Volume 19, Number 12—December 2013
Dispatch
Lack of MERS Coronavirus Neutralizing Antibodies in Humans, Eastern Province, Saudi Arabia
Figure 1
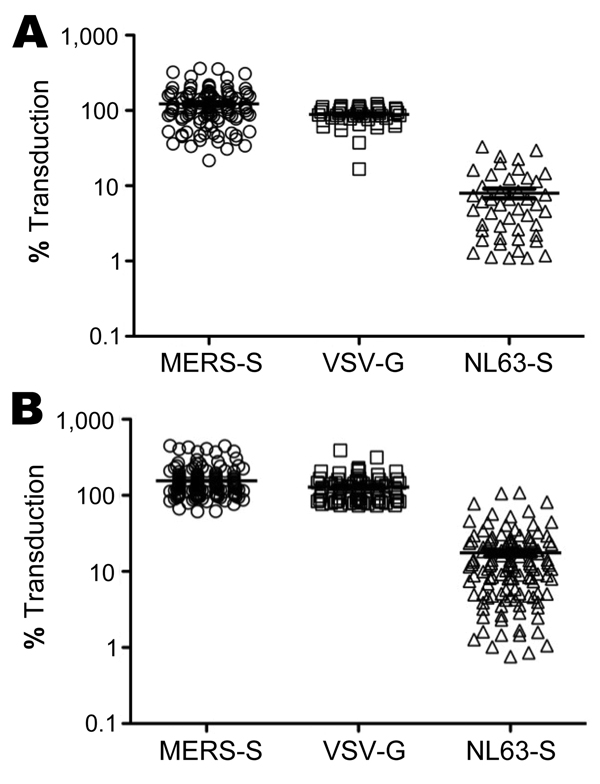
Figure 1. . Neutralizing activity of serum and plasma samples obtained from patients at King Fahd Hospital of the University in Alkhobar, Saudi Arabia. A) Lentiviral vectors encoding luciferase and bearing the indicated viral glycoproteins were incubated with 1:20 dilutions of plasma from healthy adults, obtained during December 2012, and then added to target cells. Transduction efficiency was measured by quantification of luciferase activities in cell lysates and is shown relative to transduction of cells in the absence of serum, which was set at 100%. All 110 plasma samples available were tested for neutralization of Middle East respiratory syndrome coronavirus spike protein (MERS-S)–dependent transduction; subsets were also tested for neutralization of transduction driven by the G protein of vesicular stomatitis virus (VSV-G) (46/110) and the S protein of human coronavirus NL63 (NL63-S) (46/110). B) Analysis conducted as described for panel A using 158 serum samples from children with lower respiratory tract infections, obtained during May 2010–May 2011. All samples were analyzed for neutralization of MERS-S–mediated transduction; subsets were also tested for neutralization of transduction driven by VSV-G (76/158) and NL63-S (123/158). Horizontal lines indicate mean ±SEM.
1These authors contributed equally to this article.