Volume 12, Number 2—February 2006
Dispatch
Sequencing and Staphylococci Identification
Cite This Article
Citation for Media
Abstract
The emerging clinical importance of staphylococcal infections prompted us to establish a reference database for partial RNA polymerase B (rpoB; nucleotides 1444–1928) gene sequences from type strains of all staphylococcal species and subspecies. This database correctly identified 55 clinical staphylococcal isolates; all were correctly identified at the species level. At the subspecies level, rpoB misidentified only 2 isolates.
The emerging clinical importance of Staphylococcus aureus and coagulase-negative staphylococci (1) in connection with the expanding number of staphylococcal subspecies described requires accurate identification to the subspecies level. Currently, the genus Staphylococcus is divided into 36 species and 21 subspecies. Staphylococcal subspecies not included in the databases of commercial identification systems, as well as phenotypic variants (e.g., small-colony variants), are often misidentified (2).
We recently described the usefulness of genotypic identification of staphylococcal subspecies by using partial 16S rDNA sequences in comparison with phenotypic tests (3). However, the partial 16S rDNA sequences used were not discriminative enough to differentiate all staphylococcal subspecies. When searching for a molecular target for discrimination of staphylococci, several genes have been evaluated, e.g., heat shock protein 60 (hsp60) (4), superoxide dismutase A (sodA) (5), and RNA polymerase B (rpoB) (6). However, these studies concentrated only on a limited number of staphylococcal species. Therefore, a complete reference database of partial rpoB gene sequences from type strains (n = 47) and other culture collection strains, including all validly described staphylococcal subspecies, was created for this study. This reference database was then evaluated with clinical isolates. Results were compared with those previously obtained by 16S rDNA sequencing and conventional phenotypic tests.
We analyzed 82 type and other culture collection strains encompassing all validly described staphylococcal species (n = 38) and subspecies (n = 21; according to the current List of Bacterial Names with Standing in Nomenclature, updated May 14, 2005) (7). Two strains of the recently proposed candidate species S. pettenkoferi (8) were added to complete the rpoB sequence reference database. Using this database, we analyzed 55 clinical staphylococcal isolates collected from human (n = 52) and animal (S. intermedius, n = 2; S. felis, n = 1) specimens; 6 of the human isolates exhibited the small-colony variant (SCV) phenotype.
This strain collection was previously analyzed by the API ID 32 Staph and VITEK 2 systems (both obtained from bioMérieux, Marcy l'Etoile, France), partial 16S rDNA sequencing, chemotaxonomy, and riboprinting to determine species designation (3). The thermal cycling condition to amplify the partial rpoB gene (899 bp) was 35 cycles of denaturation at 94°C for 45 s (300 s for the first cycle), annealing (60 s at 52°C), and extension (90 s at 72°C, 600 s for the last cycle). The Staphylococcus-specific primers used for amplification and sequencing of rpoB are shown in Table 1. Sequencing reactions were performed in a total volume of 10 μL containing 0.5 μL premix (ABI Prism BigDye Terminator v3.0 Ready Reaction Cycle Sequencing Kit, Applied Biosystems, Darmstadt, Germany), 1.8 μL 400 mmol/L Tris-HCl, 10 mmol/L MgCl2, 10 pmol sequencing primer, and 2 μL polymerase chain reaction product. The sequencing products were purified by using the Centri-Sep Spin Columns (Princeton Separations, Adelphia, NJ, USA) and analyzed with the ABI Prism 3100 Avant Genetic Analyzer (Applied Biosystems) according to the manufacturer's instructions. For further analysis, nucleotides 1444–1928 (corresponding to S. aureus rpoB gene positions of the GenBank accession no. X64172) of the rpoB gene were used. The sequences were analyzed by using Ridom TraceEditPro version 1.0 software (Ridom GmbH, Würzburg, Germany). Staphylococcal partial rpoB reference sequences determined in this study were deposited in GenBank under accession nos. DQ120729–DQ120752.
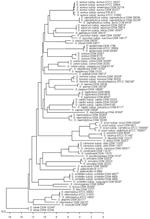
Partial rpoB sequences were determined for 82 culture collection strains and 55 clinical isolates. All staphylococcal type strains were distinguishable by rpoB; the only exception was the S. equorum subspecies that shared the same sequence (Figure A1). The mean pairwise distance of all type and other culture collection strains exhibiting a unique rpoB sequence (n = 68) was 13.7% (range 0%–21.4%) and the standard deviation was 3.3%. When assuming a normal distribution for the distances and choosing a reporting criterion >94.0%, the similarity for a distinct species correlates with a statistical error probability of 1.0% (9).
The definitive identification of 55 clinical isolates and the rpoB gene sequence similarity search results are shown in Table 2. At the species level, the correct species designation for all 55 clinical isolates was made by rpoB sequence similarity search (sequence similarity >94.0%). Of 21 clinical isolates belonging to species currently divided into subspecies, 17 isolates were correctly identified to the subspecies level. Subspecies identification for isolates M26 and M53 was unsuccessful by rpoB or partial 16S rDNA sequencing, riboprinting, and chemotaxonomy (data not shown). Only isolates M20 and M39 were misidentified by rpoB sequencing as S. saprophyticus subsp. saprophyticus instead of subsp. bovis.
Our previous study demonstrated the superiority of sequence-based methods over phenotypic approaches using the API ID 32 Staph and VITEK 2 systems (3). The advantage of a sequence-based method became most evident when differentiating isolates with the SCV phenotype, in which the API ID 32 Staph and VITEK 2 systems misidentified 2 and 4 isolates, respectively. When both sequence-based approaches used were compared, rpoB sequencing was superior to partial 16S rDNA identification. Although the 16S rDNA procedure differentiated 50 (90.9%) of all tested clinical isolates at species level, rpoB identified 100%. Therefore, if an unknown organism needs to be identified, 16S rDNA sequencing is the method of choice because of the availability of universal primers (10). However, if the genus is already known, the rpoB method should be used.
Compared with other published molecular probes, rpoB showed the highest discriminatory power, e.g., hsp60 and sodA sequencing did not differentiate subspecies of S. carnosus, S. cohnii, S. hominis, S. schleiferi, or S. succinus (4,5). In a previous study, rpoB sequence-based identification of Staphylococcus species has been reported (6). However, a limited number of taxa were included, and the primers used were not appropriate to detect all staphylococcal subspecies.
Sequencing of rpoB was also used to identify other bacterial species (11,12). A higher discrimination with rpoB sequencing compared with 16S rDNA sequencing has been demonstrated for the genera Corynebacterium (13) and Bacillus (14). DNA sequencing is a rapid alternative to biochemical and other phenotypic procedures for the differentiation of bacterial pathogens because of its decreased costs and increased automation (15). Thus, rpoB is a useful molecular target for differentiating staphylococcal isolates to the species and subspecies level.
Dr Mellmann is a consultant for medical microbiology, hygiene, and infectious diseases at the University Hospital Münster. His professional interests include molecular identification and epidemiology of bacterial pathogens.
References
- von Eiff C, Peters G, Heilmann C. Pathogenesis of infections due to coagulase-negative staphylococci. Lancet Infect Dis. 2002;2:677–85. DOIPubMedGoogle Scholar
- Seifert H, Wisplinghoff H, Schnabel P, von Eiff C. Small colony variants of Staphylococcus aureus and pacemaker-related infection. Emerg Infect Dis. 2003;9:1316–8.PubMedGoogle Scholar
- Becker K, Harmsen D, Mellmann A, Meier C, Schumann P, Peters G, Development and evaluation of a quality-controlled ribosomal sequence database for 16S ribosomal DNA-based identification of Staphylococcus species. J Clin Microbiol. 2004;42:4988–95. DOIPubMedGoogle Scholar
- Kwok AY, Su SC, Reynolds RP, Bay SJ, Av-Gay Y, Dovichi NJ, Species identification and phylogenetic relationships based on partial HSP60 gene sequences within the genus Staphylococcus. Int J Syst Bacteriol. 1999;49:1181–92. DOIPubMedGoogle Scholar
- Poyart C, Quesne G, Boumaila C, Trieu-Cuot P. Rapid and accurate species-level identification of coagulase-negative staphylococci by using the sodA gene as a target. J Clin Microbiol. 2001;39:4296–301. DOIPubMedGoogle Scholar
- Drancourt M, Raoult D. rpoB gene sequence-based identification of Staphylococcus species. J Clin Microbiol. 2002;40:1333–8. DOIPubMedGoogle Scholar
- Euzeby JP. List of bacterial names with standing in nomenclature: a folder available on the internet. Int J Syst Bacteriol. 1997;47:590–2. DOIPubMedGoogle Scholar
- Trülzsch K, Rinder H, Trcek J, Bader L, Wilhelm U, Heesemann J. 'Staphylococcus pettenkoferi' a novel staphylococcal species isolated from clinical specimens. Diagn Microbiol Infect Dis. 2002;43:175–82. DOIPubMedGoogle Scholar
- Harmsen D, Karch H. 16S rDNA for diagnosing pathogens: a living tree. ASM News. 2004;70:19–24.
- Clarridge JE. Impact of 16S rRNA gene sequence analysis for identification of bacteria on clinical microbiology and infectious diseases. Clin Microbiol Rev. 2004;17:840–62. DOIPubMedGoogle Scholar
- Drancourt M, Roux V, Fournier P, Raoult D. rpoB gene sequence-based identification of aerobic gram-positive cocci of the genera Streptococcus, Enterococcus, Gemella, Abiotrophia, and Granulicatella. J Clin Microbiol. 2004;42:497–504. DOIPubMedGoogle Scholar
- Mollet C, Drancourt M, Raoult D. rpoB sequence analysis as a novel basis for bacterial identification. Mol Microbiol. 1997;26:1005–11. DOIPubMedGoogle Scholar
- Khamis A, Raoult D, La Scola B. Comparison between rpoB and 16S rRNA gene sequencing for molecular identification of 168 clinical isolates of Corynebacterium. J Clin Microbiol. 2005;43:1934–6. DOIPubMedGoogle Scholar
- Blackwood KS, Turenne CY, Harmsen D, Kabani AM. Reassessment of sequence-based targets for identification of Bacillus species. J Clin Microbiol. 2004;42:1626–30. DOIPubMedGoogle Scholar
- Cook VJ, Turenne CY, Wolfe J, Pauls R, Kabani A. Conventional methods versus 16S ribosomal DNA sequencing for identification of nontuberculous mycobacteria: cost analysis. J Clin Microbiol. 2003;41:1010–5. DOIPubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 12, Number 2—February 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Alexander Mellmann, Institut für Hygiene, Universitätsklinikum Münster, Robert-Koch-Strasse 41, D-48149 Münster, Germany; fax: 49-251-83-55688
Top