Volume 18, Number 11—November 2012
CME ACTIVITY - Research
Nasopharyngeal Bacterial Interactions in Children
Cite This Article
Citation for Media
Introduction

Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 70% minimum passing score and complete the evaluation at www.medscape.org/journal/eid; (4) view/print certificate.
Release date: October 19, 2012; Expiration date: October 19, 2013
Learning Objectives
Upon completion of this activity, participants will be able to:
• Describe patterns of nasopharyngeal bacterial colonization and interaction in healthy young children, based on an observational study
• Describe the role of Haemophilus influenzae in acute otitis media (AOM) affecting young children, based on an observational study
• Describe the role of other bacteria in AOM affecting young children, based on an observational study
CME Editor
P. Lynne Stockton, VMD, MS, ELS(D), Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: P. Lynne Stockton, VMD, MS, ELS(D), has disclosed no relevant financial relationships.
CME Author
Laurie Barclay, MD, freelance writer and reviewer, Medscape, LLC. Disclosure: Laurie Barclay, MD, has disclosed no relevant financial relationships.
Authors
Disclosures: Qingfu Xu, PhD; Anthony Almudervar, PhD; and Michael E. Pichichero, MD, have disclosed no relevant financial relationships. Janet R. Casey, MD, has disclosed the following relevant financial relationships: received grants for clinical research from Pfizer, Sanofi-Aventis.
Abstract
Antimicrobial treatments and vaccines can alter bacterial interactions in the nasopharynx, thereby altering disease processes. To better understand these interactions, we examined colonization rates of 3 respiratory bacterial pathogens among 320 children when healthy and at onset of acute otitis media (AOM). Bacterial interactions were analyzed with a repeated measures logistic regression model. Among healthy children, Streptococcus pneumoniae and Moraxella catarrhalis were synergistically (positively) associated. Colonization with S. pneumoniae when healthy, but not at onset of AOM, was competitively (negatively) associated with Staphylococcus aureus. Among children with AOM, competitive associations were found between Haemophilus influenzae and S. pneumoniae and between H. influenzae and M. catarrhalis; rates of colonization with H. influenzae were higher. Bacterial interactions result in differing pathogen prevalence during periods of health and at onset of AOM. H. influenzae might become a more common cause of AOM among children who receive pneumococcal conjugate vaccine.
Respiratory bacterial infections, including pneumonia, acute exacerbations of bronchitis, acute sinusitis, and acute otitis media (AOM) among children and adults create major clinical concerns (1,2). The most common bacteria that cause upper respiratory tract infections are Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis (2). The human nasopharynx is an ecologic reservoir of these and other bacteria. A broad variety of commensal bacteria and potential bacterial pathogens colonize the nasopharynx (3,4). Colonization of the nasopharynx is a first, and essential, step toward development of respiratory bacterial infections (3). Viruses can join the microbial mix as a prelude to secondary bacterial infections of the respiratory tract (5–7).
More information about microbial interactions in the nasopharynx is needed (8). These interactions can be altered by therapeutic (e.g., antimicrobial drug) and vaccine (e.g., pneumococcal conjugate vaccination) interventions, resulting in synergistic or competitive outcomes. Information about interactions of the major bacterial respiratory pathogens in the nasopharynx and the conditions conducive to progression to infection (e.g., concurrent viral upper respiratory infections) is limited.
Microbial species can interact synergistically to promote persistence of colonization (positive, or synergistic, association) or they can compete (negative, or competitive, association) (4,9). Interactions between bacteria can alter the composition of a microbial community and affect incidence of disease (4). Several studies have reported competitive associations between colonized S. pneumoniae and S. aureus in the nasopharynx of children, raising concerns that eradication of S. pneumoniae from the nasopharynx by the heptavalent pneumococcal conjugate vaccine (PCV7) might lead to increased S. aureus colonization and subsequent infections (10–13). The introduction of the 13-valent pneumococcal conjugate vaccine will probably exacerbate this effect. Other variables that alter nasopharynx colonization patterns in children include age, gender, daycare attendance, history of having been breast-fed, environmental exposure to tobacco smoke, and otitis-prone condition (14,15).
Several recent reports have described interactions among the 3 major pathogens—S. pneumoniae, H. influenzae, and M. catarrhalis—in young children (8,10,11,16), but the results were contradictory (9). We investigated the interactions of these 3 pathogens in the nasopharynx of young children while healthy (healthy visits) and at onset of AOM (AOM visits). Our aims were to understand differences in nasopharynx colonization rates and bacterial interactions according to the child’s health status.
Study Design and Participants
We analyzed data collected during June 2006–May 2011 from children enrolled in a 5-year prospective study supported by the National Institute of Deafness and Other Communication Disorders. In that study, healthy children with no previous episodes of pneumonia, sinusitis, or AOM were enrolled at 6 months of age from 5 middle-class, suburban pediatric practices in Rochester, New York, USA. Nasopharyngeal and oropharyngeal samples were obtained from healthy children at 6, 9, 12, 15, 18, and 24 months of age and examined for S. pneumoniae, H. influenzae, M. catarrhalis, and S. aureus. If symptoms compatible with an AOM infection developed, a tympanocentesis was performed, as described, to confirm the diagnosis (17). At the time of diagnosis, nasopharyngeal and oropharyngeal samples were obtained for bacterial pathogen cultures. All children received age-appropriate standard vaccinations, including pneumococcal conjugate vaccine (PCV7) (Prevnar; Wyeth Pharmaceuticals, Collegeville, PA, USA).
We analyzed culture data from nasopharyngeal samples collected during 1,183 healthy visits and 334 AOM visits among 320 children 6–24 months of age. All samples included in this study were from children who had not received antimicrobial therapy for at least 3 weeks. Nasopharynx colonization at healthy versus AOM visits was compared among children at 6, 9, 12, 15, 18, and 24 months of age. This time frame includes peak incidence of AOM infection caused by S. pneumoniae, H. influenzae, and M. catarrhalis.
Nasopharyngeal and oropharyngeal samples were obtained for culture as described (18). The pathogens S. pneumoniae, H. influenzae, M. catarrhalis and S. aureus were isolated and identified according to the Manual of Clinical Microbiology (19).
The study was approved by the Institutional Review Board of the University of Rochester and the Rochester General Hospital. Written informed consent was obtained from parents or guardians before the children were enrolled.
Statistical Analyses
The rates of nasopharynx colonization among children of the same age at healthy and AOM visits were compared by using the Fisher exact test and GraphPad Prism software (www.graphpad.com). Bacterial interactions were analyzed by using repeated measures logistic regression models. Predicted outcomes of colonization with S. pneumoniae, H. influenzae, and M. catarrhalis were examined by using multivariate logistic regression. Generalized estimating equations were used to model exchangeable correlation within participants (20). Two logistic regression models (1 for healthy visits and 1 for AOM visits) were calculated by using R version 2.13.2 (www.r-project.org/). To examine the effects of covariates on each of the 3 pathogens, we modeled colonization of each pathogen separately by using the remaining 2 pathogens as predictors and including the interaction term (8). Because few S. aureus were isolated, we did not separately model colonization outcome for S. aureus (8). For each model, we estimated odds ratios (ORs) for the response pathogen given the presence of each predictor pathogen alone, then jointly; synergistic associations between bacteria are indicated by OR>1; competitive associations, by OR<1. The absence of both predictor pathogens was used as the reference condition. p≤0.05 was considered significant; p≤0.01, strongly significant; p≤ 0.1, weakly significant. The model was also used to estimate the OR for each pathogen relative to the risk factors of sex, age, daycare attendance, history of having been breast-fed, environmental exposure to tobacco smoke, and otitis-prone condition.
Polymicrobial Nasopharynx Colonization
Differences in nasopharynx colonization rates between healthy and AOM visits remained generally similar across the longitudinal samplings (Table 1). The rate of nasopharynx colonization by S. pneumoniae for children of all ages at healthy visits, when neither H. influenzae nor M. catarrhalis was present, was 14.2%; this rate did not differ statistically from that at AOM visits (14.4%; p = 0.93) (Table 1). In contrast, the rate of nasopharynx colonization by H. influenzae for children of all ages at AOM visits, when neither S. pneumoniae nor M. catarrhalis was present, was 19.5%; this rate was >4-fold higher than the rate at healthy visits (4.5%; p<0.0001) (Table 1). The rate of nasopharynx colonization by M. catarrhalis for children of all ages at healthy visits, when neither H. influenzae or S. pneumoniae was present, was 20.6%; this rate was 2-fold higher than that for AOM visits (10.5%; p<0.0001) (Table 1).
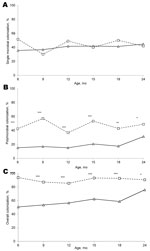
Polymicrobial colonization of the nasopharynx was significantly less common at healthy visits than at AOM visits. At the onset of an infection, polymicrobial colonization increased by 1.5–2.8-fold overall for children of all ages (p<0.05 for all). At healthy visits, the proportion of children with polymicrobial nasopharynx colonization was 18.1% (214/1,183); whereas at AOM visits, the proportion was 45.5% (152/334) (p<0.0001). A comparison of single-pathogen colonization and polymicrobial colonization at ages 6, 9, 12, 15, 18, and 24 months and differences between healthy visits and AOM visits are shown in the Figure.
The overall colonization rates for any of the 3 pathogens were 57.4% (679/1,183) at healthy visits and 89.8% (300/334) at AOM visits (p<0.0001). The culture-positive rates at healthy visits were 30.3% (358/1183) for S. pneumoniae, 11.7% (138/1,183) for H. influenzae, and 36.3% (429/1,183) for M. catarrhalis; whereas at AOM visits, rates were 52.7% (176/334), 47.9% (160/334), and 43.4% (145/334), respectively (p<0.0001 for all) (Table 1).
Bacterial Interactions
The predicted outcome of nasopharynx colonization with S. pneumoniae, H. influenzae, and M. catarrhalis is shown in Table 2. At healthy visits, when colonization with S. pneumoniae was the predicted outcome, S. pneumoniae was synergistically associated with M. catarrhalis colonization (OR 1.42, p = 0.015) but not with H. influenzae colonization (OR 1.33, p = 0.28). When colonization with H. influenzae was the predicted outcome, no significant associations were found between H. influenzae and S. pneumoniae (OR 1.43, p = 0.19) or between H. influenzae and M. catarrhalis (OR 0.81, p = 0.43). When colonization with M. catarrhalis was the predicted outcome, M. catarrhalis colonization was synergistically associated with S. pneumoniae colonization (OR 1.51, p = 0.0059) but not with H. influenzae colonization (OR 0.83, p = 0.49).
At onset of AOM infection, the predictions differed greatly. When colonization with S. pneumoniae was the predicted outcome, S. pneumoniae colonization was competitively associated with H. influenzae colonization (OR 0.40, p = 0.0014) but not with M. catarrhalis colonization (OR 1.02, p = 0.94). When colonization with H. influenzae was the predicted outcome, H. influenzae colonization was competitively associated with S. pneumoniae colonization (OR 0.41, p = 0.0021) and M. catarrhalis colonization (OR 0.37, p = 0.0022). When colonization with M. catarrhalis was the predicted outcome, M. catarrhalis colonization was competitively associated with H. influenzae colonization (OR 0.35, p = 0.0015) but not with S. pneumoniae colonization (OR 1.02, p = 0.95).
The higher prevalence of a potential otopathogen in the nasopharynx need not imply greater association. For example, the marginal rates for the pathogens in the healthy group (all ages) were 0.303, 0.117, and 0.363, the product of which is 0.0129 (Table 1). The actual rate for the occurrence of all 3 is 0.028, indicating an increase in association compared with chance. In contrast, the corresponding rates for the AOM group are 0.527, 0.479, 0.434, the product of which is 0.1096, compared with an actual rate of occurrence of 0.087 for all 3, indicating a decrease in association compared with chance. These totals are therefore compatible with the ORs reported in Table 2.
Effects of Other Risk Factors on Bacterial Colonization
Analysis of the effects of S. aureus colonization and host factors on nasopharynx colonization by S. pneumoniae, H. influenzae, and M. catarrhalis in children when healthy and at onset of AOM indicated that nasopharyngeal cultures were positive for S. aureus at 7.7% (91/1,183) of healthy visits and 5.7% (19/334) of AOM visits (p = 0.23). S. aureus was competitively associated with S. pneumoniae at healthy visits (OR 0.55, p = 0.011) but not at AOM visits (OR 0.95, p = 0.89). No significant associations were identified between S. aureus and H. influenzae or between S. aureus and M. catarrhalis at either type of visit (Table 2). At the healthy visits, daycare attendance was significantly positively associated with S. pneumoniae (OR 1.87, p = 0.0001) and H. influenzae colonization (OR 1.71, p = 0.015) but not with M. catarrhalis colonization (OR 1.28, p = 0.11). At onset of AOM, daycare attendance was significantly positively associated with H. influenzae colonization (OR 2.06, p = 0.0032) but not with S. pneumoniae (OR 1.29, p = 0.28) or M. catarrhalis colonization (OR 0.91, p = 0.70). The otitis-prone condition (defined as 3 episodes of AOM infection within 6 months or 4 AOM infections within 12 months) was significantly positively associated with S. pneumoniae (OR 2.34, p = 0.016) and H. influenzae colonization (OR 3.1, p = 0.0017) at healthy visits but not at AOM visits. We found no significant association between smoking exposures and colonization or between a history of breastfeeding and colonization at either type of visit (Table 2). Being male was positively associated with M. catarrhalis colonization (OR 1.94, p = 0.005) at AOM visits but not at healthy visits.
We found that patterns of nasopharynx colonization associations in children differed at onset of AOM and when healthy. H. influenzae colonization was competitively associated with S. pneumoniae and M. catarrhalis colonization at AOM visits but not at healthy visits; the rates of nasopharynx colonization by H. influenzae were 4-fold higher during AOM visits than during healthy visits.
Our findings suggest that during the PCV era, H. influenzae might increase as a bacterial pathogen of AOM. Our data among young children show that nasopharynx colonization studies of healthy children might not reflect the polymicrobial mix at the time of onset of AOM; the nasopharyngeal environment during onset of AOM favors H. influenzae colonization. It is the mix of bacteria at time of infection that determines which organisms are most likely to cause infection (12). We have previously shown that among children, the changes in nasopharynx colonization by S. pneumoniae and H. influenzae caused by PCV7 resulted in a remarkable proportionate decrease in AOM infection caused by S. pneumoniae and a proportionate increase in AOM infection caused by H. influenzae (17,21,22). At onset of AOM, when H. influenzae co-colonizes with S. pneumoniae or M. catarrhalis, H. influenzae predominates over these 2 bacteria to cause AOM (18). The elimination of S. pneumoniae strains expressing PCV7 serotypes has resulted in the remaining S. pneumoniae strains, except serotype 19A, competing less effectively with H. influenzae in the nasopharynx (18). Therefore, with a further reduction in nasopharynx colonization by S. pneumoniae, including strains expressing serotype 19A (as will probably result from the recent introduction of PCV13 in some countries), our data suggest that H. influenzae might fill the nasopharyngeal niche at the onset of AOM, and consequently, H. influenzae might become a more prominent cause of AOM.
S. aureus also can cause respiratory infections; several reports have suggested that S. aureus might be replacing S. pneumoniae as a dominant nasopharynx colonizer as a consequence of the introduction of PCV. Concern has been expressed that S. aureus might emerge as a more prominent respiratory pathogen (10–13,23). However, our results do not support that S. aureus has emerged as a frequent pathogen of AOM after introduction PCV7.
Knowledge regarding interactions among S. pneumoniae, H. influenzae, and M. catarrhalis is limited, appears contradictory, and is confined to studies among children. Zemlickova et al. found no significant association between S. pneumoniae and H. influenzae or between S. pneumoniae and M. catarrhalis in the nasopharynx of 425 healthy children, 3–6 years of age, in the Czech Republic (11). Madhi et al. found synergistic associations between S. pneumoniae and H. influenzae in PCV9-vaccinated healthy children, 5 years of age, in South Africa (10). Jacoby et al. found synergistic associations between nasopharynx colonization by S. pneumoniae, H. influenzae, and M. catarrhalis in healthy children, 1–24 months of age, but no significant association between S. pneumoniae and S. aureus or between H. influenzae and S. aureus (16). At onset of respiratory viral infections and in association with AOM infection, Pettigrew et al. (8) found competitive associations between colonization by S. pneumoniae and H. influenzae, H. influenzae and M. catarrhalis, S. pneumoniae and S. aureus, and H. influenzae and S. aureus in children 6–36 months of age. Thus, our results comparing nasopharynx colonization patterns during times of health with patterns at onset of AOM help explain the prior contradictory results.
Although we found a synergistic association between S. pneumoniae and M. catarrhalis at healthy visits and a significant increase of polymicrobial colonization at AOM visits, the tendency toward polymicrobial colonization did not result in synergistic associations at AOM visits. On the contrary, competitive associations between H. influenzae and S. pneumoniae and between H. influenzae and M. catarrhalis were found at AOM visits. The increases in colonization of individual bacterial pathogens during AOM might randomly result in an increase of polymicrobial colonization and might not result from synergistic associations among the potential pathogens. Further study on the mechanism is needed.
The mechanisms to explain competitive and synergistic interactions among S. pneumoniae, H. influenzae, M. catarrhalis, and S. aureus have been explored. In a mouse model, Lysenkoet al. found that when H. influenzae colonized with S. pneumoniae in the nasopharynx, S. pneumoniae was rapidly cleared (24). The competitive interaction was dependent on cellular components of H. influenzae activating the host innate immune response involving complement and neutrophils; the end result was the killing of S. pneumoniae (24). The mouse model results are consistent with our observations among children but differ from results observed during in vitro experiments that predict that S. pneumoniae should inhibit the growth of H. influenzae (25,26). A competitive association between S. pneumoniae and S. aureus might be mediated by S. pneumoniae production of hydrogen peroxide (25,27). Our results and those of others suggest that this mechanism might be active during times of health but not, on the basis of our observation, at onset of AOM (10,11,13,28).
We did evaluate the effects of age, sex, daycare attendance, breast-feeding history, exposure to tobacco smoke, and an immunologically driven increased susceptibility to AOM (15,29) in our study population. The results differed at healthy versus AOM visits except for the effect of daycare attendance on H. influenzae colonization; daycare attendance had a positive association with H. influenzae colonization at both healthy and AOM visits. Others have shown daycare attendance to be consistently associated with increased colonization by S. pneumoniae, H. influenzae, and M. catarrhalis in healthy children and in children with viral upper respiratory infections (8). Our previous studies have shown that children who are prone to otitis have much weaker immune responses to S. pneumoniae surface antigens than those who are not prone (15,30). We therefore assessed the otitis-prone condition as a predictor in the models in this study. We found that the otitis-prone condition was positively associated with S. pneumoniae and H. influenzae colonization at healthy visits but not at AOM visits.
The rates of nasopharynx colonization with S. pneumoniae, H. influenzae, and M. catarrhalis and the mix at onset of AOM that we observed occurred mostly in the context of a concurrent viral upper respiratory tract infection. A limitation of our study is that we did not evaluate bacterial–viral interactions. Revai et al. (31) and Chonmaitree et al. (32) found that among young children, viral upper respiratory tract infections preceded >90% of AOM infections. At onset of AOM, 93% of the children in our study had clinical signs of a viral upper respiratory tract infection; the predominant viruses detected were influenza, parainfluenza, respiratory syncytial, and adenovirus (A. Chang, unpub. data). The culture and PCR methods used in that study did not identify rhinovirus, metapneumovirus, or bocavirus. Previous studies have shown that viral upper respiratory tract infections affect bacterial nasopharynx colonization (5,7). Respiratory viruses up-regulate epithelial cell receptors for some species of respiratory bacteria (7); they cause inflammation and down-regulate innate and adaptive host defenses (7,33). Different viruses have varying effects on different bacteria (34–36). The comparison of healthy visits with AOM visits suggests that viral infections alter the host nasopharynx environment by facilitating a shift in the polymicrobial mix and enhancing polymicrobial colonization.
We did not study the additional effect of antimicrobial drugs on nasopharynx colonization. Such treatment modifies nasopharynx colonization patterns. Pettigrew et al. (8) showed that antimicrobial drug therapy was associated with a lower prevalence of colonization with S. pneumoniae and M. catarrhalis but not with H. influenzae. Varon et al. (37) showed that colonization by S. pneumoniae, H. influenzae, and M. catarrhalis decreased after antimicrobial drug thereapy; the reduction in colonization was less for H. influenzae than for S. pneumoniae or M. catarrhalis. Current national guidelines endorse the use of amoxicillin for first-line treatment of AOM, sinusitis, and community-acquired pneumonia in children (38–40). Amoxicillin is ineffective for eradicating β-lactamase–producing H. influenzae and M. catarrhalis. We have previously shown that ≈65% of H. influenzae and 100% of M. catarrhalis colonizing the nasopharynx of children in our study population elaborate β-lactamase (17,21,22). Therefore, empiric treatment with amoxicillin would probably increase H. influenzae and M. catarrhalis nasopharynx colonization in children.
Our results led us to 2 conclusions. First, nasopharyngeal bacterial interactions among S. pneumoniae, H. influenzae, and M. catarrhalis differ during health and at onset of AOM in young children. Second, at the onset of AOM, the nasopharynx environment among children vaccinated with PCV7 is favorable for H. influenzae colonization. Consequently, our results predict that H. influenzae might be become a more prominent bacterial pathogen of AOM in the era of PCV. Further studies of virus–bacterium–host interactions in the nasopharynx and additional studies of the mechanisms driving the observed shifts in bacterial species and polymicrobial makeup are needed.
Dr Xu is a research scientist II at Rochester General Hospital Research Institute. His primary research interests include molecular mechanisms of bacterial–vial interactions and co-pathogenesis of respiratory tract infections, mucosal immune responses, and vaccine development.
Acknowledgments
We thank Sally Thomas and the nurses, staff, and collaborating pediatricians from Long Pond Pediatrics, Genesis Pediatrics, Rainbow Pediatrics, Lewis Pediatrics, and Legacy Pediatrics and the parents who consented to the long and challenging study. We also thank Diana G. Adlowitz, Arthur Chang, and Jennifer Wills for assistance with pathogen identification by culture.
This work was supported by the National Institutes of Health, National Institute on Deafness and Other Communication Disorders (RO1DC008671-01A2), by the Thrasher Research Fund (Salt Lake City, UT, USA) award no. 02823-2, and by an investigator-initiated research grant from Wyeth Pharmaceuticals (Collegeville, PA, USA) (all to M.E.P.).
References
- Brunton S. Treating community-acquired bacterial respiratory tract infections: update on etiology, diagnosis, and antimicrobial therapy. J Fam Pract. 2005;54:357–64.PubMedGoogle Scholar
- Cappelletty D. Microbiology of bacterial respiratory infections. Pediatr Infect Dis J. 1998;17(Suppl):S55–61. DOIPubMedGoogle Scholar
- Leiberman A, Dagan R, Leibovitz E, Yagupsky P, Fliss DM. The bacteriology of the nasopharynx in childhood. Int J Pediatr Otorhinolaryngol. 1999;49(Suppl 1):S151–3. DOIPubMedGoogle Scholar
- Laufer AS, Metlay JP, Gent JF, Fennie KP, Kong Y, Pettigrew MM. Microbial communities of the upper respiratory tract and otitis media in children. MBio. 2011;2:e00245–10. DOIPubMedGoogle Scholar
- Heikkinen T, Chonmaitree T. Importance of respiratory viruses in acute otitis media. Clin Microbiol Rev. 2003;16:230–41. DOIPubMedGoogle Scholar
- Wald ER, Guerra N, Byers C. Upper respiratory tract infections in young children: duration of and frequency of complications. Pediatrics. 1991;87:129–33.PubMedGoogle Scholar
- Bakaletz LO. Immunopathogenesis of polymicrobial otitis media. J Leukoc Biol. 2010;87:213–22. DOIPubMedGoogle Scholar
- Pettigrew MM, Gent JF, Revai K, Patel JA, Chonmaitree T. Microbial interactions during upper respiratory tract infections. Emerg Infect Dis. 2008;14:1584–91. DOIPubMedGoogle Scholar
- Murphy TF, Bakaletz LO, Smeesters PR. Microbial interactions in the respiratory tract. Pediatr Infect Dis J. 2009;28(Suppl):S121–6. DOIPubMedGoogle Scholar
- Madhi SA, Adrian P, Kuwanda L, Cutland C, Albrich WC, Klugman KP. Long-term effect of pneumococcal conjugate vaccine on nasopharyngeal colonization by Streptococcus pneumoniae—and associated interactions with Staphylococcus aureus and Haemophilus influenzae colonization—in HIV-infected and HIV-uninfected children. J Infect Dis. 2007;196:1662–6. DOIPubMedGoogle Scholar
- Zemlicková H, Urbásková P, Adámková V, Motlová J, Lebedová V, Procházka B. Characteristics of Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis and Staphylococcus aureus isolated from the nasopharynx of healthy children attending day-care centres in the Czech Republic. Epidemiol Infect. 2006;134:1179–87. DOIPubMedGoogle Scholar
- Bogaert D, van Belkum A, Sluijter M, Luijendijk A, de Groot R, Rumke HC, Colonisation by Streptococcus pneumoniae and Staphylococcus aureus in healthy children. Lancet. 2004;363:1871–2. DOIPubMedGoogle Scholar
- Regev-Yochay G, Dagan R, Raz M, Carmeli Y, Shainberg B, Derazne E, Association between carriage of Streptococcus pneumoniae and Staphylococcus aureus in children. JAMA. 2004;292:716–20. DOIPubMedGoogle Scholar
- Stenström C, Ingvarsson L. Otitis-prone children and controls: a study of possible predisposing factors. 2. Physical findings, frequency of illness, allergy, day care and parental smoking. Acta Otolaryngol. 1997;117:696–703. DOIPubMedGoogle Scholar
- Sharma SK, Casey JR, Pichichero ME. Reduced memory CD4+ T-cell generation in the circulation of young children may contribute to the otitis-prone condition. J Infect Dis. 2011;204:645–53. DOIPubMedGoogle Scholar
- Jacoby P, Watson K, Bowman J, Taylor A, Riley TV, Smith DW, Modelling the co-occurrence of Streptococcus pneumoniae with other bacterial and viral pathogens in the upper respiratory tract. Vaccine. 2007;25:2458–64. DOIPubMedGoogle Scholar
- Casey JR, Adlowitz DG, Pichichero ME. New patterns in the otopathogens causing acute otitis media six to eight years after introduction of pneumococcal conjugate vaccine. Pediatr Infect Dis J. 2010;29:304–9.PubMedGoogle Scholar
- Xu Q, Casey JR, Chang A, Pichichero M. When co-colonizing the nasopharynx Haemophilus influenzae predominates over Streptococcus pneumoniae except serotype 19A strains to cause acute otitis media. Pediatr Infect Dis J. 2012;31:638–40.
- Murray PR, Pfaller MA, Jorgensen JH, Yolken RH, editors. Manual of clinical microbiology. 8th ed; 2003. p. 412–29, 636–48, 770–802, 390–411.
- Prentice RL, Zhao LP. Estimating equations for parameters in means and covariances of multivariate discrete and continuous responses. Biometrics. 1991;47:825–39. DOIPubMedGoogle Scholar
- Pichichero ME, Casey JR. Emergence of a multiresistant serotype 19A pneumococcal strain not included in the 7-valent conjugate vaccine as an otopathogen in children. JAMA. 2007;298:1772–8. DOIPubMedGoogle Scholar
- Casey JR, Pichichero ME. Changes in frequency and pathogens causing acute otitis media in 1995–2003. Pediatr Infect Dis J. 2004;23:824–8. DOIPubMedGoogle Scholar
- Veenhoven R, Bogaert D, Uiterwaal C, Brouwer C, Kiezebrink H, Bruin J, Effect of conjugate pneumococcal vaccine followed by polysaccharide pneumococcal vaccine on recurrent acute otitis media: a randomised study. Lancet. 2003;361:2189–95. DOIPubMedGoogle Scholar
- Lysenko ES, Ratner AJ, Nelson AL, Weiser JN. The role of innate immune responses in the outcome of interspecies competition for colonization of mucosal surfaces. PLoS Pathog. 2005;1:e1. DOIPubMedGoogle Scholar
- Pericone CD, Overweg K, Hermans PW, Weiser JN. Inhibitory and bactericidal effects of hydrogen peroxide production by Streptococcus pneumoniae on other inhabitants of the upper respiratory tract. Infect Immun. 2000;68:3990–7. DOIPubMedGoogle Scholar
- Shakhnovich EA, King SJ, Weiser JN. Neuraminidase expressed by Streptococcus pneumoniae desialylates the lipopolysaccharide of Neisseria meningitidis and Haemophilus influenzae: a paradigm for interbacterial competition among pathogens of the human respiratory tract. Infect Immun. 2002;70:7161–4. DOIPubMedGoogle Scholar
- Regev-Yochay G, Trzcinski K, Thompson CM, Malley R, Lipsitch M. Interference between Streptococcus pneumoniae and Staphylococcus aureus: in vitro hydrogen peroxide–mediated killing by Streptococcus pneumoniae. J Bacteriol. 2006;188:4996–5001. DOIPubMedGoogle Scholar
- Regev-Yochay G, Raz M, Dagan R, Porat N, Shainberg B, Pinco E, Nasopharyngeal carriage of Streptococcus pneumoniae by adults and children in community and family settings. Clin Infect Dis. 2004;38:632–9. DOIPubMedGoogle Scholar
- Sharma SK, Casey JR, Pichichero ME. Reduced serum IgG responses to pneumococcal antigens in otitis prone children may be due to poor memory B-cell generation. J Infect Dis. 2011;204:645–53..PubMedGoogle Scholar
- Kaur R, Casey JR, Pichichero ME. Serum antibody response to five Streptococcus pneumoniae proteins during acute otitis media in otitis-prone and non-otitis-prone children. Pediatr Infect Dis J. 2011;30:645–50. DOIPubMedGoogle Scholar
- Revai K, Mamidi D, Chonmaitree T. Association of nasopharyngeal bacterial colonization during upper respiratory tract infection and the development of acute otitis media. Clin Infect Dis. 2008;46:e34–7. DOIPubMedGoogle Scholar
- Chonmaitree T, Revai K, Grady JJ, Clos A, Patel JA, Nair S, Viral upper respiratory tract infection and otitis media complication in young children. Clin Infect Dis. 2008;46:815–23. DOIPubMedGoogle Scholar
- Ratner AJ, Lysenko ES, Paul MN, Weiser JN. Synergistic proinflammatory responses induced by polymicrobial colonization of epithelial surfaces. Proc Natl Acad Sci U S A. 2005;102:3429–34. DOIPubMedGoogle Scholar
- Tong HH, Fisher LM, Kosunick GM, DeMaria TF. Effect of adenovirus type 1 and influenza A virus on Streptococcus pneumoniae nasopharyngeal colonization and otitis media in the chinchilla. Ann Otol Rhinol Laryngol. 2000;109:1021–7.PubMedGoogle Scholar
- Suzuki K, Bakaletz LO. Synergistic effect of adenovirus type 1 and nontypeable Haemophilus influenzae in a chinchilla model of experimental otitis media. Infect Immun. 1994;62:1710–8.PubMedGoogle Scholar
- Bakaletz LO, Murwin DM, Billy JM. Adenovirus serotype 1 does not act synergistically with Moraxella (Branhamella) catarrhalis to induce otitis media in the chinchilla. Infect Immun. 1995;63:4188–90.PubMedGoogle Scholar
- Varon E, Levy C, De La Rocque F, Boucherat M, Deforche D, Podglajen I, Impact of antimicrobial therapy on nasopharyngeal carriage of Streptococcus pneumoniae, Haemophilus influenzae, and Branhamella catarrhalis in children with respiratory tract infections. Clin Infect Dis. 2000;31:477–81. DOIPubMedGoogle Scholar
- Cincinnati Children’s Hospital Medical Center. Evidence-based care guideline for community acquired pneumonia in children 60 days through 17 years of age. Rockville (MD): Agency for Healthcare Research and Quality, National Guideline Clearinghouse; 2001 (revised 2006).
- Cincinnati Children’s Hospital Medical Center. Evidence-based care guideline for management of acute bacterial sinusitis in children 1 to 18 years of age. Rockville (MD): Agency for Healthcare Research and Quality, National Guideline Clearinghouse; 2000 (revised 2006).
- University of Michigan Health System. Otitis media. Rockville (MD): Agency for Healthcare Research and Quality, National Guideline Clearinghouse; 2002 (revised 2007).
Figure
Tables
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 70% passing score) and earn continuing medical education (CME) credit, please go to www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/category/2922.html. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title: Nasopharyngeal Bacterial Interactions in Children
CME Questions
1. Based on the observational study by Dr. Xu and colleagues, which of the following statements about patterns of nasopharyngeal bacterial colonization and interaction in healthy young children is most likely correct?
A. Streptococcus pneumoniae is positively associated with Staphylococcus aureus
B. S. pneumoniae is positively associated with Moraxella catarrhalis
C. M. catarrhalis is positively associated with S. aureus
D. Patterns of nasopharyngeal bacterial colonization and interaction in health are likely to predict those at onset of acute otitis media (AOM)
2. Your patient is a 1-year-old, PCV7-vaccinated male with AOM. Based on the observational study by Dr. Xu and colleagues, which of the following statements about the role of Haemophilus influenzae is most likely correct?
A. The nasopharyngeal environment is likely to be unfavorable to H. influenzae colonization
B. At AOM onset, H. influenzae is positively associated with S. pneumoniae
C. At AOM onset, H. influenzae is positively associated with M. catarrhalis
D. H. influenzae may become a more important cause of AOM in pneumococcal conjugate vaccinated children
3. Based on the observational study by Dr. Xu and colleagues, which of the following statements about the role of other bacteria in AOM affecting the patient described in question 2 would most likely be correct?
A. During AOM, S. pneumoniae colonization is negatively associated with S. aureus
B. Viral-bacterial-host interactions in the nasopharynx during AOM are completely characterized
C. Findings of this study support the role of S. aureus as a frequent pathogen of AOM
D. Elimination of S. pneumoniae PCV7 strains has resulted in the remaining strains competing less effectively with H. influenzae in the nasopharynx and as a cause of AOM infection
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
Related Links
Table of Contents – Volume 18, Number 11—November 2012
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Michael E. Pichichero, Rochester General Hospital Research Institute, Center for Infectious Diseases and Immunology, 1425 Portland Ave, Rochester, NY 14621, USA
Top