Volume 12, Number 10—October 2006
Dispatch
ICD-9 Codes and Surveillance for Clostridium difficile–associated Disease
Cite This Article
Citation for Media
Abstract
We conducted a retrospective cohort study to compare Clostridium difficile–associated disease rates determined by C. difficile–toxin assays and International Classification of Diseases, 9th Revision (ICD-9) codes. The correlation between toxin assay results and ICD-9 codes was good (κ = 0.72, p<0.01). The sensitivity of the ICD-9 codes was 78% and the specificity was 99.7%.
Clostridium difficile–associated disease (CDAD) is the most common infectious cause of healthcare-associated diarrhea (1). Recent studies suggest both the incidence and severity of CDAD may be increasing (2–9), but no national surveillance system exists to track CDAD rates. Some studies have used International Classification of Diseases, 9th Revision (ICD-9) codes of hospital discharges to study CDAD rates (4,10,11). The validity of this method has not been studied. We compared CDAD rates determined by ICD-9 codes to rates determined by C. difficile–toxin assays at a tertiary-care hospital to determine the sensitivity and specificity of ICD-9 code–based CDAD surveillance.
Data were collected electronically on a retrospective cohort of patients admitted to Barnes-Jewish Hospital in Saint Louis from January 1, 2003, through December 31, 2003. Patients who had only 1 admission of <48 hours and neonates were excluded. Electronic charts were reviewed for patients who had a positive C. difficile–toxin assay or the ICD-9 code indicating C. difficile–associated disease (008.45) (Appendix) .
A case of CDAD was defined as a patient with a positive C. difficile–toxin assay (Tech Laboratory C. difficile tox A/B II toxin assay [Tech Laboratory, Blacksburg, VA, USA]) or pseudomembranes seen on colonoscopy. Because the hospital laboratory performs a C. difficile–toxin assay only on unformed stool samples and stool toxin testing is ordered based on clinical suspicion of CDAD, all patients with a positive toxin assay were considered CDAD case-patients.
Data were analyzed with SPSS 12.0 for Windows (SPSS, Inc., Chicago, IL, USA). Statistical analyses included κ, χ2, and Mann-Whitney U tests. A 2-sided p value of 0.05 was considered significant. This study was approved by the Washington University Human Studies Committee.
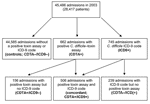
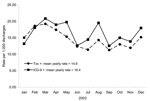
A total of 45,486 admissions among 28,417 unique patients were included in the analysis (Figure 1). A C. difficile–toxin assay was ordered during hospitalization for 3,630 (8%) of these admissions. Toxin assays were positive (CDTA+) in 662 (18%) admissions. The C. difficile ICD-9 code was assigned to 745 admissions (ICD9+). The breakdown of admissions, according to toxin assay and ICD-9 status, was as follows: 506 had both a positive toxin assay and received the ICD-9 code (concordant; CDTA+/ICD9+), 156 had a positive toxin assay but no ICD-9 code (CDTA+/ICD9–), and 239 received the ICD-9 code but did not have a positive toxin assay (CDTA–/ICD9+) (Figure 1). The concordance between toxin assays and ICD-9 codes was good (κ = 0.72, p<0.01). The overall mean CDAD rate by ICD-9 codes (16.4/1,000 admissions) was significantly higher then the mean rate by toxin assays (14.6/1,000 admissions) (Figure 2; rate ratio [RR] 1.13, 95% confidence interval [CI] 1.01–1.25).
The median number of days from admission to stool collection was greater in admissions with a positive toxin assay but no ICD-9 code (CDTA+/ICD9–) than in concordant (CDTA+/ICD9+) admissions (6.0 days vs 3.0 days, p<0.01) (Table 1). The first positive stool sample was collected within 48 hours of discharge for 68 (44%) of admissions with a positive toxin assay only (CDTA+/ICD9–) admissions, compared with 72 (14%) of concordant (CDTA+/ICD9+) admissions (p<0.01).
Upon chart review, documentation of a previous history of CDAD was evident in 142 (59%) of the ICD-9 code only (CDTA–/ICD9+) admissions. A C. difficile–toxin assay had been ordered in 137 (57%) of all ICD-9 code only (CDTA-/ICD9+) admissions. One-hundred thirty (54%) had at least 1 stool negative for C. difficile toxin.
Overall, 92% of patients with positive toxin assay (CDTA+) and 90% of patients with ICD-9 code only (CDTA–/ICD9+) received antimicrobial drug treatment for CDAD (Table 2). Metronidazole was prescribed in 187 (78%) of the ICD-9 code only (CDTA–/ICD9+) admissions, compared with 591 (89%) of patients with a positive toxin assay (CDTA+) (p<0.01). For 75 (31%) of the ICD-9 code only (CDTA–/ICD9+) admissions, oral vancomycin was prescribed, compared with 130 (20%) of patients with a positive toxin assay (CDTA+) (p<0.01).
Thirty-four cases of CDAD were missed by C. difficile–toxin assay results and subsequently identified through chart review (3 missed positive toxins, 26 CDAD patients transferred from other facilities, 3 positive outpatient toxin assays, and 2 diagnosed by colonoscopy), which brought the total number of cases with positive CDAD diagnostics to 696. ICD-9 codes correctly identified 540 of these cases and correctly classified 44,741 admissions as non-CDAD admissions (sensitivity 78%, specificity 99.7%). When the CDAD rate by toxin assays was adjusted for the additional cases, the adjusted CDAD rate was 15.3/1,000 admissions. This rate was not significantly different from the unadjusted CDAD rate by toxin assay results (RR 0.95, 95% CI 0.86–1.06) or the rate by ICD-9 codes alone (RR 1.07, 95% CI 0.97–1.19).
Overall, there was good correlation between C. difficile–toxin assay results and ICD-9 codes. Initially, the CDAD rate by ICD-9 codes appeared higher than the rate by toxin assays. However, once the additional CDAD cases identified through chart review were added, this difference was not significant.
Admissions with only a positive C. difficile–toxin assay and no C. difficile ICD-9 code (CDTA+/ICD9–) were more likely than concordant (CDTA+/ICD9+) admissions to have their first positive toxin assay within 48 hours of discharge. For these admissions, toxin assay results may not yet have been back at the time of discharge or CDAD may not have been considered a primary diagnosis by the physician and therefore not captured by the medical coders.
Antimicrobial drug treatment patterns suggest ICD-9 code only (CDTA–/ICD9+) admissions were patients who were more likely to have a history of CDAD. Metronidazole is first-line therapy for CDAD at our institution. Oral vancomycin is reserved for recurrent or severe cases. The observation that more ICD-9 code only (CDTA–/ICD9+) patients received oral vancomycin indicates that recurrent CDAD may have been suspected in these patients. In these patients, CDAD appears to have been diagnosed on the basis of the patient's history and symptoms instead of by a positive C. difficile–toxin assay. This pattern has been previously reported (12).
True CDAD cases may have been misclassified among the controls. A patient who did not have a positive C. difficile–toxin assay, who was not assigned the CDAD ICD-9 code, and whose diagnosis was made by colonoscopy would have been missed. However, misclassification is unlikely for two reasons. First, after charts were reviewed, only 2 additional patients were identified whose diagnosis was made by colonoscopy alone. Second, the detection of CDAD cases transferred from other institutions indicates that CDAD cases diagnosed by methods other than the toxin assays are being captured by ICD-9 codes.
Use of ICD-9 codes to study CDAD rates has advantages and disadvantages. ICD-9 codes are readily available from billing databases. In the absence of a national surveillance system for CDAD, ICD-9 codes provide a standard method for determining CDAD rates that can be used at all types of hospitals. However, because ICD-9 codes are assigned at discharge and not on the date of diagnosis, determining which cases are hospital acquired and which are community acquired is difficult. Also, ICD-9 codes are assigned by medical coders, who may not be able to accurately identify a patient's principal diagnoses as well as a physician or medical professional. Despite these limitations, ICD-9 codes can likely be used to identify CDAD cases and track CDAD rates when C. difficile–toxin assay results are not available.
Details on ICD-9 Codes
The International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9) code used in this study was 008.45, "intestinal infection due to Clostridium difficile," and is the only ICD-9 code related to CDAD. To apply this code, medical coders must have documentation in a patient's medical record by the treating medical providers that a patient's gastroenteritis or colitis is due to C. difficile. Positive laboratory tests alone are not sufficient to warrant application of the code. At our institution, ICD-9 coding occurs, on average, 5–7 days after a patient is discharged from the hospital.
The ICD-9 system of classifying hospital discharge diagnoses is used throughout the United States. The definition for the code 008.45 is consistent between hospitals, although individual coding practices may vary. Although ICD-9 codes have limitations, they are readily available from administrative databases and have been used frequently to identify diagnoses and classify comorbidities (1).
A move to the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10) system is anticipated for US hospitals but the exact time of this transition is not yet known. The ICD-10 system does include a code for CDAD (A04.7, Enterocolitis due to C. difficile), so the ICD-based system presented here could be modified to be used with the updated coding system.
Appendix Reference
Klabunde CN, Warren JL, Legler JM. Assessing comorbidity using claims data: an overview. Med Care. 2002;40(8 Suppl):IV-25–35.
Dr Dubberke is a clinical instructor of medicine in the Division of Infectious Diseases, Department of Medicine, at Washington University School of Medicine. His research interests include C. difficile–associated disease, hospital epidemiology, and infections in transplant recipients.
Acknowledgments
We thank Margaret Olsen for statistical advice and Cherie Hill for technical assistance.
This work was supported by grants from the Centers for Disease Control and Prevention (UR8/CCU715087-06/1, 1U01C1000333-01) and the National Institutes of Health (1K24AI06779401). Preliminary data were presented in part at the 15th Annual Scientific Meeting of the Society for Healthcare Epidemiology of America, Los Angeles, California, USA, April 9–12, 2005.
References
- Johnson S, Gerding DN. Clostridium difficile–associated diarrhea. Clin Infect Dis. 1998;26:1027–34. DOIPubMedGoogle Scholar
- Archibald LK, Banerjee SN, Jarvis WR. Secular trends in hospital-acquired Clostridium difficile disease in the United States, 1987–2001. J Infect Dis. 2004;189:1585–9. DOIPubMedGoogle Scholar
- Dallal RM, Harbrecht BG, Boujoukas AJ, Sirio CA, Farkas LM, Lee KK, Fulminant Clostridium difficile: an underappreciated and increasing cause of death and complications. Ann Surg. 2002;235:363–72. DOIPubMedGoogle Scholar
- McDonald LC, Owings M, Jernigan DB. Clostridium difficile infection in patients discharged from US short-stay hospitals, 1996–2003. Emerg Infect Dis. 2006;12:409–15.PubMedGoogle Scholar
- Morris AM, Jobe BA, Stoney M, Sheppard BC, Deveney CW, Deveney KE. Clostridium difficile colitis: an increasingly aggressive iatrogenic disease? Arch Surg. 2002;137:1096–100. DOIPubMedGoogle Scholar
- Pepin J, Valiquette L, Alary ME, Villemure P, Pelletier A, Forget K, Clostridium difficile-associated diarrhea in a region of Quebec from 1991 to 2003: a changing pattern of disease severity. CMAJ. 2004;171:466–72. DOIPubMedGoogle Scholar
- Pepin J, Alary ME, Valiquette L, Raiche E, Ruel J, Fulop K, Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40:1591–7. DOIPubMedGoogle Scholar
- Rubin MS, Bodenstein LE, Kent KC. Severe Clostridium difficile colitis. Dis Colon Rectum. 1995;38:350–4. DOIPubMedGoogle Scholar
- Siemann M, Koch-Dorfler M, Rabenhorst G. Clostridium difficile–associated diseases. The clinical courses of 18 fatal cases. Intensive Care Med. 2000;26:416–21. DOIPubMedGoogle Scholar
- Frost F, Craun GF, Calderon RL. Increasing hospitalization and death possibly due to Clostridium difficile diarrheal disease. Emerg Infect Dis. 1998;4:619–25. DOIPubMedGoogle Scholar
- Peterson CA, Calderon RL. Trends in enteric disease as a cause of death in the United States, 1989–1996. Am J Epidemiol. 2003;157:58–65. DOIPubMedGoogle Scholar
- McFarland LV, Surawicz CM, Rubin MS, Fekety R, Elmer GW, Grover H, Recurrent Clostridium difficile disease: epidemiology and clinical characteristics. Infect Control Hosp Epidemiol. 1999;20:43–9. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 12, Number 10—October 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Erik R. Dubberke, Campus Box 8051 660 S Euclid, Saint Louis, MO 63110, USA
Top