Volume 20, Number 6—June 2014
Dispatch
Fatal Monkeypox in Wild-Living Sooty Mangabey, Côte d’Ivoire, 2012
Cite This Article
Citation for Media
Abstract
We isolated a monkeypox virus from a wild-living monkey, a sooty mangabey, found dead in Taï National Park, Côte d’Ivoire, in March 2012. The whole-genome sequence obtained from this isolate and directly from clinical specimens showed its close relationship to monkeypox viruses from Western Africa.
Among the poxviruses are several species of orthopoxviruses (OPVs) that are pathogenic to humans, including monkeypox virus (MPXV) and variola virus (VARV). MPXV was first discovered in laboratory captive monkeys in Copenhagen in 1958 (1). After the eradication of VARV during the 1970s, MPXV became the highest pathogenic OPV infection in humans. On the basis of epidemiologic and sequence data, strains of MPXV can be assigned to a West African or a Congo Basin clade; viruses from the Congo Basin clade show more pronounced illness, death, viremia, and human-to-human transmission than do strains from the West African clade (2,3).
Serologic studies showed that monkeys from Africa have OPV antibodies, but no natural case of MPXV has been reported in wild-living primates (4,5). The only MPXV isolate obtained from an animal in the wild was from a Thomas’s rope squirrel (Funisciurus anerythrus) caught in Democratic Republic of the Congo in 1985 (6). Here we describe natural MPXV infection in a sooty mangabey (Cercocebus atys) found dead in Taï National Park (TNP), Côte d’Ivoire.
During a long-term program to monitor deaths in wildlife, an infant mangabey was found dead in the TNP in March 2012. The body did not show any apparent injuries, and the animal had died relatively recently, as indicated by the presence of blowfly eggs but absence of maggots. Multiple skin lesions typical of MPXV infection occurred as dark red crusts 5–7 mm in diameter, partly confluent, which were disseminated over the body. Extremities were mainly affected; fewer lesions were seen on the belly and none on the back.
A full necropsy was performed under high-level safety measures, and samples of all organs and blood were collected and preserved in liquid nitrogen and 10% buffered formalin (7). Histologic analysis of the skin showed eosinophilic inclusion bodies, suggesting that an OPV infection had caused the ulcers. Severe bacterial secondary infection of the ulcers also was observed, as well as bacteremia that might have contributed to the pathologic changes and death.
DNA was extracted from different tissues, and quantitative PCR for OPV DNA (rpo18) and a cellular target (c-myc) were performed as described (8). We found a high viral DNA load in relation to cellular DNA in all tissues, except muscle, indicating a systemic infection, with particularly high loads in a skin lesion and from a throat swab sample (Table). An immunofluorescence assay performed on MPXV-infected cells showed titers of 320 for IgG and 80 for IgM, indicating an acute OPV infection.
Virus from skin tissue was propagated in HEp-2 cells, infected cells were harvested, and DNA was extracted. We performed library preparation and sequencing on an Ion Torrent PGM with an Ion PGM Sequencing 200 Kit (Life Technologies, Darmstadt, Germany) (average read length = 93 bases). Sequences were analyzed by using Geneious (Biomatters, Auckland, NZ). At first, a de novo assembly from PGM fastq-data was conducted, followed by mapping of the resulting contigs to all 11 MPXV genomes published in the National Center for Biotechnology Information to obtain orientation and to form a consensus sequence. The reads were mapped again to the consensus sequences to identify assembly errors. These efforts resulted in a single 197,571-bp genome. Additionally, DNA preparations from the mangabey’s skin and throat swab specimens were subjected directly to an Illumina HiSeq 1500 (San Diego, CA, USA), sequencing 150 + 150 bases (paired end). The genome could also be assembled to 190,562 bp by using Illumina data only (2.9% of the 285 million Illumina reads could be used for the MPXV assembly, compared with 45% of the 3.5 million reads from the PGM), indicating that the viral genome can be assembled by sequencing specimens of high viral load directly without previous virus propagation.
Finally, we obtained a 12,395-fold coverage of the MPXV genome from Illumina data and a 732-fold coverage for data from the PGM. Eighty-four percent of the reads from the skin sample could be mapped to Staphylococcus aureus. These data provide evidence of the usability of direct Illumina sequencing for metagenomic analysis. Combining gained sequence data showed a full genome of MPXV-TNP with 200,035 bp (GenBank accession no. KJ136820).
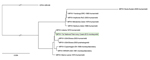
All known MPXV genomes, including MPXV-TNP, and the genome of cowpox virus GRI-90 (used as outgroup) were truncated to include the sequence information between the first and last coding region for further phylogenetic analysis (MrBayes, v. 3.2.1 (http://mrbayes.sourceforge.net/), with gaps as binary model). MPXV-TNP is closely related to a human MPXV isolate detected in the neighboring country of Liberia in 1970 (Figure 1). Together with other isolates from this geographic region, MPXV-TNP belongs to the West African clade of MPXV (2,9). Additionally, protein sequences of MPXV-TNP were more similar to those of sequences from the West African clade than to those of the Congo Basin clade (Figure 2).
Sequence comparison of West African and Congo Basin MPXVs by other researchers showed individual genes that are conserved across the 2 clades and are speculated to be responsible for the different pathogenicity of the viruses (2,3). For example, we analyzed the sequence of the immunomodulatory protein IL-1β receptor (B14R), which moderated the severity of vaccinia virus infection in a mouse model (10). MPXV-TNP encodes a further truncated sequence variant of the IL-1β protein (confirmed by Sanger sequencing) that is unique among known MPXV strains. Unfortunately, the effects of IL-1β receptor fragmentation on functionality and on virulence of MPXV is not known (11).
The case described here suggests that mangabeys can be fatally infected with MPXV in nature and have high viral loads found in various tissues. Phylogenetic analysis showed that the newly identified MPXV-TNP strain is closely related to MPXV isolated from humans in Liberia and Sierra Leone in 1970 and can be assigned to the less virulent West African clade of MPXV, which seems surprising. A possible explanation is that MPXV disease progression appears to be rare in wild-living monkeys, and this case was the first observed in both regions: Central Africa, with more lethal strains, and West Africa, with less lethal strains. Also, the mangabey was only a few weeks of age and might not have had a fully developed immune system, and this factor and its secondary bacterial infection were likely to have contributed to disease severity. Moreover, individual immunologic defects cannot be ruled out. Nevertheless, very few monkeys are under systematic observation by humans, and infection of humans with the MPXV-TNP strain cannot be ruled out, even though no cases in humans were recognized in this area during the same period.
This case demonstrates that wild primates can serve as indicators for specific pathogens in certain regions (12). The local human population hunts and eats potential reservoir species, such as rodents, and therefore follow-up investigations of human infections and the prevalence of MPXV in reservoir species are needed to pinpoint the zoonotic risk posed by MPXV in the area. Additionally, studying the role of MPXV infections in wild nonhuman primates could enhance understanding of the natural history of this virus.
Dr Radonić is a research scientist at the Robert Koch Institute. His primary research focus is the implementation, development, validation, and diagnostic applications of next-generation sequencing approaches.
Acknowledgment
We thank the Ivorian authorities for long-term support, especially the Ministry of the Environment and Forests, as well as the Ministry of Research, the directorship of the Taï National Park and the Swiss Research Center. We also thank Jule Hinzmann, Ines Müller, Jung-Won Sim-Brandenburg, and Delia Barz for their excellent technical assistance and Ursula Erikli for copy editing.
References
- Magnus PV, Andersen EK, Petersen KB, Birch-Andersen A. A pox-like disease in cynomolgus monkeys. Acta Pathol Microbiol Scand. 1959;46:156–76. DOIGoogle Scholar
- Likos AM, Sammons SA, Olson VA, Frace AM, Li Y, Olsen-Rasmussen M, A tale of two clades: monkeypox viruses. J Gen Virol. 2005;86:2661–72 . DOIPubMedGoogle Scholar
- Chen N, Li G, Liszewski MK, Atkinson JP, Jahrling PB, Feng Z, Virulence differences between monkeypox virus isolates from West Africa and the Congo basin. Virology. 2005;340:46–63. DOIPubMedGoogle Scholar
- Parker S, Buller RM. A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Future Virol. 2013;8:129–57.
- Parker S, Nuara A, Buller RM, Schultz DA. Human monkeypox: an emerging zoonotic disease. Future Microbiol. 2007;2:17–34. DOIPubMedGoogle Scholar
- Khodakevich L, Jezek Z, Kinzanzka K. Isolation of monkeypox virus from wild squirrel infected in nature. Lancet. 1986;1:98–9. DOIPubMedGoogle Scholar
- Leendertz FH, Pauli G, Mätz-Rensing K, Boardman W, Nunn C, Ellerbrok H, Pathogens as drivers of population declines: the importance of systematic monitoring in great apes and other threatened mammals. Biol Conserv. 2006;131:325–37. DOIGoogle Scholar
- Kramski M, Mätz-Rensing K, Stahl-Hennig C, Kaup FJ, Nitsche A, Pauli G, A novel highly reproducible and lethal nonhuman primate model for orthopox virus infection. PLoS ONE. 2010;5:e10412. DOIPubMedGoogle Scholar
- Nakazawa Y, Emerson GL, Carroll DS, Zhao H, Li Y, Reynolds MG, Phylogenetic and ecologic perspectives of a monkeypox outbreak, southern Sudan, 2005. Emerg Infect Dis. 2013;19:237–45. DOIPubMedGoogle Scholar
- Alcamí A, Smith GL. A soluble receptor for interleukin-1 beta encoded by vaccinia virus: a novel mechanism of virus modulation of the host response to infection. Cell. 1992;71:153–67. DOIPubMedGoogle Scholar
- Weaver JR, Isaacs SN. Monkeypox virus and insights into its immunomodulatory proteins. Immunol Rev. 2008;225:96–113. DOIPubMedGoogle Scholar
- Calvignac-Spencer S, Leendertz SA, Gillespie TR, Leendertz FH. Wild great apes as sentinels and sources of infectious disease. Clin Microbiol Infect. 2012;18:521–7. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 20, Number 6—June 2014
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Andreas Nitsche, Centre for Biological Threats and Special Pathogens, Highly Pathogenic Viruses, Robert Koch Institute, Nordufer 20, 13353 Berlin, Germany
Top