Volume 18, Number 11—November 2012
Research
Seroprevalence of Antibodies against Chikungunya, Dengue, and Rift Valley Fever Viruses after Febrile Illness Outbreak, Madagascar
Cite This Article
Citation for Media
Abstract
In October 2009, two–3 months after an outbreak of a febrile disease with joint pain on the eastern coast of Madagascar, we assessed serologic markers for chikungunya virus (CHIKV), dengue virus (DENV), and Rift Valley fever virus (RVFV) in 1,244 pregnant women at 6 locations. In 2 eastern coast towns, IgG seroprevalence against CHIKV was 45% and 23%; IgM seroprevalence was 28% and 5%. IgG seroprevalence against DENV was 17% and 11%. No anti-DENV IgM was detected. At 4 locations, 450–1,300 m high, IgG seroprevalence against CHIKV was 0%–3%, suggesting CHIKV had not spread to higher inland-altitudes. Four women had IgG against RVFV, probably antibodies from a 2008 epidemic. Most (78%) women from coastal locations with CHIKV-specific IgG reported joint pain and stiffness; 21% reported no symptoms. CHIKV infection was significantly associated with high bodyweight. The outbreak was an isolated CHIKV epidemic without relevant DENV co-transmission.
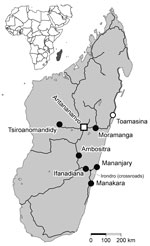
In October 2009, the sentinel surveillance system for early outbreak detection in Madagascar (1) reported an increase of cases of fever with joint pain on the eastern coast. At the beginning of February 2010, chikungunya virus (CHIKV) infection was diagnosed in a patient from the Mananjary district. The International Federation of Red Cross and Red Crescent Societies reported 702 clinically diagnosed cases of chikungunya during February 9–February 15, 2010. Six hundred occurred in the coastal city of Mananjary, and 96 occurred in the small village of Irondro at the crossroads between the towns of Mananjary, Manakara, and Ifanadiana, indicating that the area was a focal point of the epidemic (Figure 1) (2).
Arthropod-borne viruses (arboviruses) such as CHIKV (3), dengue virus (DENV), and Rift Valley fever virus (RVFV) (4,5) are emerging pathogens in the southwestern Indian Ocean region. In 2005–2006, CHIKV caused outbreaks and epidemics on La Réunion, Mauritius, Mayotte, and the Seychelles (6), which caused considerable illness and death (7). Chikungunya appears to occur as an epidemic and an endemic disease in this region. The endemic disease affects mainly populations with high levels of IgG against CHIKV who live in rural areas in Africa (8). The epidemic disease occurs in Asia and the Indian Ocean region in populations in which herd immunity is weak, often in urban areas where Aedes aegypti and Ae. albopictus mosquitoes are the main transmission vectors. During epidemics, humans are the primary reservoirs. Monkeys, rodents, birds, and cattle have been identified as animal reservoirs (9–11). The onset of chikungunya epidemics is acute with high attack rates as seen in 2005–2006 on La Réunion (12). Concurrent epidemics of dengue and chikungunya have been reported from Asia (13) and Africa (14). In 2006, a combined outbreak of dengue fever and chikungunya fever occurred near the Madagascan city of Toamasina (15), but the rest of the country remained unaffected by this epidemic. An increased number of RVFV infections was noticed in Madagascar during the rainy seasons of 2008 and 2009 with 476 (19 fatal) and 236 (7 fatal) suspected cases, respectively (4).
The sudden emergence of chikungunya in the Mananjary area indicated a lack of herd immunity in the affected population. The previous outbreak of CHIKV infection in Madagascar in 2006 in the Toamasina region occurred in conjunction with DENV infection. Because of the recent reports of RVFV infections in animals and humans, our investigation of the recent outbreak in Madagascar included assessment of serologic parameters against CHIKV, DENV, and RVFV.
Approximately 2–3 months after the peak and 1–2 months after the decline of the outbreak of chikungunya, we retrospectively assessed the serologic markers and reported clinical features of women who came for routine pregnancy follow-up visits at 6 geographic locations. By focusing on pregnant women, we could reduce the need to stratify for age and sex and thus minimize the fragmentation of data. The focus could then to be placed on 1) assessing a possible inward spread of the epidemic, 2) evaluating whether the epidemic was limited to CHIKV or due to a simultaneous occurrence of DENV or RVFV infections, and 3) detecting factors associated with an increased risk of CHIKV infection.
The cooperative project of the University of Antananarivo and the Bernhard Nocht Institute for Tropical Medicine was carried out during May–July 2010. Overall, 1,244 pregnant women were included from 6 different sites (Figure 1). Investigations were conducted in 2 coastal cities: Mananjary, the suspected epicenter of the chikungunya epidemic in February, and Manakara. Inland study sites included Ifanadiana, located at the ascending road from the above-mentioned cities to the highlands, the 2 highland cities of Tsiroanomandidy and Ambositra, and Moramanga, a highland city between Antananarivo and Toamasina. The study sites were chosen to include the coastal cities where the suspected chikungunya outbreak was reported (Manajary and Manakara), a city lying at the main road leading to the outbreak region on a moderate elevation level of 466 m (Ifanadiana), and 3 arbitrarily chosen cities in the highlands (Moramanga, Tsiroanomandidy, and Ambositra). Pregnancy follow-up services were chosen for a population comprehensive enough to allow the collection of ≈200 samples within a week. All women attending the routine pregnancy follow-up services were included.
The study was approved by the “Comité d’ethique de la Vice Primature Chargée de la Santé Publique” and discussed with representatives of the World Health Organization (WHO) during a meeting at the WHO office in Antananarivo on April 23, 2010. The study was explained to every participant, and informed consent was either signed (or, in the case of illiteracy, a fingerprint was obtained), and signature of a witness was acquired. Data on the course of pregnancy were obtained from interviews and review of the pregnancy follow-up booklet. To assess symptoms of CHIKV infections, researchers questioned the participants regarding current symptoms, symptoms that occurred since the time of their last menorrhea, or symptoms they recalled from a recent confirmed or suspected CHIKV infection.
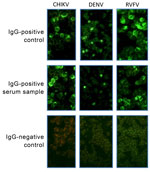
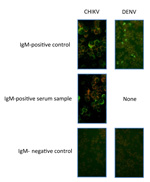
A venous blood sample collected into EDTA was taken for measurement of IgG against CHIKV, DENV, and RVFV. To detect acute CHIKV or DENV infections, we measured levels of IgM against CHIKV and DENV. Immunofluorescence assays (IFAs) for CHIKV, DENV, and RVFV were performed with virus-infected Vero E6 cells as described (16). In brief, Vero cells were spread onto slides, air dried, and fixed in acetone. Plasma samples were serially diluted in phosphate-buffered saline, starting with an initial dilution of 1:10, added to the cells, and incubated for 90 minutes at 37°C. After slides were washed with phosphate-buffered saline, they were incubated with fluorescein isothiocyanate–labeled rabbit antihuman IgG and IgM (SIFIN, Berlin, Germany) at 37°C for 25 minutes (Figure 2, Figure 3). IgG or IgM titers >100 were considered positive.
The serologic controls used for the IFA were the standard routine controls of the WHO Collaborating Centre for Arbovirus and Haemorrhagic Fever Reference and Research in Hamburg, Germany. Moreover, the controls used in this serologic survey were used as external quality assessment samples by the WHO Collaborating Centre for Quality Assurance and Standardization in Laboratory Medicine, Berlin, Germany. The sensitivity and specificity of the IFAs were demonstrated to be 100%, according to results of external quality assessments organized by the European Network for Diagnostics of Imported Viral Diseases (17). The DENV nonstructural protein 1 (NS1) antigen ELISA was performed according to the instructions of the manufacturer (Bio-Rad Laboratories, Hercules, CA, USA). The DENV NS1 antigen is a marker for DENV antigen circulation and thus detects acute infections up to day 21 after onset of symptoms.
The following retrospectively self-reported symptoms were assessed: fever, joint pain, and stiffness, skin symptoms and rashes, hip vibrations and pain, conjunctivitis, and stooped posture. For the 2 coastal locations where seroprevalence of CHIKV was highest, we analyzed potential risk factors for CHIKV infection, taking into account patient’s age, mosquito protective measures (bednet, fan, repellent, air conditioner), and because our study population consisted of pregnant women, we also considered pregnancy-related factors such as parity, weight, and trimester of pregnancy.
Statistical analysis was performed by using STATA version 10 (www.stata.com). The risk factor analysis was done by using univariable and multivariable logistic regressions. If the odds ratios suggested a trend over several categories, a nonparametric test for trend was conducted in the univariable analysis. For multivariable models, a Mantel-Haenszel test for trend, adjusted for the other variables in the model, was performed.
A total of 1,244 women from 6 locations in Madagascar were included in the study. All locations, when data were presented graphically, showed a right-skewed age distribution. The median age was 25 years (range 12–50 years). The median age of women from the 2 coastal locations (Mananjary and Manakara) was not significantly different from that of women from higher altitudes (p = 0.95, by Wilcoxon rank sum test). No relevant difference was found regarding the proportion of primiparas by location (28.4% in the highlands compared with 27.4% at the coast, p = 0.70, by χ2 test). None of the participants had an air conditioner at home; 3 women who lived on the coast reported using a fan. Two women who lived in the highlands reported using mosquito repellents. The only noteworthy mosquito protective measure used frequently was bednets (70.3%). Bednet use was significantly higher for women from the coastal cities of Mananjary (88.2%) and Manakara (90.8%) than for those living in the highland cities of Moramanga (65.7%), Ambositra (21.5%), and Tsiroanomandidy (56.7%). Of those from Ifanadiana, a coastal city at 450-m altitude, 94% used bednets.
Table 1 shows the levels of IgG against CHIKV, DENV, and RVFV, and the results of the IFAs that measured IgM against CHIKV and DENV. IgM against CHIKV was only detected in samples from Mananjary and Manakara taken ≈2–4 months after the peak of the epidemic. Although 27.5% of the samples from Mananjary and 5.2% of the samples from Manakara were positive for IgM against CHIKV, IgM against CHIKV was not detected in samples from other noncoastal locations. IgM against DENV and DENV NS1 antigen, indicators of DENV viremia, were not detected in any location.
A total of 154 of the 1,244 pregnant women (12.4%) were positive for IgG against CHIKV, and 116 of them (75.3%) had reported a history of symptoms of CHIKV infection since their last menorrhea. The highest rates of IgG against CHIKV (Mananjary 44.6%, Manakara 22.7%) and IgM against DENV (Mananjary 17.4%, Manakara 10.8%) were found in the 2 coastal cities (Figure 1; Table 1). IgG against CHIKV and DENV were found in 7.2% and 2.4% in Mananjary and Manakara, respectively. Differences in seroprevalences at the other locations, all at higher altitudes, were negligible, apart from the high frequency of IgG against DENV in Moramanga (11.7%) and Tsiroanomandidy (3.9%). Among persons with IgG against CHIKV, the proportion of women who reported a history of symptoms related to CHIKV infection during the recent outbreak was the same in Mananjary and Manakara (79%). None of the women interviewed from Moramanga reported a history of recent symptoms, even if they had IgG against CHIKV or DENV.
According to the results, only persons with IgG against CHIKV from Mananjary and Manakara could be confidently assigned to the 2009–2010 outbreak. Therefore, reported symptoms and analysis of risk factors for CHIKV infection was confined to these 2 locations (Table 2, Table 3). Risk for previous CHIKV infection increased with body weight (Table 2). This association persisted after adjusting for parity, bednet use, and age (Table 3) and after additionally adjusting for the trimester of pregnancy (Table 3).
In contrast to the outbreak in the Toamasina area of Madagascar in 2006, the outbreak investigated here appears to be exclusively caused by CHIKV infections without concomitant DENV infection. Although one third of the participants in coastal cities at the epicenter of the outbreak were infected with CHIKV, the epidemic did not spread to higher altitudes and further inland. The chikungunya epidemic curve reached its peak in Mananjary in February and abated in March 2010; in Manakara, the epidemic occurred ≈1 month later. The duration of the epidemic roughly corresponds to the rainy season in Madagascar from November to April. Anti-CHIKV IgG was detected in all samples positive for anti-CHIKV IgM, and we assumed that the outbreak had ended before the investigation described here was started. In the noncoastal locations, all samples were negative for IgM against CHIKV, which suggests that the epidemic was constrained to the coast.
By focusing on pregnant women, we achieved a relatively homogenous study population, which facilitated the comparison of differences between study sites by reducing differences between persons (e.g., age or sex) and overfragmentation of the data. In addition to DENV, the presence of RVFV was assessed because this virus has been recently reported in Madagascar (4,18). In 22 persons, IgG against CHIKV and IgG against DENV were detected. The major mosquito vectors of both viruses are equally susceptible to CHIKV and DENV, with simultaneous transmission being confirmed in experimental settings (19,20). Although a considerable number of the samples from Mananjary (17.4%) and from Manakara (10.8%) were positive for IgG against DENV, none was positive for DENV IgM or DENV NS1 antigen, which suggests past DENV infections, independent of the recent chikungunya epidemic.
A limitation of the study is the possibility that some women may have had prior CHIKV infections before the outbreak investigated in this study. A CHIKV outbreak was reported in the Toamasina region in 2006 (15). However, it seems unlikely that a relevant proportion of the resident population of the Mananjary region acquired CHIKV immunity during the Toamasina outbreak because this region is 400 km distant from Mananjary, and no tarred road exists along the coast. The elevated seroprevalance of IgG and IgM against CHIKV in the coastal city of Manakara, which lies 100 km south of Mananjary, suggests that the outbreak spread southwards along the coast.
The International Federation of Red Cross and Red Crescent Societies reported that the town of Irondo, where the inbound roads from Mananjary and Manakara meet, was also affected by the outbreak (2).The town is situated 30 km from the coastline at an altitude of ≈40 m. Although an altitude of 400–500 m does not avert mosquito survival, the low seroprevalence in Ifanadiana (altitude 466 m, 70 km inland) provides evidence against an upward and inbound spread of the epidemic. The long travel distance between Irondro and Ifanadiana, with serpentine roads and long uninhabited stretches between small settlements, may have interrupted transmission chains. Samples taken from the highland cities of Ambositra (1,280 m), Tsiroanomandidy (860 m), and Moramanga (920 m) indicate that the highland population of Madagascar remained unaffected by the recent chikungunya outbreak.
According to our data, 21% of the women from the coastal cities who were positive for IgG against CHIKV did not report symptoms corresponding to those of chikungunya during the outbreak. Similarly, the proportion of asymptomatic infections during a CHIKV outbreak in a naïve population from northeastern Italy in 2007 was 18% (21). In contrast, during the 2005–2006 chikungunya outbreak on La Réunion, only 5% of infections remained asymptomatic (22,23).
Entomologic data were not collected during the chikungunya outbreak on Madagascar. The main vectors for CHIKV are Ae. albopictus and Ae. aegypti mosquitoes. In a study from Gabon, Ae. albopictus mosquitoes outnumbered Ae. aegypti mosquitoes in most suburban areas, and in urban areas, where Ae. aegypti mosquitoes were more commonly found, CHIKV and DENV were only found in Ae. albopictus (19) mosquitoes. Ae. albopictus mosquitoes were the main vectors on La Réunion during the 2005–2006 epidemic.
Recent entomologic data for Madagascar were scarce in 2010, when the chikungunya outbreak took place. In a report from 1989, Ae. albopictus was identified as the predominant Aedes species on the eastern coast and on the central highland plateau, where all our study sites were located (24). A recent study provides evidence for an expansion of an invasive lineage of Ae. albopictus that has spread throughout the island and possibly caused a decline of Ae. aegypti at least in urban areas (25). More research on the vector dynamics of Ae. albopictus and Ae. aegypti mosquitoes in Madagascar is needed.
The analysis of risk and protective factors for infection was confined to the epidemic coastal cities. The finding that bednet use had no influence on the risk of CHIKV infection is in accordance with the fact that the chikungunya vectors Ae. albopictus and Ae. aegypti mosquitoes bite during the daytime. The results indicate a positive association between body weight and the risk for CHIKV infection but the causality could not be assessed in this cross-sectional study. Notably, a study from India found evidence of a higher risk for chronic sequelae among obese persons who acquired chikungunya (26). Future studies on risk factors for chikungunya should include body weight as a possible influence.
The 2009–2010 arboviral outbreak in coastal eastern Madagascar was an isolated CHIKV epidemic without relevant DENV co-transmission. With more than one third of all women affected in the epicenter, the infection rate in the population was high. Data from other locations suggest that the epidemic did not spread to higher altitudes and inland.
Dr Schwarz is a physician and epidemiologist. He currently works in the Infectious Disease Epidemiology Research Group at the Bernhard Nocht Institute for Tropical Medicine in Hamburg, Germany. His scientific interests are the epidemiology of infectious diseases and the training of public health scientists in epidemiology.
Acknowledgments
We thank the women who participated in the study and the health center staff in Mananjary, Manakara, Ifanadiana, Ambositra, Tsiroanomandidy, and Moramanga for their hospitality and support. We also thank the authorities of the Ministry of Health of Madagascar and the Madagascar office of WHO for supporting the project, the administration of the University of Antananarivo, and the Bernhard Nocht Instititute, whose support facilitated the initiation of this bilateral cooperation. In addition, we are grateful to Klaus Jürries for sketching a map of Madagascar and to Lutz Ehlkes for a final revision of the manuscript with a focus on fluency and language.
We gratefully acknowledge financial support from the Deutsche Forschungsgemeinschaft (DFG, grant SCHW 1549/1-1).
References
- Randrianasolo L, Raoelina Y, Ratsitorahina M, Ravolomanana L, Andriamandimby S, Heraud JM, Sentinel surveillance system for early outbreak detection in Madagascar. BMC Public Health. 2010;10:31. DOIPubMedGoogle Scholar
- International Federation of Red Cross and Red Crescent Societies. DREF [Disaster Relief and Emergency Fund] operation. Madagascar: chikungunya epidemic, February 24, 2010. DREF operation no. MDRMG005 [cited 2010 Oct 7]. http://reliefweb.int/node/346169
- Andriamandimby SF, Randrianarivo-Solofoniaina AE, Jeanmaire EM, Ravololomanana L, Razafimanantsoa LT, Rakotojoelinandrasana T, Rift Valley fever during rainy seasons, Madagascar, 2008 and 2009. Emerg Infect Dis. 2010;16:963–70. DOIPubMedGoogle Scholar
- Morvan J, Rollin PE, Laventure S, Rakotoarivony I, Roux J. Rift Valley fever epizootic in the central highlands of Madagascar. Res Virol. 1992;143:407–15. DOIPubMedGoogle Scholar
- World Health Organization. Outbreak news. Chikungunya and dengue, south-west Indian Ocean. Wkly Epidemiol Rec. 2006;81:106–8.PubMedGoogle Scholar
- Renault P, Josseran L, Pierre V. Chikungunya-related fatality rates, Mauritius, India, and Réunion Island. Emerg Infect Dis. 2008;14:1327. DOIPubMedGoogle Scholar
- Pialoux G, Gauzere BA, Jaureguiberry S, Strobel M. Chikungunya, an epidemic arbovirosis. Lancet Infect Dis. 2007;7:319–27. DOIPubMedGoogle Scholar
- Diallo M, Thonnon J, Traore-Lamizana M, Fontenille D. Vectors of Chikungunya virus in Senegal: current data and transmission cycles. Am J Trop Med Hyg. 1999;60:281–6.PubMedGoogle Scholar
- Inoue S, Morita K, Matias RR, Tuplano JV, Resuello RR, Candelario JR, Distribution of three arbovirus antibodies among monkeys (Macaca fascicularis) in the Philippines. J Med Primatol. 2003;32:89–94. DOIPubMedGoogle Scholar
- Wolfe ND, Kilbourn AM, Karesh WB, Rahman HA, Bosi EJ, Cropp BC, Sylvatic transmission of arboviruses among Bornean orangutans. Am J Trop Med Hyg. 2001;64:310–6.PubMedGoogle Scholar
- Renault P, Solet JL, Sissoko D, Balleydier E, Larrieu S, Filleul L, A major epidemic of chikungunya virus infection on Réunion Island, France, 2005–2006. Am J Trop Med Hyg. 2007;77:727–31.PubMedGoogle Scholar
- Kularatne SA, Gihan MC, Weerasinghe SC, Gunasena S. Concurrent outbreaks of chikungunya and dengue fever in Kandy, Sri Lanka, 2006–07: a comparative analysis of clinical and laboratory features. Postgrad Med J. 2009;85:342–6. DOIPubMedGoogle Scholar
- Leroy EM, Nkoghe D, Ollomo B, Nze-Nkogue C, Becquart P, Grard G, Concurrent chikungunya and dengue virus infections during simultaneous outbreaks, Gabon, 2007. Emerg Infect Dis. 2009;15:591–3. DOIPubMedGoogle Scholar
- Ratsitorahina M, Harisoa J, Ratovonjato J, Biacabe S, Reynes JM, Zeller H, Outbreak of dengue and chikungunya fevers, Toamasina, Madagascar, 2006. Emerg Infect Dis. 2008;14:1135–7. DOIPubMedGoogle Scholar
- Tappe D, Schmidt-Chanasit J, Gunther S, Schulze MH, Stich A. Chikungunya and dengue virus antibodies in a traveller with severe arthralgia returning from India. J Clin Virol. 2010;49:148–50. DOIPubMedGoogle Scholar
- Tappe D, Schmidt-Chanasit J, Ries A, Ziegler U, Muller A, Stich A. Ross River virus infection in a traveller returning from northern Australia. Med Microbiol Immunol (Berl). 2009;198:271–3. DOIPubMedGoogle Scholar
- Jeanmaire EM, Rabenarivahiny R, Biarmann M, Rabibisoa L, Ravaomanana F, Randriamparany T, Prevalence of Rift Valley fever infection in ruminants in Madagascar after the 2008 outbreak. Vector Borne Zoonotic Dis. 2011;11:395–402. DOIPubMedGoogle Scholar
- Paupy C, Ollomo B, Kamgang B, Moutailler S, Rousset D, Demanou M, Comparative role of Aedes albopictus and Aedes aegypti in the emergence of dengue and chikungunya in central Africa. Vector Borne Zoonotic Dis. 2010;10:259–66. DOIPubMedGoogle Scholar
- Vazeille M, Mousson L, Martin E, Failloux AB. Orally co-infected Aedes albopictus from La Réunion Island, Indian Ocean, can deliver both dengue and chikungunya infectious viral particles in their saliva. PLoS Negl Trop Dis. 2010;4:e706. DOIPubMedGoogle Scholar
- Moro ML, Gagliotti C, Silvi G, Angelini R, Sambri V, Rezza G, Chikungunya virus in north-eastern Italy: a seroprevalence survey. Am J Trop Med Hyg. 2010;82:508–11. DOIPubMedGoogle Scholar
- Staikowsky F, Le Roux K, Schuffenecker I, Laurent P, Grivard P, Develay A, Retrospective survey of Chikungunya disease in Reunion Island hospital staff. Epidemiol Infect. 2008;136:196–206. DOIPubMedGoogle Scholar
- Staikowsky F, Talarmin F, Grivard P, Souab A, Schuffenecker I, Le Roux K, Prospective study of chikungunya virus acute infection in the Island of La Réunion during the 2005–2006 outbreak. PLoS ONE. 2009;4:e7603. DOIPubMedGoogle Scholar
- Fontenille D, Rodhain F. Biology and distribution of Aedes albopictus and Aedes aegypti in Madagascar. J Am Mosq Control Assoc. 1989;5:219–25.PubMedGoogle Scholar
- Delatte H, Bagny L, Brengue C, Bouetard A, Paupy C, Fontenille D. The invaders: Phylogeography of dengue and chikungunya viruses Aedes vectors, on the South West islands of the Indian Ocean. Infect Genet Evol. 2011;11:1769–81. DOIPubMedGoogle Scholar
- Padmakumar B, Jayan JB, Menon R, Kottarathara AJ. Clinical profile of chikungunya sequelae, association with obesity and rest during acute phase. Southeast Asian J Trop Med Public Health. 2010;41:85–91.PubMedGoogle Scholar
Figures
Tables
Cite This Article1These authors contributed equally to this article.
Table of Contents – Volume 18, Number 11—November 2012
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Norbert G. Schwarz, Bernhard Nocht Institute for Tropical Medicine, Infectious Disease Epidemiology, Bernhard-Nocht-Strasse 74, 20359 Hamburg, Germany
Top