Volume 13, Number 7—July 2007
Dispatch
Human Influenza A (H5N1) Cases, Urban Areas of People’s Republic of China, 2005–2006
Cite This Article
Citation for Media
Abstract
We investigated potential sources of infection for 6 confirmed influenza A (H5N1) patients who resided in urban areas of People’s Republic of China. None had known exposure to sick poultry or poultry that died from illness, but all had visited wet poultry markets before illness.
Although >280 confirmed human cases of avian influenza A (H5N1) virus infection from 12 countries have been reported (1), detailed data on sources of infection for most patients are limited (2). In Vietnam, 8 of 9 patients with influenza A (H5N1) reported close contact with sick or dead poultry (3). In Thailand, 9 of 12 such patients lived in households where backyard chickens died, and 8 reported direct contact with dead chickens (4). Case-control studies in Thailand and Vietnam found that the most statistically significant risk factor was recent exposure to sick or dead poultry, especially directly touching dead poultry (5,6).
Avian influenza (H5N1) poultry outbreaks have been reported in mainland People’s Republic of China since 2004 (7); since late 2005, human cases have also been reported (8). Most Chinese patients had exposure to backyard poultry, although some had no apparent direct exposure to poultry that were sick or died. We describe findings of investigations of urban patients with influenza A (H5N1), who had no known direct contact with sick poultry or poultry that died of illness in China.
Enhanced surveillance for influenza-like illness and pneumonia of unknown origin was established in China after the outbreak of severe acute respiratory syndrome (SARS) (9). All suspected cases of influenza A (H5N1) are reported through a national surveillance system to the Chinese Center for Disease Control and Prevention (China CDC). Laboratory testing is performed by the National Influenza Center of China CDC. A confirmed case of influenza (H5N1) was defined according to World Health Organization case definitions (10). This study was part of an ongoing public health outbreak investigation and determined by the ministry of health to be exempt from institutional review board assessment.
China CDC conducted epidemiologic investigations by interviewing confirmed influenza (H5N1) patients and their relatives, reviewing medical records, and visiting patient households and places visited by patients within 2 weeks of illness onset. A standardized questionnaire was used to collect demographic, clinical, and exposure history data. For 3 severely ill patients, only relatives and contacts were interviewed to assess possible influenza (H5N1) subtype exposures. Particular attention was paid to potential exposures such as contact with well-appearing, sick, or dead poultry; visits to poultry markets; or contact with persons with febrile respiratory symptoms in the 2 weeks before onset. A rural case was 1 that occurred in a village resident; an urban case was 1 that occurred in a city resident.
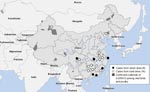
From October 2005 through October 2006, 20 confirmed cases were reported from 12 provinces. Six cases were identified in 6 cities of 5 provinces; each city had an average population of 8.3 million and was at least 112 km away from cities with another case (Figure 1). More urban cases were reported in 2006 (5 [42%] of 12) than in 2005 (1 [13%] of 8), but this difference was not statistically significant (p = 0.325) (Figure 2). Demographic and clinical characteristics of the 6 urban patients with influenza (H5N1) cases are presented in Table 1. All 6 urban patients were adults, median age 30 years (range 21 to 41); 5 died.
Five of the 6 urban case-patients had no direct contact with poultry. One patient prepared freshly slaughtered chicken that she purchased for cooking at a live (wet) poultry market. No patients kept poultry or other animals at home, and no poultry or poultry outbreaks were identified in their neighborhoods. Five patients had visited wet poultry markets within a week of illness onset, and all had visited a wet market during the 2 weeks before their illness. Three patients visited wet markets at least once a day before illness onset. Only 1 patient (case-patient 5) had any travel history in the 2 weeks before illness onset. That patient had visited his parents’ home in a rural area, where healthy backyard poultry were kept outside the house, and he had visited a wet market in the same area 2 weeks before illness onset (Table 2).
All 6 patients had no known contact with other confirmed influenza A (H5N1) patients or with anyone with febrile respiratory symptoms. A total of 640 persons were followed up for medical observation for 2 weeks, including 136 close contacts of the 6 patients, 389 healthcare workers who provided care for them, and 115 persons who worked in the poultry markets visited by the patients. Febrile respiratory illness developed in 5 contacts: case-patient 1’s mother, case-patient 3’s girlfriend, case-patient 6’s daughter, a nurse who cared for case-patient 6, and a patient hospitalized on the same ward as case-patient 6. All 5 ill contacts recovered, and all respiratory specimens collected from them tested negative for influenza A (H5N1) by reverse-transcriptase–PCR. Paired acute- and convalescent-phase serum samples collected from these 5 ill contacts tested negative for subtype (H5N1) neutralizing antibodies by microneutralization assay.
Our study suggests that exposure to wet poultry markets may be an important influenza A (H5N1) risk factor for persons in urban areas of China. None of the 6 case-patients had known direct contact with poultry that were sick or died of illness. Two patients (case-patients 1 and 3) had no identified potential exposures except for visiting a wet poultry market during the week before illness onset. Four other case-patients visited wet markets, although other exposures could have potentially led to virus transmission. Case-patient 2 was an egg seller and could have also been infected by contact with fecally contaminated eggs. In 2005, influenza A (H5N1) virus was isolated from eggs brought to China by travelers from Vietnam (11). Case-patient 4 could potentially have been exposed to the virus through preparation of freshly slaughtered chickens purchased at a wet market. Case-patient 5 could have been exposed to the virus by visiting his parents’ home, which had healthy backyard poultry outside, or by transporting eggs. Case-patient 6 could have been exposed to the virus at home when his wife prepared a freshly slaughtered chicken purchased from a wet market. No epidemiologic evidence suggested human-to-human transmission of influenza A (H5N1) associated with the urban patients.
These observations are consistent with results of a case-control study conducted during the 1997 influenza A (H5N1) outbreak in Hong Kong Special Administrative Region, which found the most statistically significant influenza (H5N1) risk factor was visiting a live poultry market the week before illness onset (12). During that outbreak, widespread subtype (H5N1) poultry dieoffs occurred in wet markets, but these dieoffs have not been observed in urban China. The role of poultry vaccination in decreasing poultry outbreaks in wet markets in China is unknown. A recent simulation study showed that silent spread of the virus can occur in poultry because of incomplete protection at the flock level, even if a poultry vaccine is effective in individual birds (13).
In China, wet markets are sustained by demand for freshly slaughtered poultry. Wet markets are considered a reservoir and amplifier of avian influenza A viruses because they bring together avian host species in a high-density setting that can facilitate viral persistence, cross-species infection, and genetic reassortment (14). Our findings suggest that wet markets pose a risk that is likely to be low for avian-to-human transmission of subtype (H5N1) in urban settings. Viral RNA for subtype (H5N1) was detected in a specimen collected from a goose cage at an urban wet market visited by case-patient 3 (15), which suggests the potential for influenza (H5N1) transmission through environmental contamination.
We were limited by inability to elicit complete exposure histories from all case-patients because of their severe illness or death. However, we interviewed household and family members, friends, and co-workers, and investigated places that patients had visited in the 2 weeks before illness onset. We were unable to ascertain how human infection with the virus occurred through the patients’ visits to wet poultry markets. Possibilities include self-inoculation of the respiratory tract after touching subtype (H5N1)–contaminated surfaces, or inhalation of aerosolized debris with influenza (H5N1) virus.
We did not perform testing on poultry or environmental specimens and can only speculate about potential exposures and sources of influenza A subtype (H5N1) infection in wet markets, homes, and neighborhoods. Future studies should test tracheal, cloacal, and blood specimens from poultry; swabs of fecal material, cages, and other potentially contaminated surfaces; and air specimens in wet markets, for evidence of influenza (H5N1). More research studies, including case-control studies, are needed to better clarify the risk for subtype (H5N1) transmission that occurs from visiting wet poultry markets. Close collaborations are needed between animal health and public health agencies to reduce the public health risk for this virus in wet poultry markets and to understand the impact of poultry vaccination on the risk for transmission.
Dr Yu is a medical epidemiologist in the Office for Disease Control and Emergency Response, China CDC. His research interests include surveillance, prevention, and control of emerging infections, including SARS, avian influenza, and Streptococcus suis.
Acknowledgments
We thank the CDCs of Chengdu, Sanming, Shenzhen, Guangzhou, and Wuhan Prefectures for assisting in coordinating field investigations and providing logistic support.
The Ministry of Health of China has generously facilitated this study. This study was supported by grants from the National Task Force of China (2004BA519A71).
References
- World Health Organization. Cumulative number of confirmed human cases of avian influenza A/(H5N1) reported to WHO. 2007 March 29. [cited 2007 March 29]. Available from http://www.who.int/csr/disease/avian_influenza/country/cases_table_2007_03_29/en/print.html
- World Health Organization. Epidemiology of WHO-confirmed human cases of avian influenza A (H5N1) infection. Wkly Epidemiol Rec. 2006;81:249–57.PubMedGoogle Scholar
- Tran TH, Nguyen TL, Nguyen TD, Luong TS, Pham PM, Nguyen VC, Avian influenza A (H5N1) in 10 patients in Vietnam. N Engl J Med. 2004;350:1179–88. DOIPubMedGoogle Scholar
- Chotpitayasunondh T, Ungchusak K, Hanshaoworakul W, Chunsuthiwat S, Sawanpanyalert P, Kijphati R, Human disease from influenza A (H5N1), Thailand, 2004. Emerg Infect Dis. 2005;11:201–9.PubMedGoogle Scholar
- Areechokchai D, Jiraphongsa C, Laosiritaworn Y, Hanshaoworakul W, O’Reilly M. Investigation of avian influenza (H5N1) outbreak in humans—Thailand, 2004. MMWR Morb Mortal Wkly Rep. 2006;55(Suppl 1):3–6.PubMedGoogle Scholar
- Dinh PN, Long HT, Tien NT, Hien NT, Mai le TQ, Phong le H, et al. Risk factors for human infection with avian influenza A H5N1, Vietnam, 2004. Emerg Infect Dis. 2006;12:1841–7.PubMedGoogle Scholar
- World Organization for Animal Health. Update on avian influenza in animals (type H5). [cited 2006 July 10]. Available from http://www.oie.int/downld/AVIAN%20INFLUENZA/A_AI-Asia.htm
- Yu H, Shu Y, Hu S, Zhang H, Gao Z, Chen H, The first confirmed human case of avian influenza A (H5N1) in Mainland China. Lancet. 2006;367:84. DOIPubMedGoogle Scholar
- World Health Organization. Prevention and control of avian influenza in humans in China: achieving the national objectives of the WHO Global Influenza Preparedness Plan. Wkly Epidemiol Rec. 2006;81:108–13.PubMedGoogle Scholar
- World Health Organization. WHO case definitions for human infections with influenza A (H5N1) virus. 2006 Aug 29. [cited 2006 Sep 9]. Available from http://www.who.int/csr/disease/avian_influenza/guidelines/case_definition2006_08_29/en/index.html
- Li Y, Lin Z, Shi J, Qi Q, Deng G, Li Z, Detection of Hong Kong 97-like H5N1 influenza viruses from eggs of Vietnamese waterfowl. [Epub 2006 Mar 13]. Arch Virol. 2006;151:1615–24. DOIPubMedGoogle Scholar
- Mounts AW, Kwong H, Izurieta HS, Ho Y, Au T, Lee M, Case-control study of risk factors for avian influenza A (H5N1) disease, Hong Kong, 1997. J Infect Dis. 1999;180:505–8. DOIPubMedGoogle Scholar
- Savill NJ, St Rose SG, Keeling MJ, Woolhouse MEJ. Silent spread of H5N1 in vaccinated poultry. Nature. 2006;442:757. DOIPubMedGoogle Scholar
- Webster RG. Wet markets—a continuing source of severe acute respiratory syndrome and influenza? Lancet. 2004;363:234–6. DOIPubMedGoogle Scholar
- Wang M, Di B, Zhou DH, Zheng BJ, Jing H, Lin YP, Food markets with live birds as source of avian influenza. Emerg Infect Dis. 2006;12:1773–5.PubMedGoogle Scholar
Figures
Tables
Cite This Article1These authors contributed equally to this work.
2Members of the Chinese Center for Disease Control and Prevention Avian Influenza H5N1 Study Group are Shuyun Xie, Zhitao Liu, Xuefeng Tang, and Jun Li. Members of the Provincial Center for Disease Control and Prevention Avian Influenza H5N1 Study Group are Gongping Liu, Jianming Ou, Shaojian Cai, Ting Huang, Tie Song, De Wu, and Xiaodong Sun.
Table of Contents – Volume 13, Number 7—July 2007
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Weizhong Yang, Chinese Center for Disease Control and Prevention, 27 Nanwei Rd, Beijing, 100050, People’s Republic of China;
Top