Volume 11, Number 10—October 2005
Research
Plasmodium falciparum Spatial Analysis, Western Kenya Highlands
Cite This Article
Citation for Media
Abstract
We carried out a population-based study to determine the unbiased, age-specific Plasmodium falciparum prevalence, asexual and sexual parasite density, and spatial distribution to establish rates of infection at a site in western Kenya. Three cross-sectional surveys were carried out in western Kenya highlands. Blood samples were taken from 1,388 persons from 6 months to 75 years of age. Parasite prevalence and densities in the population decreased with age and distance from valley bottoms. Children from 1 to 4 years of age had the highest parasite prevalence (38.8%–62.8%); in adults, prevalence declined to 2.9%–24.1%. Malaria prevalence declined by an average of 19% from July to December 2002 across age groups. These observations suggest that parasite transmission is intense at this altitude. Asexual parasite density indicated clustering near major vector breeding habitats. Variability in seasonal prevalence indicates transmission instability and susceptibility to epidemics.
The reemergence of epidemic malaria in the East African highlands (elevation >1,500 m above sea level) is a public health problem (1). Research indicates that the mechanisms leading to epidemic malaria in the highlands are complex and are probably due to the concerted effects of factors such as topography (2), hydrology (3), climate variability (4), land-use/land-cover change (5), and drug resistance (6). Effective disease control calls for a clear understanding of the interaction between these epidemiologic factors.
Perennial malaria transmission in the lowlands has been attributed to high vector densities throughout the year (7). Inhabitants of the basin region of Lake Victoria, western Kenya, experienced up to 300 infective bites per year (8,9). Vector density and transmission intensity in the highlands are much lower than in the lowlands. For example, a transect study from lowland (300 m elevation) to highland (1,700 m elevation) in the Usambara Mountains in Tanzania found a >1,000-fold reduction in transmission intensity between the holoendemic lowland and the hypoendemic highland plateau (7). At high altitudes in the highlands and on hilltops, where malaria transmission intensity is low, human populations have poorly developed immunity to malaria because exposures are infrequent (10), and persons are vulnerable to severe clinical illness and complications from Plasmodium infection (11,12). High risk for severe malaria is seen in persons living in areas with low-to-moderate transmission intensities (13). In such areas, the proportion of asymptomatic persons is usually lower than in high-transmission areas, where P. falciparum prevalence and parasite density varies little between seasons (14).
Although malaria incidence has been increasing in the East African highlands (15), the extent and distribution of malaria infections in the asymptomatic human population are largely unknown. Data on malaria in the western Kenya highlands are derived from hospital clinical records (16,17) and do not provide a population-based, epidemiologic profile or information on the unbiased prevalence of malaria in local highland populations. Bias in hospital data can arise from poverty, self-medication, and lack of access to hospitals. Detailed parasitologic data are also not normally available from hospital clinical records. Because data on malaria prevalence in local residents are unavailable, we do not know whether whole populations are susceptible to malaria or the risk is skewed toward children, as is seen in malaria-holoendemic regions. In this study, we investigated malaria parasitologic profiles in a population living in a highland zone in the Kakamega district, western Kenya, where epidemics have been reported (4) to determine age-specific parasitemia prevalence, age-specific parasite densities, and the spatial distribution of infections.
Study Site
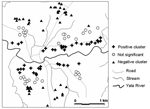
The study site is located in Iguhu village (0°17´N, 34°74´E, elevation 1,450–1,580 m above sea level), Kakamega district, western Kenya, with population ≈11,000. This area experiences 2 rainy seasons and averages 2,000 mm rainfall per year. The long rainy season usually occurs between April and May, with an average monthly rainfall 150–260 mm, while the short rainy season usually occurs between September and October, with an average monthly rainfall 165 mm. Malaria prevalence peaks usually lag 1–2 months after the rain. The mean annual daily temperature is 20.8°C. The area has experienced extensive deforestation and swamp reclamation in recent years as a result of rapid human population growth and the demand for settlement and agricultural land; therefore, only patches of forest remain. Malaria vectors in the area are Anopheles gambiae sensu stricto and A. funestus (4,18). Maize is the principal subsistence crop, although vegetables are grown on small, irrigated plots in valley bottoms. The area is bisected by the Yala River (Figure 1); most mosquito larval habitats were found on river banks in the bottom of the valley and on the banks of streams during both dry and rainy seasons (18).
Study Design
The working hypothesis was that no difference would be seen in the prevalence among various age groups and that infections would be randomly distributed. The binomial model was used to estimate the confidence interval (CI). Because the prevalence of malaria was not well known in the area, a 50% estimate, which gives the best sample size, was used for the peak-transmission season, and a 25% estimate was used for the low-transmission season. We calculated the 2 sample sizes with a 95% CI and precision level of 5%:
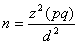
In this equation, n is the sample size, z is the critical value of the standard normal distribution at the 5% level (1.96), p is the malaria prevalence estimate, q = 1 – p, and d is the precision level. The population size was estimated to be 3,000. The sample size obtained was 341 persons for the peak-transmission season and 263 for the low-transmission season.
Study Population and Data Collection
The study, which involved 1,388 participants, was carried out in 3 cross-sectional surveys undertaken in July 2002 (n = 396), December 2002 (n = 283), and June 2003 (n = 709). Peak malaria transmission occurs 1–2 months after the rainy season; thus, surveys were conducted 1 month after the rains. The short rains before December are usually not adequate to increase transmission, and this period is considered entomologically dry. Figure 1 shows the spatial distribution of residents examined for Plasmodium infections in July 2002. The sample size for each age group during each sampling period is shown in the Table. Adults (>18 years of age) were recruited into the study upon giving an informed consent; consent for children (<18 years) was provided by the participants and their parents or guardians. Scientific and ethical clearance was given by Kenya Medical Research Institute and State University of New York at Buffalo ethical review boards. Inclusion criteria were provision of informed consent, age >6 months at recruitment, and no reported chronic or acute illness except malaria. Blood samples were collected by the standard finger-prick method, and thick and thin smears were prepared on labeled slides. The thin and thick blood smears were air dried. The thin smears were fixed in methanol and stained in 4% Giemsa for 30 min with the thick smear. Two experienced technicians examined the slides under ×1,000 oil immersion to identify and count the parasite species. Random checks were carried out on the slide counts by independent microscopists to ensure quality control. Parasite density was scored against 200 leukocytes when the slide was positive; otherwise, the whole slide was carefully scanned before being declared negative. Parasite densities were converted to number of parasites per microliter of blood, assuming a leukocyte count of 8,000 cells/μL (19).
We examined the monthly malaria infection dynamics in primary school children, age 6–14, in the study area from June 2002 to June 2003, with the microscopic test. Daily rainfall data were collected by using automated weather stations (HOBO, Onset Computer Corporation, Bourne, MA, USA). These data were offloaded monthly from the weather stations.
Data Analysis
Most of the homes of the participants in the prevalence survey were georeferenced by using a global positioning system (GPS) unit. A database was created with participants' names, ages, GPS location, and parasite density. Participants' names were coded for confidentiality. The relationship between P. falciparum prevalence and parasite density was determined across age, season, and distance to the nearest stream/valley. To examine the effects of age on parasite prevalence and infection density, ages were stratified into 6 age groups: <1, 1–4, 5–9, 10–14, 15–19, and >19 years (20). Age-specific prevalence was determined by expressing positive blood smears as a percentage of all examined blood smears; only positive slides were considered when the geometric mean parasite density was calculated for each age category. The χ2 test was used to determine differences in prevalence among age groups for each survey. Analysis of variance (ANOVA) with logarithm-transformed parasite density was used to examine the difference among age groups and among surveys in parasite density. Nonspatial statistical analyses were conducted by using the JMP statistical package (SAS Institute Inc, Cary, NC, USA).
To determine the spatial distribution pattern of malaria-infected residents, ArcView 3.3 (Environmental Systems Research Institute, Redlands, CA, USA) was used to create spatial-distribution maps of infected and uninfected participants for the 3 surveys. P. falciparum infections were tested for clustering by household with the global spatial statistic, the K function, weighted by parasite density (21–23) by using Point Pattern Analysis software (San Diego State University, San Diego, CA, USA). The global weighted K function, L(d), examines the spatial dependence of malaria infection by household over a wide range of scales of pattern (21). The observed L(d) function values were tested against the null hypothesis that the spatial distribution of all infected residents in the study area was random, by using 1,000 Monte Carlo iterations. Because children and adults showed significantly different prevalence and parasite density, the global spatial cluster analysis was conducted separately for the 0- to 9-year age group, the 10- to 19-year age group, and adults (>19 years old). The local spatial statistic Gi*(d) (24) was computed to assess clustering of high parasite densities near a particular transmission source, the Yala River, where most vector breeding sites were found (18), for age groups 0–9 and 10–19 years, but not for adults because sample sizes were too small. To correct for multiple comparisons when using Gi*(d), significance levels were determined by using Table 3 in reference (25).
Plasmodium Prevalence and P. falciparum Density
In addition to P. falciparum, we found P. malariae and P. ovale in our study populations. P. falciparum constituted 97.1%, 94.9%, and 94.6% of malaria cases in July and December 2002, and June 2003 surveys, respectively. P. malariae constituted 2.9%, 5.1%, and 4.6% of malaria cases, while P. ovale was observed only in June 2003 (0.8%). P. malariae was often seen in mixed infection with P. falciparum. For example, 83.3% of P. malariae infections in July 2002 occurred with P. falciparum, while 50% of P. malariae infections in December 2002, and 81.8% of P. malariae infections in June 2003, were accompanied by P. falciparum.
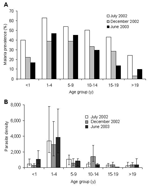
P. falciparum prevalence varied significantly among age groups (homogeneity χ2 = 95.82, df = 5, p<0.001) and sampling months (homogeneity χ2 = 46.81, df = 2, p<0.001; Figure 2A). Malaria prevalence for all age groups ranged from 50.1% in July 2002 to 27.1% in December 2002. Children 1–9 years of age consistently had the highest prevalence (average 47.0%) in the 3 cross-sectional surveys, while adults >19 years of age showed the lowest prevalence (average 9.5%). The mean parasite density did not vary significantly among survey months (ANOVA, F = 0.53, df = 2, 483, p = 0.59) but did vary significantly among age groups (F = 21.17, df = 5, 483, p<0.001; Figure 2B). For example, children 1–4 years of age had the highest parasite density (geometric mean of 3,469.7 infected erythrocytes/μL blood, 95% CI 2,328.0–5,171.3). This amount was >7-fold higher than the average parasite density among all other age groups (477.8 infected erythrocytes/μL, 95% CI 265.6–715.1; Tukey-Kramer HSD [honestly significant difference] test, p<0.001; Figure 2B).
P. falciparum Gametocyte Prevalence and Density
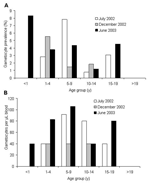
Overall, P. falciparum gametocyte prevalence did not differ significantly among the 3 surveys (homogeneity χ2 = 1.34, df = 2, p = 0.51) or age groups (homogeneity χ2 = 9.48, df = 5, p = 0.09; Figure 3A). The mean gametocyte prevalence was 2.7% (range 0%–8.3%; Figure 3A). Similarly, gametocyte density did not vary significantly among sampling occasions (ANOVA, F = 0.71, df = 2, 31, p = 0.50) or age groups (F = 0.84, df = 4, 31, P = 0.51; Figure 3B). The geometric mean gametocyte density was 71.7 gametocytes/μL (95% CI 53.8–95.6).
During the study period (June 2002 to June 2003), malaria prevalence showed a general decreasing trend (Figure 4). However, the geometric mean parasite density varied among months. The months with peak parasite density appeared to be 1–2 months behind rainfall peaks.
Spatial Distribution of Parasite Density
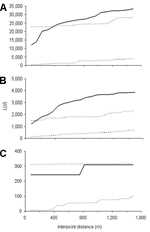
The global weighted K function, L(d), was used to examine the spatial dependence of malaria infection by household over an interpoint distance of 100–1,400 m for each of the 3 age groups (0.6–9, 10–19, and >19 years). Figure 5 shows measures of the observed L(d) and the 95% CI plotted for various values of interpoint distance for the July 2002 survey. The spatial distribution of infections is considered evenly dispersed if the observed K function values are below the lower limit of the 95% CI, clustered if above the upper limit, or random if within the 95% CI. The weighted K function indicated that the parasite density distribution pattern was significantly different than expected under complete spatial randomness for age groups 0.6–9 (Figure 5A) and 10–19 years (Figure 5B), but was random in adults (Figure 5C). Spatial clustering for age groups 0.6–9 and 10–19 years occurred at an interpoint distance >150 m. This pattern was consistent with 2 other surveys done in December 2002 and June 2003 (data not shown).
Local spatial clustering analysis was performed on parasite densities to determine whether higher infection densities were clustered near the Yala River, where most vector breeding sites existed. Figure 6 shows an example of local spatial clustering for age group 0.6–9 years for the July 2002 survey. We found that parasite densities were positively clustered for children living <500 m from the Yala River and negatively clustered for children residing >1,000 m from the river. That is, children living near the river had significantly higher parasite densities than would be expected in random distribution, whereas those farther away from the river exhibited significantly lower parasite densities. Clustering of parasite densities in age group 10–19 years showed a similar pattern, but fewer households had either positive or negative clustering. Similar results were found for the December 2002 and June 2003 surveys (data not shown). Therefore, the patterns of P. falciparum parasite density clustering, as shown in Figures 5 and 6, suggest a relationship between a house's distance from the Yala River and clustering of high parasite densities.
In this study, we found that children 1–4 years of age had a 7-fold greater number of parasites in their blood as compared to persons >5 years of age, irrespective of season. The rapid fall in parasite density as age increases suggests age-dependent immunity to P. falciparum parasites. The low prevalence of infections among adults further supports this notion. However, the highland topography affects drainage and the distribution of mosquito breeding habitats and therefore also affects exposure to infections. For example, 90% of adult mosquitoes in the study area were collected <300 m from valley bottoms (26). Nonuniform exposure patterns to infections as a result of topography and hydrology would lead to similar spatial immunologic profiles. Thus, within the highland human populations, varying states of transmission stability are found.
John et al. carried out an epidemiologic study at an altitude of 2,134 m in the Uasin Gishu district of western Kenya (27). They found no difference in reinfection rates between children and adults, which indicates that malaria transmission could be unstable at this altitude. Whether infections translated into clinical disease at the same rate in children and adults is not clear (28). Hay et al. (17) reported that approximately two thirds of hospital admissions in the Kisii district (elevation 1,500–1,800 m) in the western Kenya highlands were children <15 years of age. Our population-based malaria prevalence data from a village in the Kakamega district (elevation 1,450–1,580 m) indicate that children (1–4 years of age) have the highest malaria prevalence and parasite density, a condition similar to that seen in the holoendemic lowlands. Few records exist of detailed population-based parasitologic and clinical studies of malaria in the highlands of western Kenya, which is why classifying the stability of transmission is difficult. Shililu et al. (3) reported prevalence rates in asymptomatic children from 44% in the dry season to 55.4% in the wet season in Mumias, Kakamega district (≈1,500 m above sea level), western Kenya. During the 1990 malaria epidemic in the Uasin Gishu district in western Kenya (elevation 2,000 m), Some (29) reported a prevalence of 72% in the general population. In the same year, Ayisi et al. (30) observed a mean prevalence of 39.2% in the general population at the Belgut division of the Kericho district (elevation 1,800 m). Our data fall within the range observed by other investigators at similar altitudes. However, we also observed a significant interannual and seasonal variability in malaria prevalence at this highland site. The variation in parasite prevalence between the wet and dry seasons could be explained by differences in vector abundance. For example, the mean monthly anopheline vector abundances increased by 6- to 8-fold in the long rainy season compared to the dry season (26).
The prevalence of P. falciparum malaria in school children in the low-altitude region of Lake Victoria basin (elevation ≈1,200 m) adjacent to the highlands reaches >80% (31–34), which is much higher than malaria prevalence in the highlands. Therefore, in the highlands, a high proportion of residents was susceptible to infection and clinical disease. Under conditions of hypertransmission, outbreaks and epidemics would result because medical facilities would be unable to cope with the number of persons affected. Malaria instability in this area is related to the proportion of susceptible persons and the variability in transmission. Children 1–4 years of age had the highest parasite densities in their blood; after 5 years of age, densities dropped dramatically. The low prevalence of infections in adults suggests that these persons have developed some degree of immunity as a result of lifelong exposure.
The mean parasite density observed in the 1- to 4-years age group at Iguhu was similar to that observed in the <1-year age group in the lowlands (32), which suggests immunity develops more slowly in the highlands. Still, older persons had 7-fold lower parasite loads, which indicates functional, age-dependent immunity.
The age-specific profiles of gametocyte prevalence in the highland site were much lower than those observed in the lowlands; e.g., infants (1–9 months) had a prevalence of 86% in the lowlands, while in the highlands prevalence in this group was 2.9%. Children 5–9 years of age had the highest rate of infection with gametocytes (4.9%) at Iguhu, which is comparatively lower than that reported in the lowland (40%) for the same age group (32). The reservoir of malaria infections in the highlands is much lower than in the lowlands. For example, Ayisi et al. (30) reported a mean gametocyte prevalence of 1.8% in the Kericho district in the western Kenya highlands, while we observed a mean gametocyte rate of 2.8% in the Kakamega district. By contrast, Githeko et al. (32) reported a gametocyte prevalence of 39.1% during the rainy season in the Kisumu district and a rate of 10.8% in the dry season in the Suba district (35). Similar to observations made in the lowlands (32), the mean gametocyte density in highland residents did not vary significantly among age groups. Compared to what was seen in the lowlands, the proportion of persons carrying gametocytes (the infectious reservoir) in our highland site was 5- to 100-fold less, which suggests a weak transmission system in the highlands.
Spatial analysis of asexual parasite densities indicated clustering in relation to distance from major larval breeding habitats and in relation to age. This phenomenon was not observed in persons living >1,000 m from major breeding sites, which suggests that the rate of parasite transmission may be higher closer to major breeding habitats. This finding is consistent with the spatial distribution of indoor resting vector densities in the area. Our results are consistent with the fact that the risk of malaria is strongly associated with distance from breeding sites (36,37).
This cross-sectional study in a highland site where malaria epidemics have been reported in the past shows that transmission is intense, particularly after the rainy season, but variable with regard to season and distance from major mosquito breeding habitats. The risk of infection is highly variable within the site, and subsequently, the stability of transmission may reflect this variability. A large host population is available for infection before periods of hypertransmission. Although the population has a functional age-dependent immunity to malaria, as indicated by parasite densities, its development is slower than that found in the holoendemic lowlands, which suggests that this population may be more susceptible to malaria infections and more prone to epidemics compared to the lowland populations.
Mr Munyekenye is a research associate at the Center for Vector Biology and Control Research, Kenya Medical Research Institute, Kisumu, Kenya. His research focuses on malaria parasite ecology and vector-parasite interactions.
Acknowledgments
We thank all the participants in the study and the technical assistance of J. Otete, C. Okoth, S. Apopo, and J. Maritim.
This article is published with permission of the Director of Kenya Medical Research Institute. The work was supported by NIH grants R01 A150243 and D43 TW01505 (to G.Y.).
References
- Githeko A, Clive S. The history of malaria control in Africa: lessons learned and future perspectives. In: Ebi K, Smith J, Burton I, editors. Integration of public health with adaptation to climate change: lessons learned and new directions. London: Francis and Taylor; 2005.
- Balls M, Bodker R, Thomas C, Kisinza W, Msangeni H, Lindsay S. Effect of topography on the risk of malaria infection in the Usambara Mountains, Tanzania. Trans R Soc Trop Med Hyg. 2004;98:400–8. DOIPubMedGoogle Scholar
- Shililu J, Maier W, Seitz H, Orago A. Seasonal density, sporozoite rates and entomological inoculation rates of Anopheles gambiae and Anopheles funestus in a high-altitude sugarcane growing zone in Western Kenya. Trop Med Int Health. 1998;9:706–10. DOIPubMedGoogle Scholar
- Githeko A, Ndegwa W. Predicting malaria epidemics using climate data in Kenyan highlands: a tool for decision makers. Glob Change Hum Health. 2001;2:54–63. DOIGoogle Scholar
- Lindblade K, Walker E, Onapa A, Katungu J, Wilson M. Land use change alters malaria transmission parameters by modifying temperature in a highland area of Uganda. Trop Med Int Health. 2000;5:263–74. DOIPubMedGoogle Scholar
- Githeko A, Lindsay S, Confalonieri U, Partz J. Climate changes and vector borne diseases: a regional analysis. Bull World Health Organ. 2000;78:1136–47.PubMedGoogle Scholar
- Bodker R, Akida J, Shayo D, Kisinza W, Msangeni H, Pedersen E, Relationship between altitude and intensity of malaria transmission in the Usambara Mountains, Tanzania. J Med Entomol. 2003;40:706–17. DOIPubMedGoogle Scholar
- Beier JC, Perkins PV, Onyango FK, Gargan TP, Oster CN, Whitemire RE, Characterization of malaria transmission by Anopheles (Diptera: Culicidae) in western Kenya in preparation for malaria vaccine trials. J Med Entomol. 1990;27:570–7.PubMedGoogle Scholar
- Githeko A, Service M, Mbogo C, Atieli F, Juma F. Plasmodium falciparum sporozoite and entomological inoculation rates at the Ahero rice irrigation scheme and the Miwani sugar-belt in western Kenya. Ann Trop Med Parasitol. 1993;87:379–91.PubMedGoogle Scholar
- McGregor I. The development and maintenance of immunity to malaria in highly endemic areas. Clin Trop Med Commun Dis. 1986;1:29–53.
- Marsh K, Snow R. Malaria transmission and morbidity. Parassitologia. 1999;41:241–6.PubMedGoogle Scholar
- Snow R, Marsh K. The consequences of reducing transmission of Plasmodium falciparum in Africa. Adv Parasitol. 2002;52:235–64. DOIPubMedGoogle Scholar
- Snow R, Omumbo J, Lowe B, Molyneux C, Obiero J, Palmer A, Relation between severe malaria morbidity in children and level of Plasmodium falciparum transmission in Africa. Lancet. 1997;349:1650–4. DOIPubMedGoogle Scholar
- Smith T, Charlwood J, Kihonda J, Mwankusye S, Billingsley P, Meuwissen J, Absence of seasonal variation in malaria parasitaemia in an area of intense seasonal transmission. Acta Trop. 1993;54:55–72. DOIPubMedGoogle Scholar
- Lindsay S, Martens W. Malaria in the African highlands: past, present and future. Bull World Health Organ. 1998;76:33–45.PubMedGoogle Scholar
- Malakooti M, Biomndo K, Shanks G. Reemerging of epidemic malaria in the highlands of western Kenya. Emerg Infect Dis. 1998;4:671–6. DOIPubMedGoogle Scholar
- Hay S, Noor A, Simba M, Busolo M, Guyatt H, Ochola S, Clinical epidemiology of malaria in the highlands of western Kenya. Emerg Infect Dis. 2002;8:543–8.PubMedGoogle Scholar
- Minakawa N, Sonye G, Mogi M, Yan G. Habitat characteristics of Anopheles gambiae s.s. larvae in a Kenyan highland. Med Vet Entomol. 2004;18:301–5. DOIPubMedGoogle Scholar
- Slutsker L, Taylor T, Wirima J, Steketee R. In-hospital morbidity and mortality due to malaria-associated severe anaemia in two areas of Malawi with different patterns of malaria infection. Trans R Soc Trop Med Hyg. 1994;88:548–51. DOIPubMedGoogle Scholar
- Prybylski D, Khaliq A, Fox E, Sarwari A, Strickland G. Parasite density and malaria morbidity in the Pakistani Punjab. Am J Trop Med Hyg. 1999;61:791–801.PubMedGoogle Scholar
- Ripley B. The second-order analysis of stationary point processes. J Appl Probab. 1976;13:255–66. DOIGoogle Scholar
- Getis A, Franklin J. Second-order neighborhood analysis of mapped point patterns. Ecology. 1987;68:473–7. DOIGoogle Scholar
- Haase P. Spatial pattern analysis in ecology based on Ripley's K-function: introduction and methods of edge correction. J Veg Sci. 1995;6:575–82. DOIGoogle Scholar
- Getis A, Ord JK. The analysis of spatial association by use of distance statistics. Geogr Anal. 1992;24:189–206. DOIGoogle Scholar
- Ord J, Getis A. Local spatial autocorrelation statistics: distributional issue and an application. Geogr Anal. 1995;27:286–306. DOIGoogle Scholar
- Zhou G, Minakawa N, Githeko A, Yan G. Spatial distribution patterns of malaria vectors and sample size determination in spatially heterogeneous environments: a case study in the west Kenyan highlands. J Med Entomol. 2004;41:1001–9. DOIPubMedGoogle Scholar
- John C, Koech D, Sumba P, Ouma J. Risk of Plasmodium falciparum infection during a malaria epidemic in highland Kenya, 1997. Acta Trop. 2004;92:55–61. DOIPubMedGoogle Scholar
- Sokhna C, Faye F, Spiegel A, Dieng H, Trape J. Rapid reappearance of Plasmodium falciparum after drug treatment among Senegalese adults exposed to moderate seasonal transmission. Am J Trop Med Hyg. 2001;65:167–70.PubMedGoogle Scholar
- Some ES. Effects and control of highland malaria epidemic in Uasin Gishu District, Kenya. East Afr Med J. 1994;71:2–8.PubMedGoogle Scholar
- Ayisi J, Githeko A, Owaga M, Ekisa W, Anyona D, Obala A, A survey of malaria endemicity in Kericho District, Kenya June 1990. In: Were J, Mutugi M, Githure J, editors. Proceedings of the Annual Medical Scientific Conference: 1992. Nairobi, Kenya.
- Spencer H, Kaseje D, Collins W, Shehata M, Turner A, Stanfill P, Community based malaria control in Saradidi, Kenya: description of the programme and impact on parasitemia rates and antimalarial antibodies. Ann Trop Med Parasitol. 1987;81:13–23.PubMedGoogle Scholar
- Githeko A, Brandling-Bennett A, Beier M, Atieli F, Owaga M, Collins F. The reservoir of Plasmodium falciparum malaria in a holoendemic area of western Kenya. Trans R Soc Trop Med Hyg. 1992;86:355–8. DOIPubMedGoogle Scholar
- Githeko A, Brandling-Bennett A, Beier M, Atieli F, Owaga M, Juma F, Confirmation that Plasmodium falciparum has aperiodic infectivity to Anopheles gambiae. J Med Vet Entomol. 1993;7:373–6. DOIPubMedGoogle Scholar
- Oloo A, Githeko A, Adungo N, Karanja D, Vulule J, Kisia-Abok , Field trial of permethrin impregnated sisal curtains in malaria control in western Kenya. East Afr Med J. 1996;73:735–40.PubMedGoogle Scholar
- Bousema J, Gouagna L, Drakeley C, Meutstege A, Okech B, Akim I, Plasmodium falciparum gametocyte carriage in asymptomatic children in western Kenya. Malar J. 2004;3:18. DOIPubMedGoogle Scholar
- Trape J, Lefebvre-Zante E, Legros F, Ndiaye G, Bouganali H, Druilhe P, Vector density gradients and the epidemiology of urban malaria in Dakar, Senegal. Am J Trop Med Hyg. 1992;47:181–9.PubMedGoogle Scholar
- Staedke S, Nottingham E, Cox J, Kamya M, Rosenthal P, Dorsey G. Short report: proximity to mosquito breeding sites as a risk factor for clinical malaria episodes in an urban cohort of Ugandan children. Am J Trop Med Hyg. 2003;69:244–6.PubMedGoogle Scholar
Figures
Table
Cite This Article1Current affiliation: Saga University, Saga, Japan.
Table of Contents – Volume 11, Number 10—October 2005
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Guiyun Yan, Department of Biological Sciences, 109 Cooke Hall, State University of New York, Buffalo, NY 14260, USA; fax: 716-645-2975
Top