prevention and control (12). Surveillance activities conducted in January and February 2000 for overwintering Culex mosquitoes in New York City identified WN virus-infected specimens in underground hibernacula (13), indicating that there was a risk of virus maintenance in vector species and potential for reemergence as a human disease risk during the spring and summer months. Our report describes subsequent state and local agency efforts to establish, implement, and evaluate the mosquito and WN virus surveillance programs undertaken during 2000 to minimize risks for human and animal infection.
Local Health Department (LHD) Response
The New York State Department of Health (NYSDOH) developed a system of mosquito pool allocations that would allow rapid detection of virus should WN virus appear anywhere in the area affected by the 1999 outbreak or in peripheral counties in the general metropolitan area or surrounding lower Hudson Valley area. Based on data from the 1999 surveillance, initial priority was placed on mosquito species considered likely first indicators of virus circulation, primarily in the Culex genus. LHDs conducted early season larval surveillance and habitat descriptions, initiated habitat management and larval control in habitats where immature mosquitoes were present (focusing on container breeding, storm drain, or catch basin habitats), and developed a local database of these habitats. Adult mosquito surveillance activities could start at any time in the individual counties, and mosquito pools could be submitted to the NYSDOH Arbovirus Research Laboratory beginning the first week of June. The NYSDOH provided CO2-baited CDC miniature light traps (14) and gravid mosquito traps (15) to LHDs to conduct adult mosquito surveillance. Anticipating a total statewide weekly submission of 400 mosquito pools (up to 50 mosquitoes/pool), we provided LHDs with a weekly pool allocation and scheduled day of submission for all mosquito pools to be tested for arboviruses.
The department also provided other surveillance supplies that LHDs would need to conduct initial mosquito surveillance activities or to enhance existing programs. We divided the surveillance equipment among the counties on the basis of 1) 1999 surveillance data and recognized geographic distribution of WN virus and 2) the anticipated geographic distribution of WN virus in the metropolitan NYC and lower Hudson Valley area during 2000. We also allocated pools for eastern equine encephalitis surveillance conducted annually in Long Island and central New York. Mosquito pool allocations for 2000 were divided among the LHDs in NYC and the rest of the state on the basis of human population density and distance from the 1999 epicenter. Weekly pool allocations ranged from 90 pools for NYC, 45 pools for each county on Long Island, 40 pools for Westchester County, and 10-15 pools for counties further upstate that were conducting adult mosquito surveillance.
Mosquitoes submitted for virus testing arrived at the Arbovirus Research Laboratory on a weekly basis; some counties submitted specimens midweek and others at the end of the week to split the initial 400-pool weekly load. Mosquito pools contained specimens from a single species (or combined species such as Cx. pipiens and Cx. restuans) collected at a single site during the week. In some cases where numbers were below the target of 50 specimens per pool, collections from 2 sequential weeks or from closely adjacent sites were combined. Although the NYSDOH initially requested separation, it is unlikely that all counties effectively removed all blood-fed or gravid females from weekly collections. Therefore, some pools positive by polymerase chain reaction (PCR) may have contained blood-fed or gravid mosquitoes. Mosquito pools submitted to the Arbovirus Research Laboratory were tested for viral RNA by reverse transcription (RT)-PCR techniques following established protocols and reported as described (16). Briefly, mosquitoes were tested by TaqMan RT-PCR with two primer-probe sets. Virus isolation attempts in Vero cell culture followed the detection of PCR-positive pools, as described (16).
State Analysis of Surveillance Data
Surveillance data related to adult mosquito populations, adult mosquito pool submissions for virus testing, and laboratory test results (as well as bird, animal, and human data) were entered into the NYSDOH Health Information Network, a secure statewide health agency communication network. These surveillance data were maintained on a daily basis and available to NYSDOH and the contributing LHD. Local agency data were held confidential for 24 hours before all other LHDs were provided summary information.
All LHDs were responsible for the entry of mosquito surveillance data in the Health Information Network, as described. All surveillance mechanisms (mosquito, bird, animal, and human) provided data for this secure database. Program and laboratory review of the surveillance database, as well as laboratory entry of virus detection results, provided a current picture of mosquito populations and a virus infection registry of statewide data. When used fully and properly by the LHDs, mosquito population dynamics, rates, and location of arbovirus activity could be derived from regular review of this comprehensive database. All analyses conducted for this report were developed through the summary of the information extracted from that network.
Mosquito Surveillance
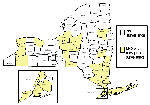
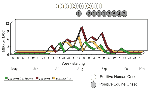
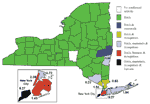
Counties conducting adult mosquito surveillance during 2000 along with a summary of their general surveillance efforts are listed in Table 1. Although 26 counties collected and submitted pools of mosquitoes for virus testing (Figure 1), PCR-positive mosquitoes were found only in the five NYC boroughs, both counties on Long Island, and four counties in the lower Hudson River Valley. Figure 2 provides the weekly numbers of mosquitoes collected by genus through the 26-week collection season from May 13 to November 4, 2000. A total of 317,676 mosquitoes were collected: 192,538 (60.6%) Culex spp., 86,034 (27.1%) Aedes (or Ochlerotatus) spp., and 39,104 (12.3%) other species. LHDs collected 28 species of mosquitoes during the 2000 adult mosquito surveillance season (3 Aedes spp., 4 Anopheles spp., 1 Coquillettidia sp., 4 Culex spp., 2 Culiseta spp., 11 Ochlerotatus spp., 1 Orthopodomyia sp., 1 Psorophora sp., and 1 Uranotaenia sp.). Of the 9,952 pools submitted for testing, 5,851 (58.8%) were Culex spp., 3,072 (30.9%) were Aedes or Ochlerotatus spp., and 1,029 (10.3%) were other species. Despite the original agreement with LHDs for a maximum weekly submission of 400 pools, submissions exceeded the maximum during 11 weeks of the season, with a peak of 1,200 pools submitted the week of August 5. Laboratory protocols were initially established at a maximum of 100 mosquitoes per pool, but that figure was reduced to 50 mosquitoes per pool in May to enhance the ability to detect virus.
Virus Surveillance in Mosquitoes
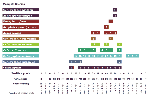
Quantified summary data related to virus distribution by county and by mosquito species are provided in Table 2. NYSDOH surveillance and laboratory testing activities during 2000 led to the identification of 363 PCR WN virus-positive mosquito pools. Results from virus isolation attempts on these PCR-positive pools are included in Table 3. We are also aware of the detection of a single PCR-positive pool of Culex pipiens/restuans collected by U.S. Army Center for Health Promotion and Preventive Medicine staff in Brooklyn (B. Pagac, pers. comm.; data not included). Although NYC and Long Island LHDs submitted only 5,324 (53%) of the 9,952 total mosquito pools, 298 (82%) of 363 PCR WN virus-positive pools were collected in these counties. Only 65 (18%) of the 363 PCR WN virus-positive pools were identified in four counties north of NYC and Long Island, despite accounting for 26% (2,549/9,952) of the total pools submitted. Most PCR-positive pools (72%, 47/65) from upstate were from Rockland County. Similarly, 341 (94%) of the 363 PCR-positive pools were Culex mosquitoes, primarily in the group of Cx. pipiens/restuans mosquitoes that were not separated because of loss of morphologic characteristics during collection or natural aging. Although we found 79 pools of Cx. pipiens PCR positive for WN virus, none of 238 pools of Cx. restuans were positive. Twenty PCR WN virus-positive pools of Aedes or Ochlerotatus species mosquitoes were identified, primarily from Ae. vexans (10 pools) and Oc. japonicus (5 pools).
Temporal analysis of mosquito-based WN virus surveillance is illustrated in Figure 3 for all mosquito species (or groups) during the 2000 season. The first PCR evidence of WN virus infection in mosquitoes occurred in specimens collected the week of July 1, 2000, with one WN virus-positive pool each in Cx. pipiens collected in Richmond County (NYC) and Oc. japonicus collected in Westchester County. WN virus activity continued to amplify as the season progressed, with increasing numbers of PCR-positive mosquito pools, increasing geographic distribution of infection, and WN virus infection identified in additional mosquito species. By the end of the season, WNV was identified by PCR in eight mosquito species from a wide geographic area of Long Island and the lower Hudson River Valley, north to Columbia County. The northernmost PCR WN virus-positive mosquito pool was Oc. japonicus collected in Columbia County the week of September 16, 2000. The latest PCR WN virus-positive pool was identified in Cx. pipiens/restuans collected the week of October 14, 2000, from Suffolk County. (The latest pools of positive mosquitoes collected in 1999 were also collected during mid-October.) A complete list of PCR WN virus-positive mosquito species detected in 2000 appears in Table 2.
We calculated trap-type productivity by species for the six most abundant mosquito species collected statewide during 2000. Dry ice-baited CDC light traps captured 98% of the Ae. vexans, 83% of the Oc. triseriatus, 77% of the Cx. salinarius, 55% of both total Cx. pipiens and Cx. pipiens/restuans combined, and 21% of the Oc. japonicus. The only species collected in predominantly greater numbers by the gravid traps was Oc. japonicus (76% of total collections). Although gravid traps collected only 30% of the total number of mosquito pools submitted for virus testing during 2000, 56% of the PCR-positive pools came from gravid trap collections. None of the mosquito pools (n=135) from diurnal resting boxes or aspiration collections (n=11) had evidence of virus infection. However, 205 (6.9%) of 2,957 pools from gravid traps and 158 (2.3%) of 6,828 pools from CDC light traps were PCR positive for WN virus. Since mosquitoes attracted to gravid traps for oviposition would have fed on a host several days previously, a widespread presence of infected hosts could have resulted in these mosquitoes being more likely to acquire virus, thereby explaining the threefold difference in PCR-positive rates in gravid trap collections.
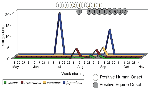
We also investigated the contribution of species minimum infection ratios (MIRs) per 1,000 mosquitoes throughout the season. MIRs (based on PCR-positive tests) were evaluated by individual pool sizes for each species and by analysis of weekly and seasonal aggregated species data within and among LHDs. Figures 4 and 5 illustrate the weekly MIRs by species for each genus (Aedes, Ochlerotatus, and Culex) through the adult mosquito surveillance season in counties that had evidence of human or equine cases (8). (These figures represent the recapitulation of results after all laboratory data were completed.) WN virus infection (confirmed by PCR) in Culex spp. began in early July and continued through the summer, concurrent with human case onsets; weekly MIRs ranged from 1 to 3.5 per 1,000, with a subsequent peak in early October. Although 80 to 100 pools of Aedes or Ochlerotatus were submitted during June and July, the PCR WN virus-positive Oc. japonicus in early July resulted in relatively high genus-specific MIRs. A second peak of elevated Aedes and Ochlerotatus MIRs occurred in late August and September, concurrent with an observed peak of equine cases. Further review of these data from counties where human or equine cases occurred, using MIR and multifactorial analyses, is required.
By the end of the 2000 surveillance season in New York, PCR WN virus-positive specimens were detected throughout the state (Figure 6). While human disease was restricted to four NYC boroughs (Staten Island, Brooklyn, Manhattan, and Queens), PCR evidence of WN virus infection in mosquitoes was detected in all five NYC boroughs, four adjacent metropolitan counties, and two counties further upstate. Evidence of infection in avian hosts, however, was detected throughout the state (except for Chenango County) (17). Despite the submission of 2,660 pools of primary vector species from 17 upstate counties outside the immediate metropolitan area, we detected PCR evidence of WN virus only in Orange (two pools of Oc. japonicus and one each of Cx. pipiens and Cx. pipiens/restuans) and Columbia (one pool of Oc. japonicus) counties. However, MIRs generally reflected the pattern of human cases and dead crow sightings per square mile (17) in three categories: 1) a combination of high number of human cases, dead crow sightings per square mile, and an overall seasonal MIR of 5.27/1,000 tested in Staten Island; 2) a small number of human cases, moderate number of dead crow sightings per square mile, and seasonal MIRs ranging from 0.18 to 2.36/1,000 for counties in and near NYC; and 3) no human cases, low number of dead crow sightings per square mile, and seasonal MIRs ranging from 0 to 0.86/1,000 for upstate counties (Figure 6). Documented WN virus transmission to humans and horses was limited to a much smaller geographic area of New York State, and, over the last two seasons, has included only NYC, Long Island, Westchester County, and Orange County (roughly 50 miles north of NYC).
Review of specific local human and mosquito population data will be addressed in separate publications. Our work summarizes statewide surveillance components and identifies some potential flaws due to the geographic aggregation of data. In addition, surveillance may not have been uniformly applied across all municipalities. However, review of these initial surveillance data will lead investigators to design research projects that will better enable the public health community to answer questions that continue to arise (e.g., regarding virus transmission, host pathogenicity, immunology, interventions, sociologic aspects, and vaccination or treatment issues).
There may be some risk of faulty interpretation in a retrospective review of statewide field and laboratory data. In addition, we have not fully evaluated the potential effect of mosquito control operations on overall mosquito populations. However, these data provide necessary baseline information related to virus ecology and infection dynamics. The detection of MIRs associated with individual mosquito species may help public health agencies and academic institutions to focus on specific species with high levels of vectorial capacity (18).
The surveillance system in New York State during 2000 led to the submission of >300,000 mosquitoes in almost 10,000 pools for arbovirus screening; most were derived from LHDs that had not previously conducted mosquito or arbovirus surveillance. These efforts required regular internal discussions on laboratory capacity, steps that could be taken to improve it, and proper use of laboratory capacity to answer both routine surveillance and research demands on limited resources.
Although there is an inherent procedural lag between mosquito collection and the availability of laboratory results, this initial analysis supports the natural presence of WN virus PCR-positive Culex, Aedes, and Ochlerotatus mosquitoes 2 weeks before onset of human illness. In addition, peak Culex infection during the period of prominent human infection appears to be associated with a peak of PCR WN virus-positive Aedes and Ochlerotatus immediately before and during the onsets of equine cases, which occurred several weeks after the peak of human onsets. However, laboratory confirmation of positivity for all virus surveillance mechanisms (mosquitoes, birds, humans, and horses) usually lagged behind specimen collection or onset dates by several weeks. In addition, infection dates for human and horse cases are likely to precede onset dates by several days to weeks, depending on the individual incubation periods.
Interpretation of the influence of trap type on yield of PCR-positive pools is complicated by the potential for detecting virus that may have been present in the host on which individual mosquitoes may have fed. Ideally, blood-fed or gravid mosquitoes should be separated from unfed females before laboratory testing. Clearly, however, a blood-fed female mosquito one week will become a host-seeking female a week or two later, and full understanding of the transmission dynamics of WN virus may require this separation, along with specific determination of host meals. Data indicate that agencies with limited budgets may be able to identify the local presence and natural circulation of WN virus more efficiently by focusing on gravid trap-based programs.
Initial analyses of MIRs associated with individual mosquito species indicate that certain species, especially Cx. salinarius, Cx. pipiens, Oc. japonicus and Oc. triseriatus, may play major roles in the natural transmission of WN virus among animal hosts. Several of these species (or species complexes) may be involved with an as-yet-unknown reservoir host capable of maintaining the virus in the absence of overt host pathogenicity. Note, however, that no infectious virus was isolated in approximately half of the mosquito pools tested with detectable RNA. WN virus RNA was detected in five pools of Oc. japonicus, for example, but no infectious virus was isolated from these mosquitoes (19). The level of infectious virus may have been too low to be detected and may have increased with further extrinsic incubation, complicating the interpretation of MIRs.
The interrelationships, ecologic niches, and host-meal preferences, for example, of the various Culex species may play a major role in the entire ecology of the virus now that it has been introduced to a completely naive hemisphere. In addition, the MIRs of Culex and other species associated with the apparently bimodal human and equine case distribution may point to a complex mosquito vector relationship not only among the various Culex species, especially Cx. salinarius and Cx. pipiens and perhaps Cx. restuans, but also with other more prominent mammal feeding species such as Ae. vexans, Oc. triseriatus, and Oc. japonicus. What will happen as this virus continues to spread into other areas of North or Central America where aggressive and more numerous mammal-feeding species exist? How will human disease risks increase if Oc. sollicitans, Cx. tarsalis, Ae. aegypti, or Anopheles species become competent vectors under normal field conditions? Are non-mosquito vectors (such as ticks, mites, or black flies) contributing to the total ecologic cycle (e.g., transmission and overwintering) of this virus in nature? Intervention programs must be established that can account for answers to these questions and maintain our original goals of minimizing the risks for human and other animal infections. Given the recognized ability of WN virus to survive in hibernating mosquitoes in the northeastern United States (20) and the current distribution of the virus in birds, mammals, and mosquitoes along the Atlantic seaboard, public health agencies in affected and neighboring states should be prepared to address the emergence of this virus in their jurisdictions in future years.
Another major goal that public health agencies must face is developing a more timely virus surveillance system. This system should allow detection of the virus in mosquito or bird host systems in sufficient time to permit an appropriate intervention to minimize or prevent further mammal infection. Our current surveillance and laboratory testing systems have inherent time lags that may preclude our ability to intervene against infected adult mosquitoes on a timely basis, before humans are exposed.
Dr. White is the Director of the Arthropod-Borne Disease Program and Director of the Tick-Borne Disease Institute of the New York State Department of Health and Associate Professor, School of Public Health, State University of New York at Albany. His current research interests include field ecology and modeling of vector-borne diseases.
Acknowledgment
We gratefully acknowledge Y. Hagiwara, N. Panebianco, J. Rososki, J. Napoli, R. Falco, T. Daniels, L. Grady, C. Huang, M. Fage, J. Gebbia, S. Bamberger, N. Pardanani, M. Martin, S. Trock, B. Wallace, P. Smith, D. Morse, D. Whalen, and A. Novello.
References
- Centers for Disease Control and Prevention. Outbreak of West Nile-like viral encephalitis--New York, 1999. MMWR Morb Mortal Wkly Rep. 1999;48:845–9.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: West Nile Virus encephalitis--New York, 1999. MMWR Morb Mortal Wkly Rep. 1999;48:944–6, 955.
- Centers for Disease Control and Prevention. Update: West Nile-like viral encephalitis-New York, 1999. MMWR Morb Mortal Wkly Rep. 1999;48:890–2.PubMedGoogle Scholar
- Hubalek Z, Halouzka J. West Nile fever-a reemerging mosquito-borne viral disease in Europe. Emerg Infect Dis. 1999;5:643–50. DOIPubMedGoogle Scholar
- Lanciotti RS, Roehrig JT, Duebel V, Smith J, Parker M, Steele K, Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science. 1999;286:2333–7. DOIPubMedGoogle Scholar
- Hayes CG. West Nile fever. In: Monath RD, editor. The arboviruses: epidemiology and ecology. Boca Raton (FL): CRC Press, Inc.; 1989.
- Savage HM, Ceianu C, Nicolescu G, Karabatsos N, Lanciotti R, Vladimirescu A, Entomologic and avian investigations of an epidemic of West Nile fever in Romania in 1996, with serologic and molecular characterization of a virus isolate from mosquitoes. Am J Trop Med Hyg. 1999;61:600–11.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: West Nile virus activity--eastern United States, 2000. MMWR Morb Mortal Wkly Rep. 2000;49:1044–7.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. West Nile virus activity--New York and New Jersey, 2000. MMWR Morb Mortal Wkly Rep. 2000;49:640–2.PubMedGoogle Scholar
- Anderson JF, Andreadis TG, Vossbrinck CR, Tirrell S, Wakem EM, French RA, Isolation of West Nile virus from mosquitoes, crows and Cooper's hawk in Connecticut. Science. 1999;286:2331–3. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Guidelines for surveillance, prevention, and control of West Nile virus infection--United States. MMWR Morb Mortal Wkly Rep. 2000;49:25–8.PubMedGoogle Scholar
- Nasci RS, White DJ, Stirling H, Oliver J, Daniels TJ, Falco RS, West Nile virus isolates from mosquitoes in New York and New Jersey, 1999. Emerg Infect Dis. 2001;7:626–30. DOIPubMedGoogle Scholar
- Newhouse VR, Chamberlain RW, Johnston JF, Sudia WD. Use of dry ice to increase mosquito catches of the CDC miniature light trap. Mosq News. 1966;26:30–5.
- Reiter P. A portable, battery-powered trap for collecting gravid Culex mosquitoes. Mosq News. 1983;43:496–8.
- Shi P-Y, Kauffman EB, Ren P, Felten A, Tai JH, Dupuis AP II, High throughput detection of West Nile virus RNA. J Clin Microbiol. 2001;39:1264–71. DOIPubMedGoogle Scholar
- Eidson M, Miller J, Kramer L, Cherry B, Hagiwara Y; WNV Bird Mortality Analysis Group. Dead crow densities and human cases of West Nile virus, New York State, 1999. Emerg Infect Dis. 2001;7:662–4. DOIPubMedGoogle Scholar
- Spielman A, James AA. Transmission of vector-borne disease. In: Warren KS, Mahmood AAF, editors. Tropical and geographical medicine. 2nd ed. New York: McGraw Hill; 1990. p. 146-59.
- Bernard KA, Maffei JG, Jones SA, Kauffman EB, Ebel GD, Dupuis AP, West Nile virus infection in birds and mosquitoes, New York State, 2000. Emerg Infect Dis. 2001;7:679–85. DOIPubMedGoogle Scholar
- Nasci RS, Savage HM, White DJ, Miller JR, Cropp BC, Godsey MS, West Nile virus in overwintering Culex mosquitoes, New York City, 2000. Emerg Infect Dis. 2001;7:742–4. DOIPubMedGoogle Scholar
Figures
Tables
Cite This Article1E.B. Kauffman, K.A. Bernard, G.D. Ebel, P-Y. Shi, S.A. Jones, A.P. Dupuis II, J. Maffei, K.A. Ngo, D.C. Nicholas, and D. Young.
2J. Miller, City of New York Department of Health; D. Ninivaggi, Suffolk County Department of Health Services; C. Hodgeman, G. Terrillion, Nassau County Health Department; A. Huang, P. Yang-Lewis, Westchester County Health Department; M. Anand, B. Hunderfund, Rockland County Health Department; M. Smith, Orange County Health Department; J. Storm, S. Capowski, Dutchess County Health Department; D. Palen, R. Krom, G. Myers, Ulster County Health Department; S. Lukowski, Albany County Health Department; J. Zukowski, Columbia County Health Department; R. Burdick, Onondaga County Health Department; and J. Mysen, United States Military Academy, West Point, New York.
Table of Contents – Volume 7, Number 4—August 2001
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Dennis J. White, Director, Arthropod-borne Disease Program, New York State Department of Health, 632 Corning Tower, Empire State Plaza, Albany, New York 12237 USA; fax: 518-473-1708
Top