Volume 15, Number 1—January 2009
Dispatch
Isolation of Candidatus Bartonella melophagi from Human Blood1
Cite This Article
Citation for Media
Abstract
Candidatus Bartonella melophagi was isolated by blood culture from 2 women, 1 of whom was co-infected with B. henselae. Partial 16S rRNA, RNA polymerase B, and citrate synthase genes and 16S–23S internal transcribed spacer sequences indicated that human isolates were similar to Candidatus B. melophagi.
During the past decade, the number of Bartonella species that are documented human pathogens has rapidly increased (1). Currently, B. bacilliformis, B. quintana, B. henselae, B. elizabethae, B. vinsonii subsp. berkhoffii, B. vinsonii subsp. arupensis, B. koehlerae, B. alsatica, B. washoensis, B. rochalimae, and B. tamiae have been isolated or sequenced from patient samples (1–7). Sheep are the most likely reservoir hosts for Candidatus B. melophagi and sheep keds may be a vector for their transmission among sheep. We report isolation of Candidatus B. melophagi from blood cultures from 2 women. This study was reviewed and approved by the North Carolina State University Institutional Review Board.
Patient 1 was a previously healthy, 51-year-old woman. During July 2004, she visited family residing in rural Ohio and participated in a variety of outdoor activities. Although she saw many wild animals, including deer, she did not report tick attachment or insect bites. Within 24 hours of her return home to North Carolina, a nonpuritic, slightly raised, circular red lesion, approximately the size of a quarter, was noted on the medial aspect of her thigh. Within 3 days, the lesion expanded to the size of a hand. Two weeks later, she exhibited a dry cough, fatigue, muscle pain in the upper body, severe chills, and extreme pain in both feet.
During the next 2 years, these symptoms persisted, along with exertional chest pains, a previously undiagnosed ausculted II to III/VI holosystolic murmur, headaches, difficulty speaking, difficulty sleeping, weakness involving the arms, joint pain, and facial tremors. No abnormalities were shown on an electrocardiogram. An echocardiogram identified mildly thickened aortic and mitral valve leaflets, mild aortic insufficiency, and mild mitral regurgitation.
After the acute illness, the woman reported cycles of illness every 3 to 4 weeks. Results of numerous complete blood counts were normal, with the exception of persistently low neutrophil counts of 2,000–2,500 neutrophils/μL. All serum biochemical parameters remained within normal reference ranges during the 2-year illness. Borrelia burgdorferi C6 peptide and immunoglobulin (Ig) M and IgG antibodies to Babesia microti were not detected. Results of PCRs specific for Anaplasma phagocytophilum, B. microti, and B. burgdorferi were negative. Oral antimicrobial drugs resulted in transient improvement; however, symptoms returned within days after the use of these drugs was stopped. Blood culture resulted in the detection of Candidatus B. melophagi and isolation of B. henselae. Her serum was not reactive with B. henselae or B. vinsonii subsp. berkhoffii antigens.
Treatment with rifampin and azithromycin, started in January 2006, resulted in some overall improvement in symptoms. Cefuroxime was added in February, and the combination resulted in substantial improvement, after which the drugs were selectively withdrawn. For 15 years before the onset of illness, this person had worked as an animal shelter manager in West Virginia and as a veterinary office manager in Virginia. Animal contact was minimal, but she had been bitten by fleas and mosquitoes. Travel history was limited to the eastern and central United States.
Patient 2 was a 65-year-old woman whose condition had been diagnosed as pericarditis of undetermined etiology in September 2004. Six months later, because of residual fatigue and muscle weakness in the arms and legs, mostly on her right side, a blood sample was cultured in Bartonella alpha proteobacteria growth medium (BAPGM).
The woman lived on a farm in southern California with her husband and managed a large animal sanctuary that also housed ≈100 cats and ≈100 dogs. She had resided in southern California for 50 years but occasionally traveled to the southeastern United States and other countries. She was directly involved in daily care of animals and had exposure to pet cattle and sheep, wolf hybrids, lamas, emus, pigs, horses, and numerous pet bird species. Bites and scratches were a daily occurrence, and exposure to cattle and sheep occurred at least weekly. In addition, the woman reported daily exposure to biting flies, occasional exposure to ticks and mosquitoes, and infrequent exposure to fleas or lice. Sheep keds had never been observed on sheep by the attending veterinarian. Blood culture resulted in isolation of Candidatus B. melophagi. Serum was reactive at a titer of 64 to B. henselae, B. vinsonii subsp. berkhoffii, and B. quintana antigens.
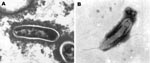
We used BAPGM and other published blood culture methods to test blood samples from both women (2,8,9). Candidatus B. melophagi DNA was amplified directly from blood of patient 2, and from the respective BAPGM enrichment cultures and 14-day subculture colonies from both patients. Sequence analysis of respective colony isolates showed B. henselae (internal transcribed spacer [ITS] sequence identical to Houston 1 strain, data not shown) and Candidatus B. melophagi from patient 1 and Candidatus B. melophagi (isolate 05-HO-1) from patient 2. Both isolates were composed of extremely small gram-negative bacilli consistent with Bartonella spp. Sequence analyses for both isolates are summarized in the Table. Unfortunately, attempts to separate B. henselae and Candidatus B. melophagi colonies from the sample of patient 1 by serial passage were unsuccessful. Bartonella sp. DNA was not amplified from an uninoculated BAPGM control culture or from sheep blood used as a supplement. Flagella, as visualized in the Candidatus B. melophagi strain K-2C isolated from sheep blood (Figure), were not visualized in the human 05-HO-2 strain by transmission electron microscopy.
Based on 16S rRNA, citrate synthase and RNA polymerase B genes, and the 16S–23S ITS region, the bacteria detected in these woman was most likely Candidatus B. melophagi, which was recently isolated from sheep blood and sheep keds (10; M. Kosoy, unpub. data). ITS sequences were nearly identical to those of Wolbachia melophagi detected in a tick removed from sheep in Peru (11). Similar to electron micrographs of the Bartonella sp. isolated from sheep blood (1), no flagella were observed by transmission electron microscopy of the 05-HO-1 human isolate, whereas the sheep ked isolate contains flagella. Because both women had had frequent contact with numerous domestic and wild animals and potential insect vectors, the route of transmission is unknown.
The clinical relevance of Candidatus B. melophagi infection in these women remains to be established. Efforts to passage Candidatus B. melophagi in our laboratory and others (D.A. Bemis, 10) have not been successful. Therefore, development of a serologic assay was not pursued. Nonspecific abnormalities, including difficulty sleeping, muscle weakness, joint pain, and facial tremors, have been reported in association with isolation of B. henselae and B. vinsonii subsp. berkhoffii (2,12). Pericardial or pleural effusions are infrequent complications of B. henselae infection in association with classical cat-scratch disease (13,14).
Before the report of Candidatus B. melophagi in commercial sheep blood sources in 2007 (10), sheep blood was used as a BAPGM supplement in our laboratory. With the exception of these 2 patients, Candidatus B. melophagi was never detected by PCR in >2,250 BAPGM enrichment blood cultures or subculture isolates obtained from animals or humans. In addition, Candidatus B. melophagi DNA was never amplified from >250 BAPGM uninoculated BAPGM enrichment control cultures, and bacterial colonies were never observed after subculture. Beginning in 2007, we also found that some batches of commercial sheep blood contained Candidatus B. melophagi DNA. Therefore, we no longer use blood as a BAPGM supplement. Recently, BAPGM was used to facilitate isolation of B. tamiae from human patients in Thailand (7), and another laboratory has published data supporting the utility of insect cell culture media for growing Bartonella spp. (15).
Dr Maggi is a research assistant professor in the Department of Clinical Sciences at North Carolina State University College of Veterinary Medicine. His research interests include development of novel or improved molecular, diagnostic, and culture methods for detection of Bartonella infections in animals and humans.
Acknowledgments
We thank the attending veterinarian for providing information on the farm, animal populations, and vector exposure for patient 2.
This study was supported by the state of North Carolina and in part by the Sigmon Trust, Bayer Animal Health, IDEXX Laboratories, and a grant from the Southeastern Center for Emerging Biological Threats. R.G.M was partially supported by IDEXX Laboratories.
References
- Chomel BB, Boulouis HJ, Maruyama S, Breitschwerdt EB. Bartonella spp. in pets and effect on human health. Emerg Infect Dis. 2006;12:389–94.PubMedGoogle Scholar
- Breitschwerdt EB, Maggi RG, Duncan AW, Nicholson WL, Hegarty BC, Woods CW. Bartonella species in blood of immunocompetent persons with animal and arthropod contact. Emerg Infect Dis. 2007;13:938–41.PubMedGoogle Scholar
- Fenollar F, Sire S, Raoult D. Bartonella vinsonii subsp. arupensis as an agent of blood culture-negative endocarditis in a human. J Clin Microbiol. 2005;43:945–7. DOIPubMedGoogle Scholar
- Kosoy M, Murray M, Gilmore RD Jr, Bai Y, Gage KL. Bartonella strains from ground squirrels are identical to Bartonella washoensis isolated from a human patient. J Clin Microbiol. 2003;41:645–50. DOIPubMedGoogle Scholar
- Raoult D, Roblot F, Rolain JM, Besnier JM, Loulergue J, Bastides F, First isolation of Bartonella alsatica from a valve of a patient with endocarditis. J Clin Microbiol. 2006;44:278–9. DOIPubMedGoogle Scholar
- Eremeeva ME, Gerns HL, Lydy SL, Goo JS, Ryan ET, Mathew SS, Bacteremia, fever, and splenomegaly caused by a newly recognized Bartonella species. N Engl J Med. 2007;356:2381–7. DOIPubMedGoogle Scholar
- Kosoy M, Morway C, Sheff KW, Bai Y, Colborn J, Chalcraft L, Bartonella tamiae sp. nov., a newly recognized pathogen isolated from three human patients from Thailand. J Clin Microbiol. 2008;46:772–5. DOIPubMedGoogle Scholar
- Duncan AW, Maggi RG, Breitschwerdt EB. A combined approach for the enhanced detection and isolation of Bartonella species in dog blood samples: pre-enrichment liquid culture followed by PCR and subculture onto agar plates. J Microbiol Methods. 2007;69:273–81. DOIPubMedGoogle Scholar
- Breitschwerdt EB, Maggi RG, Sigmon B, Nicholson WL. Isolation of Bartonella quintana from a woman and a cat following putative bite transmission. J Clin Microbiol. 2007;45:270–2. DOIPubMedGoogle Scholar
- Bemis DA, Kania SA. Isolation of Bartonella sp. from sheep blood. Emerg Infect Dis. 2007;13:1565–7.PubMedGoogle Scholar
- Parola P, Shpynov S, Montoya M, Lopez M, Houpikian P, Zeaiter Z, First molecular evidence of new Bartonella spp. in fleas and a tick from Peru. Am J Trop Med Hyg. 2002;67:135–6.PubMedGoogle Scholar
- Breitschwerdt EB, Maggi RG, Nicholson WL, Cherry NA, Woods CW. Bartonella sp. bacteremia associated with neurological and neurocognitive dysfunction. J Clin Microbiol. 2008;46:2856–61. DOIPubMedGoogle Scholar
- Levy PY, Corey R, Berger P, Habib G, Bonnet JL, Levy S, Etiologic diagnosis of 204 pericardial effusions. Medicine (Baltimore). 2003;82:385–91. DOIPubMedGoogle Scholar
- Levy PY, Fournier PE, Carta M, Raoult D. Pericardial effusion in a homeless man due to Bartonella quintana. J Clin Microbiol. 2003;41:5291–3. DOIPubMedGoogle Scholar
- Riess T, Dietrich F, Schmidt KV, Kaiser PO, Schwarz H, Schäfer A, Analysis of a novel insect cell culture medium-based growth medium for Bartonella species. Appl Environ Microbiol. 2008;74:5224–7. DOIPubMedGoogle Scholar
Figure
Table
Cite This Article1Results of this study were presented in part at the 21st Meeting of the American Society for Rickettsiology, Colorado Springs, Colorado, USA, September 8–11, 2007.
Table of Contents – Volume 15, Number 1—January 2009
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Edward B. Breitschwerdt, College of Veterinary Medicine, North Carolina State University, 4700 Hillsborough St, Raleigh, NC 27606, USA;
Top