Volume 13, Number 9—September 2007
Dispatch
Mokola Virus in Domestic Mammals, South Africa
Cite This Article
Citation for Media
Abstract
We recently identified 2 Mokola viruses from domestic mammals (a dog and a cat) in South Africa. These cases occurred 8 years after the last reported case of infection with this virus. Our findings emphasize the endemicity of rabies-related lyssaviruses in South Africa and the need to better understand the epidemiology of Mokola viruses.
Mokola virus (MOKV) is classified as genotype (gt) 3 of the genus Lyssavirus in the family Rhabdoviridae (order Mononegavirales). Apart from MOKV, the genus Lyssavirus consists of 6 gts: classic rabies virus (gt1), Lagos bat virus (gt2), Duvenhage virus (gt4), European bat lyssavirus type 1 (gt5) and type 2 (gt6), and Australian bat lyssavirus (gt7). Some novel lyssaviruses identified in bat species in the former Soviet Union are considered putative gts within this genus (1).
Although gt1 viruses have a global distribution, gt5 and gt6 viruses are restricted to Europe and gt7 viruses are limited to Australia. Natural infections with gt2, gt3, and gt4 viruses have been found only in Africa. With the exception of MOKV, all lyssavirus gts and putative gts have been isolated exclusively or most frequently from chiropteran species. MOKV has never been isolated from these species, but only from terrestrial mammals. The first MOKV was isolated from shrews (Crocidura sp.) in Nigeria in 1968. Since then, >20 isolates of this lyssavirus have been found throughout Africa (Cameroon, Central African Republic, Ethiopia, South Africa, and Zimbabwe) (2–12) (Table 1).
We report the identification and characterization of 2 cases of infection with MOKV in South Africa. The first was in a domestic dog and is, to our knowledge, the first such case in South Africa. The second was in a domestic cat, the host species in which all previous isolates were found. The cat MOKV isolate belonged to 1 of 2 previously identified South African MOKV phylogenetic lineages, but the dog MOKV isolate appeared to have a different lineage not previously encountered in South Africa or elsewhere in Africa.
In October 2004, a 3-month-old kitten (Felis domesticus) was adopted from the Society of the Prevention of Cruelty to Animals (East London, Eastern Cape Province, South Africa) and lived with its owner on a farm 23 km outside the city. It had been neutered and had been vaccinated at 10 months of age with an adjuvanted inactivated vaccine against rabies (Rabisin; Merial, Lyon, France), but no subsequent vaccinations were given. The cat spent most of the day indoors, but went out at night and returned in the morning. Unusual behavior was noticed in March 2006. It appeared dull and physically unbalanced and its pupils were dilated but it was not aggressive. The cat was humanely killed, and its brain was sent to the Onderstepoort Veterinary Institute for rabies testing.
On June 17, 2005, a 6-month-old puppy (Canis familiaris) was brought by its owner to a veterinarian in the rural town of Nkomazi (Mpumalanga Province, South Africa). The dog had a temperature of 39.8°C and no appetite. After symptoms were treated, the dog was discharged, but it was brought back 11 days later because it was paralyzed, dehydrated, and had a fixed stare. This animal had never been aggressive to other pets or humans. The dog was humanely killed, and its brain was sent to the Onderstepoort Veterinary Institute for rabies testing.
Direct immunofluorescent antibody test with an anti-rabies conjugate cross-reactive with African lyssaviruses showed numerous and strongly stained inclusion bodies in every field of impression smears of both brain samples. Isolation of virus was attempted by suckling mouse brain passage and cell culture (neuroblastoma cells; Diagnostic Hybrids, Athens, OH, USA); both methods were successful for the cat sample. However, neither method yielded an isolate from the dog sample, despite a lyssavirus-specific reaction in the original brain sample by direct immunofluorescent antibody test.
Subsequently, antigenic characterization was performed with a panel of 16 monoclonal antibodies to the nucleocapsid protein of rabies virus (Canadian Food Inspection Agency, Nepean, Ontario, Canada). Both samples showed reactivity patterns associated with MOKV (Table 2).
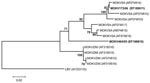
Final confirmation of MOKV in both case samples was obtained by reverse transcription–PCR, nucleotide sequencing, and phylogenetic analysis as described (12). Phylogenetic analysis (Figure) showed that the virus isolated from the cat sample (designated MOKV173/06) belonged to the same lineage of MOKV isolates that were recovered from cats in the same region of South Africa (12). However, the virus detected in the dog sample (designated MOKV404/05) appeared to represent a different South African MOKV lineage that was phylogenetically positioned between known South African and Zimbabwean lineages. This MOKV had nucleotide similarities of 88.1%–90.4% and 85.3%–88.5% with viruses from Zimbabwe and South Africa, respectively.
Infections with MOKV are rare; only 23 isolates are known. During the past 2 decades, all MOKV isolates have been found in South Africa. Because these viruses are not exclusive to South Africa (2–12), lack of isolates from other regions of Africa indicates a lack of active surveillance and limited diagnostic capabilities in many African laboratories. To our knowledge, the 2 cases of infection with MOKV we report are the first in 8 years from South Africa. These cases suggest that other cases may not have been recognized. Clinical signs in the dog and cat, including general neurologic manifestations with a lack of aggression, are often signs that warrant submitting samples for rabies testing.
We have identified regional variations in the antigenic composition of MOKV. Whether these variations are caused by neutral genetic drift or reflect different epidemiologic features is not known. Phylogenetically, divergence of these viruses into different lineages indicates active cycles and evolutionary changes that occur independently, but in close proximity (a few hundred kilometers apart).
Although the epidemiology of MOKV is incomplete, the case for a reservoir host(s) among small terrestrial animals of limited range is supported by our findings. Together with recent isolations of rabies-related lyssaviruses in a human (13) and wild animals (14,15), these reports emphasize the endemicity of these lyssaviruses in South Africa. Public health implications of African rabies-related lyssaviruses should be recognized by laboratory workers, researchers, veterinarians, wildlife personnel, gamekeepers, and pet owners. A better understanding of the epidemiology of these viruses is vital and can only be achieved by improved surveillance and awareness.
Dr Sabeta is a senior research scientist and head of the World Organisation for Animal Health Rabies Reference Laboratory in Onderstepoort, South Africa. His research interests include epidemiologic investigations of rabies and rabies-related viruses in southern Africa.
Acknowledgment
This study was supported by the Rabies Diagnostic Project OVI 15/4/P001.
References
- Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA. Virus taxonomy: the classification and nomenclature of viruses. The eighth report of the international committee on taxonomy of viruses. San Diego: Academic Press; 2004. p. 623–31.
- Shope RE, Murphy FA, Harrison AK, Causey OR, Kemp GE, Simpson DI, Two African viruses serologically and morphologically related to rabies virus. J Virol. 1970;6:690–2.PubMedGoogle Scholar
- Kemp GE, Causey OR, Moore DL, Odelola A, Fabiyi A. Mokola virus. Further studies on IbAn 27377, a new rabies-related etiologic agent of zoonosis in Nigeria. Am J Trop Med Hyg. 1972;21:356–9.PubMedGoogle Scholar
- Familusi JB, Osunkoya BO, Moore DL, Kemp GE, Fabiyi A, Moore DL. A fatal human infection with Mokola virus. Am J Trop Med Hyg. 1972;21:959–63.PubMedGoogle Scholar
- Le Gonidec G, Rickenbach A, Robin Y, Heme G. Isolation of a strain of Mokola virus in Cameroon. Ann Microbiol (Paris). 1978;129:245–9.PubMedGoogle Scholar
- Saluzzo JF, Rollin PE, Daugard C, Digoutte JP, Georges AJ, Sureau P. Premier isolement du virus Mokola a partir d’une rongeur (Lophuromys sikapusi). Ann Inst Pasteur Virol. 1984;135E:57–66. DOIGoogle Scholar
- Foggin CM. Rabies and rabies-related viruses in Zimbabwe: historical, virological and ecological aspects [doctoral dissertation]. Harare (Zimbabwe): University of Zimbabwe; 1988.
- Mebatsion T, Cox JH, Frost JW. Isolation and characterisation of 115 street rabies virus isolates from Ethiopia by using monoclonal antibodies: identification of 2 isolates of Mokola and Lagos bat viruses. J Infect Dis. 1992;166:972–7.PubMedGoogle Scholar
- Meredith CD, Nel LH, von Teichman BF. A further isolation of Mokola virus in South Africa. Vet Rec. 1996;138:119–20.PubMedGoogle Scholar
- von Teichman BF, de Koker WC, Bosch SJ, Bishop GC, Meredith CD, Bingham J. Mokola virus infection: description of recent South African cases and a review of the virus epidemiology. J S Afr Vet Assoc. 1998;69:169–71.PubMedGoogle Scholar
- Bingham J, Javangwe S, Sabeta CT, Wandeler AI, Nel LH. Report of isolations of unusual lyssaviruses (rabies and Mokola virus) identified retrospectively from Zimbabwe. J S Afr Vet Assoc. 2001;72:92–4.PubMedGoogle Scholar
- Nel L, Jacobs J, Jaftha J, von Teichman B, Bingham J. New cases of Mokola virus infection in South Africa: a genotypic comparison of Southern African virus isolates. Virus Genes. 2000;20:103–6. DOIPubMedGoogle Scholar
- Paweska JT, Blumberg LH, Liebenberg C, Hewlett RH, Grobelaar AA, Leman PA, Fatal human infection with rabies-related Duvenhage virus, South Africa. Emerg Infect Dis. 2006;12:1965–7.PubMedGoogle Scholar
- Markotter W, Randles J, Rupprecht CE, Sabeta CT, Taylor PJ, Wandeler AI, Lagos bat virus, South Africa. Emerg Infect Dis. 2006;12:504–6.PubMedGoogle Scholar
- Markotter W, Kuzmin I, Rupprecht CE, Randles J, Sabeta CT, Wandeler AI, Isolation of Lagos bat virus from water mongoose. Emerg Infect Dis. 2006;12:1913–8.PubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 13, Number 9—September 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Claude T. Sabeta, Rabies Unit, Agricultural Research Council–Onderstepoort Veterinary Institute, Private Bag X05, Onderstepoort 0110, Pretoria, South Africa;
Top