Volume 11, Number 5—May 2005
THEME ISSUE
Dengue and Dengue Hemorrhagic Fever
Dengue and Dengue Hemorrhagic Fever
Leptospirosis during Dengue Outbreak, Bangladesh
Cite This Article
Citation for Media
Abstract
We collected acute-phase serum samples from febrile patients at 2 major hospitals in Dhaka, Bangladesh, during an outbreak of dengue fever in 2001. A total of 18% of dengue-negative patients tested positive for leptospirosis. The case-fatality rate among leptospirosis patients (5%) was higher than among dengue fever patients (1.2%).
Leptospirosis is a zoonotic infection caused by spirochetes of the genus Leptospira. Infection usually results when water or soil contaminated with the urine of an infected animal comes in contact with human skin or mucous membranes (1). Clinical manifestations of leptospirosis can range from a self-limited febrile syndrome to a fatal illness (Weil disease) characterized by hemorrhage, renal failure, and jaundice. In tropical settings, leptospirosis can be indistinguishable from other febrile illnesses such as scrub typhus, malaria, or dengue.
Although leptospirosis has been reported in neighboring areas of Southeast Asia (2,3), the disease is not recognized in Bangladesh, where diagnostic tests for leptospirosis are not available. However, environmental factors, such as floods, humidity, and water contamination, are amenable to spread of the disease in Bangladesh.
An epidemic of dengue fever and dengue hemorrhagic fever began in Bangladesh in 2000 (4), and a surveillance system was established to identify patients with denguelike illness at 2 major hospitals in Dhaka. Approximately three-quarters of patients meeting surveillance criteria had laboratory evidence of dengue infection. We hypothesized that leptospirosis might be a cause of illness among febrile patients who did not have dengue fever. To assess this, we retrospectively analyzed acute-phase serum samples from all dengue-negative patients by using real-time polymerase chain reaction (PCR) for Leptospira. We used data collected as part of the surveillance program to identify distinguishing clinical characteristics of leptospirosis.
In 2000, the International Centre for Diarrhoeal Disease Research, Bangladesh (ICDDR,B) worked with staff from Dhaka Medical College and Holy Family Red Crescent Hospital to initiate surveillance for dengue as part of an emergency response to an epidemic of dengue and dengue hemorrhagic fever. Physicians at Dhaka Medical College and Holy Family Red Crescent Hospital were trained in the clinical diagnosis and management of dengue and dengue hemorrhagic fever according to World Health Organization guidelines. Patients hospitalized with fever and in whom a physician suspected dengue were enrolled in the surveillance program. Clinical and epidemiologic information, as well as acute-phase serum specimens, were systematically collected from surveillance patients. Acute-phase serum specimens were assessed for dengue virus antibodies by using a commercial immunoglobulin (Ig)G and IgM capture enzyme-linked immunosorbent assay (ELISA) (PanBio Dengue Duo, PanBio Ltd., Brisbane, Queensland, Australia) (5). In addition, serum samples collected from a subset of patients during the first 5 days of illness were evaluated for dengue virus RNA by using reverse transcriptase–polymerase chain reaction (RT-PCR), as described previously (6). Serum samples from patients with no dengue infection shown by antibody or RT-PCR testing were retrospectively assessed for leptospirosis by using a real-time PCR that amplifies the LipL32 gene (7), a virulence factor that is conserved among pathogenic Leptospira strains (8). Microplate Leptospira IgM ELISA testing (PanBio Ltd.) was conducted on all Leptospira PCR-positive serum specimens of sufficient quantity (9).
Specimens from 1,297 patients hospitalized at Dhaka Medical College and Holy Family Red Crescent Hospital between January 1 and December 31, 2001, were evaluated for dengue infection by using capture ELISA; 55 acute-phase serum samples were additionally evaluated by using RT-PCR for dengue. A total of 938 (72%) patients were diagnosed with dengue fever by serologic tests (932 patients0, RT-PCR (3 patients), or both (3 patients). Acute-phase serum specimens from the 359 patients without laboratory evidence of dengue were evaluated for leptospirosis; 63 (18%) had Leptospira detected by using PCR. Sixty-one of the PCR-positive samples were tested for Leptospira-specific IgM; 18 (30%) showed positive results and 5 (8%) showed equivocal results.
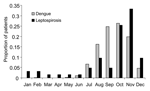
Patients with leptospirosis diagnosed by using PCR were 6 to 70 years of age (mean 28, SD 13); 74% were male. Patients with leptospirosis were of similar age and sex as patients with dengue (Table 1). Patients with leptospirosis had less education and came from households with lower income than patients with dengue. The peak occurrence of leptospirosis was in October and November, shortly after the monsoon season in Bangladesh. This overlapped with the period of highest dengue activity (July through December) (Figure).
Patients with leptospirosis reported a slightly longer duration of fever than patients with dengue (Table 2). While most patients with dengue or leptospirosis had continuous fever, an intermittent fever was more likely with leptospirosis. Reports of rash were more common with dengue fever. Aside from fever and rash, the symptoms of patients with leptospirosis and dengue were similar (Table 2): headache, myalgia, nausea, and vomiting were most common.
The median temperature and heart rate at physical examination were higher in leptospirosis patients than in dengue patients. Evidence of bleeding, including petechial rash, positive tourniquet test result, and gum bleeding, were more common in patients with dengue, although they were also found in some patients with leptospirosis. Subconjunctival hemorrhage, which may have been confused with conjunctival inflammation, was more commonly reported in patients with leptospirosis. Hepatomegaly and jaundice were more common in leptospirosis patients, but this difference was not statistically significant.
On laboratory examination, total leukocyte counts were similar in patients with dengue and patients with leptospirosis; however, lymphocytes were more likely to be predominant in patients with dengue. Hemoconcentration and thrombocytopenia were associated with dengue fever.
Of the patients whose outcome was known, 3 (5%) patients with leptospirosis died, compared with 11 (1.2%) patients with dengue (p = 0.048). Antimicrobial therapy for leptospirosis was not provided, and data on the cause of death were not available. The patients who died of leptospirosis were younger than those who died of dengue (24 ± 6 years of age vs. 36 ± 9 years, p = 0.05).
This is the first description of disease caused by Leptospira in urban Bangladesh. Our findings indicate that leptospirosis causes serious febrile illness in the densely populated city of Dhaka.
Studies conducted in other dengue-endemic areas have shown that leptospirosis can be confused with dengue fever (10–12). Most clinical symptoms of leptospirosis patients in Dhaka were nonspecific and not distinguishable from symptoms associated with dengue fever or other viral illnesses. Although fever in leptospirosis patients was higher and of longer duration than in dengue patients, there is sufficient overlap of clinical findings to suggest that clinicians caring for patients in Bangladesh should maintain a high index of suspicion for both diseases, especially during the peak incidence seasons that follow the monsoons. Recognition of leptospirosis is especially important since antimicrobial agents can reduce its severity and duration (13).
Leptospirosis patients identified in Dhaka were impoverished and poorly educated. This may reflect more frequent exposure to environments contaminated with urine from rodents or other animals. In contrast, dengue patients came from households with higher incomes and levels of education. Whether these socioeconomic differences reflect differing patterns of disease can only be determined by future population-based studies, which may in turn shed light on optimal prevention strategies.
Most of the patients hospitalized with leptospirosis and dengue virus infection in Dhaka during the period of this study were male. A recent seroprevalence study in Bangladesh did not demonstrate a sex difference in dengue seropositivity (14), but leptospirosis has been reported predominantly in men in other regions (15). Further prospective research would be useful to better define the clinical spectrum and gender distribution of disease in Bangladesh.
This study has a number of limitations. Only acute-phase serum samples were obtained as part of the hospital-based dengue surveillance program in Bangladesh. Serologic diagnosis of leptospirosis with a single specimen obtained early in infection is limited; hence, we used Leptospira-specific PCR for diagnosis in our study population. Although this molecular technique is highly sensitive and specific for the presence of leptospiremia (7), more cases may have been detected through the use of microscopic agglutination testing on paired serum samples. Notably, less than one-third of the patients with a diagnosis of leptospirosis by PCR had detectable levels of Leptospira-specific IgM. This is likely due to the sampling of acutely ill patients before seroconversion. In support of this, leptospirosis patients who had detectable levels of IgM exhibited a trend toward longer duration of fever compared with those who did not have detectable levels (6.5 vs. 5.5 days, p = 0.12).
Some cases of leptospirosis resulted in death, and the case-fatality rate among leptospirosis patients was significantly higher than among dengue fever patients. The different case-fatality rates may be related, however, to the intensive training in dengue case management that occurred during this epidemic period, or to the lack of specific antimicrobial therapy for unrecognized cases of leptospirosis. Our findings underscore the need for greater awareness of leptospirosis in the Indian subcontinent, more data on its incidence in Bangladesh, and optimal treatment regimens for leptospirosis that can be applied in resource-poor settings.
Dr. LaRocque is a clinical assistant in infectious diseases at Massachusetts General Hospital and an instructor in medicine at Harvard Medical School. She is involved in collaborative research on cholera with the ICDDR,B.
Acknowledgments
We thank Adam Cohen for assistance with preparing specimens for shipment.
This study was funded by the International Center for Tropical Disease Research of the National Institutes of Health (U01-AI58935 and GR-00100), by a cooperative agreement from the US Agency for International Development (HRN-A-00-96-90005-00), and by core donors to the ICDDR,B: Centre for Health and Population Research. Dr. LaRocque was supported by the Burroughs-Wellcome Fund Postdoctoral Fellowship in Tropical Infectious Diseases of the American Society of Tropical Medicine and Hygiene. The funding sources had no involvement in the study design, interpretation, or decision to submit this paper.
References
- Chaudhry R, Premlatha MM, Mohanty S, Dhawan B, Singh KK, Dey AB. Emerging leptospirosis, north India. Emerg Infect Dis. 2002;8:1526–7.PubMedGoogle Scholar
- Karande S, Bhatt M, Kelkar A, Kulkarni M, De A, Varaiya A. An observational study to detect leptospirosis in Mumbai, India, 2000. Arch Dis Child. 2003;88:1070–5. DOIPubMedGoogle Scholar
- Rahman M, Rahman K, Siddque AK, Shoma S, Kamal AH, Ali KS, First outbreak of dengue hemorrhagic fever, Bangladesh. Emerg Infect Dis. 2002;8:738–40.PubMedGoogle Scholar
- Vaughn DW, Nisalak A, Solomon T, Kalayanarooj S, Nguyen MD, Kneen R, Rapid serologic diagnosis of dengue virus infection using a commercial capture ELISA that distinguishes primary and secondary infections. Am J Trop Med Hyg. 1999;60:693–8.PubMedGoogle Scholar
- Lanciotti RS, Calisher CH, Gubler DJ, Chang GJ, Vorndam AV. Rapid detection and typing of dengue viruses from clinical samples by using reverse transcriptase-polymerase chain reaction. J Clin Microbiol. 1992;30:545–51.PubMedGoogle Scholar
- Levett PN, Morey RE, Galloway RL, Turner DE, Steigerwalt AG, Mayer LW. Detection of pathogenic leptospires by real-time quantitative PCR. J Med Microbiol. 2005;54:45–9. DOIPubMedGoogle Scholar
- Haake DA, Chao G, Zuerner RL, Barnett JK, Barnett D, Mazel M, The leptospiral major outer membrane protein LipL32 is a lipoprotein expressed during mammalian infection. Infect Immun. 2000;68:2276–85. DOIPubMedGoogle Scholar
- Bajani MD, Ashford DA, Bragg SL, Woods CW, Aye T, Spiegel RA, Evaluation of four commercially available rapid serologic tests for diagnosis of leptospirosis. J Clin Microbiol. 2003;41:803–9. DOIPubMedGoogle Scholar
- Levett PN, Branch SL, Edwards CN. Detection of dengue infection in patients investigated for leptospirosis in Barbados. Am J Trop Med Hyg. 2000;62:112–4.PubMedGoogle Scholar
- Sanders EJ, Rigau-Perez JG, Smits HL, Deseda CC, Vorndam VA, Aye T, Increase of leptospirosis in dengue-negative patients after a hurricane in Puerto Rico in 1996 [correction of 1966]. Am J Trop Med Hyg. 1999;61:399–404.PubMedGoogle Scholar
- Flannery B, Pereira MM, Velloso L F, Carvalho C C, de Codes LG, Orrico G S, Referral pattern of leptospirosis cases during a large urban epidemic of dengue. Am J Trop Med Hyg. 2001;65:657–63.PubMedGoogle Scholar
- Suputtamongkol Y, Niwattayakul K, Suttinont C, Losuwanaluk K, Limpaiboon R, Chierakul W, An open, randomized, controlled trial of penicillin, doxycycline, and cefotaxime for patients with severe leptospirosis. Clin Infect Dis. 2004;39:1417–24. DOIPubMedGoogle Scholar
- Hossain MA, Khatun M, Arjumand F, Nisaluk A, Breiman RF. Serologic evidence of dengue infection before onset of epidemic, Bangladesh. Emerg Infect Dis. 2003;9:1411–4.PubMedGoogle Scholar
- Laras K, Cao BV, Bounlu K, Nguyen TK, Olson JG, Thongchanh S, The importance of leptospirosis in southeast Asia. Am J Trop Med Hyg. 2002;67:278–86.PubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 11, Number 5—May 2005
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Regina C. LaRocque, Division of Infectious Diseases, Gray-Jackson 504, Massachusetts General Hospital, 55 Fruit St, Boston, MA 02114, USA; fax: 617-726-7416
Top