Volume 20, Number 7—July 2014
Research
Independent Lineages of Highly Sulfadoxine-Resistant Plasmodium falciparum Haplotypes, Eastern Africa
Figure 2
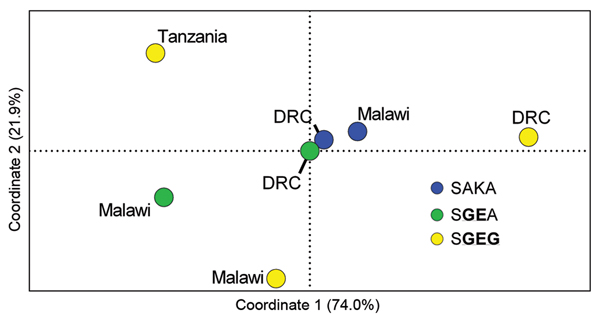
Figure 2. Principal coordinates analyses of wild-type (SAKA) and mutant (SGEA and SGEG) Plasmodium falciparum dihydropteroate synthase (dhps) halotypes from eastern Africa based on analysis of variance (RST)Pairwise RST values were computed with SPAGeDi (26) for microsatellite profiles of 7 populations of parasites defined by dhps haplotype and location: SAKA parasites from Malawi (n = 24) and the Democratic Republic of the Congo (DRC) (n = 53) (blue dots); SGEA parasites from Malawi (n = 67) and DRC (n = 17) (green dots); and SGEG parasites from DRC (n = 5), Malawi (n = 10), and Tanzania (n = 17) (yellow dots)These pairwise values were inputted into principal coordinates analyses in GenAlEx (21), in which coordinates 1 and 2 cumulatively accounted for 96% of the varianceThe dhps haplotypes are defined by amino acids at codons 436, 437, 540, and 581Mutant amino acids are underlined and in bold.
References
- Sridaran S, McClintock SK, Syphard LM, Herman KM, Barnwell JW, Udhayakumar V. Anti-folate drug resistance in Africa: meta-analysis of reported dihydrofolate reductase (dhfr) and dihydropteroate synthase (dhps) mutant genotype frequencies in African Plasmodium falciparum parasite populations. Malar J. 2010;9:247 . DOIPubMedGoogle Scholar
- Aponte JJ, Schellenberg D, Egan A, Breckenridge A, Carneiro I, Critchley J, Efficacy and safety of intermittent preventive treatment with sulfadoxine-pyrimethamine for malaria in African infants: a pooled analysis of six randomised, placebo-controlled trials. Lancet. 2009;374:1533–42 . DOIPubMedGoogle Scholar
- Dicko A, Diallo AI, Tembine I, Dicko Y, Dara N, Sidibe Y, Intermittent preventive treatment of malaria provides substantial protection against malaria in children already protected by an insecticide-treated bednet in Mali: a randomised, double-blind, placebo-controlled trial. PLoS Med. 2011;8:e1000407. DOIPubMedGoogle Scholar
- Konaté AT, Yaro JB, Ouedraogo AZ, Diarra A, Gansane A, Soulama I, Intermittent preventive treatment of malaria provides substantial protection against malaria in children already protected by an insecticide-treated bednet in Burkina Faso: a randomised, double-blind, placebo-controlled trial. PLoS Med. 2011;8:e1000408. DOIPubMedGoogle Scholar
- World Health Organization. WHO policy recommendation on intermittent preventive treatment during infancy with sulfadoxine-pyrimethamine (SP-IPTi) for Plasmodium falciparum malaria control in Africa 2010 [cited 2014 Apr 11]. http://www.who.int/malaria/news/WHO_policy_recommendation_IPTi_032010.pdf
- World Health Organization. WHO policy recommendation: seasonal malaria chemoprevention (SMC) for Plasmodium falciparum malaria control in highly seasonal transmission areas of the Sahel sub-region in Africa 2012 [cited 2014 Apr 11]. http://www.who.int/malaria/publications/atoz/who_smc_policy_recommendation/en/
- World Health Organization. WHO policy brief for the implementation of intermittent preventive treatment of malaria in pregnancy using sulfadoxine-pyrimethamine (IPTp-SP) 2013. [cited 2014 Apr 11]. http://www.who.int/malaria/publications/atoz/Policy_brief_IPTp-SP_implementation_11april2013.pdf
- Feng G, Simpson JA, Chaluluka E, Molyneux ME, Rogerson SJ. Decreasing burden of malaria in pregnancy in Malawian women and its relationship to use of intermittent preventive therapy or bed nets. PLoS ONE. 2010;5:e12012. DOIPubMedGoogle Scholar
- Harrington WE, Mutabingwa TK, Kabyemela E, Fried M, Duffy PE. Intermittent treatment to prevent pregnancy malaria does not confer benefit in an area of widespread drug resistance. Clin Infect Dis. 2011;53:224–30. DOIPubMedGoogle Scholar
- Kublin JG, Dzinjalamala FK, Kamwendo DD, Malkin EM, Cortese JF, Martino LM, Molecular markers for failure of sulfadoxine-pyrimethamine and chlorproguanil-dapsone treatment of Plasmodium falciparum malaria. J Infect Dis. 2002;185:380–8. DOIPubMedGoogle Scholar
- Gosling RD, Gesase S, Mosha JF, Carneiro I, Hashim R, Lemnge M, Protective efficacy and safety of three antimalarial regimens for intermittent preventive treatment for malaria in infants: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;374:1521–32. DOIPubMedGoogle Scholar
- Harrington WE, Mutabingwa TK, Muehlenbachs A, Sorensen B, Bolla MC, Fried M, Competitive facilitation of drug-resistant Plasmodium falciparum malaria parasites in pregnant women who receive preventive treatment. Proc Natl Acad Sci U S A. 2009;106:9027–32. DOIPubMedGoogle Scholar
- Wootton JC, Feng X, Ferdig MT, Cooper RA, Mu J, Baruch DI, Genetic diversity and chloroquine selective sweeps in Plasmodium falciparum. Nature. 2002;418:320–3. DOIPubMedGoogle Scholar
- Roper C, Pearce R, Nair S, Sharp B, Nosten F, Anderson T. Intercontinental spread of pyrimethamine-resistant malaria. Science. 2004;305:1124. DOIPubMedGoogle Scholar
- Pearce RJ, Pota H, Evehe MS. Bâ el-H, Mombo-Ngoma G, Malisa AL, et al. Multiple origins and regional dispersal of resistant dhps in African Plasmodium falciparum malaria. PLoS Med. 2009;6:e1000055.
- Vinayak S, Alam MT, Mixson-Hayden T, McCollum AM, Sem R, Shah NK, Origin and evolution of sulfadoxine resistant Plasmodium falciparum. PLoS Pathog. 2010;6:e1000830 . DOIPubMedGoogle Scholar
- Mita T, Venkatesan M, Ohashi J, Culleton R, Takahashi N, Tsukahara T, Limited geographical origin and global spread of sulfadoxine-resistant dhps alleles in Plasmodium falciparum populations. J Infect Dis. 2011;204:1980–8. DOIPubMedGoogle Scholar
- Taylor SM, Antonia AL, Chaluluka E, Mwapasa V, Feng G, Molyneux ME, Antenatal receipt of sulfadoxine-pyrimethamine does not exacerbate pregnancy-associated malaria despite the expansion of drug-resistant Plasmodium falciparum: clinical outcomes from the QuEERPAM study. Clin Infect Dis. 2012;55:42–50 . DOIPubMedGoogle Scholar
- Taylor SM, Messina JP, Hand CC, Juliano JJ, Muwonga J, Tshefu AK, Molecular malaria epidemiology: mapping and burden estimates for the Democratic Republic of the Congo, 2007. PLoS ONE. 2011;6:e16420. DOIPubMedGoogle Scholar
- Taylor SM, Antonia AL, Parobek CM, Juliano JJ, Janko M, Emch M, Plasmodium falciparum sulfadoxine resistance is geographically and genetically clustered within the DR Congo. Sci Rep. 2013;3:1165.
- Peakall R, Smouse PE. GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research–an update. Bioinformatics. 2012;28:2537–9. DOIPubMedGoogle Scholar
- Excoffier L, Smouse PE, Quattro JM. Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics. 1992;131:479–91 .PubMedGoogle Scholar
- Nei M, Roychoudhury AK. Sampling variances of heterozygosity and genetic distance. Genetics. 1974;76:379–90 .PubMedGoogle Scholar
- Fluxus Engineering. Phylogenetic network software [cited 2012 Oct 19]. http://www.fluxus-engineering.com/sharenet.htm
- Bandelt HJ, Forster P, Rohl A. Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol. 1999;16:37–48 . DOIPubMedGoogle Scholar
- Hardy OJ, Vekemans X. SPAGeDi: a versatile computer program to analyse spatial genetic structure at the individual or population levels. Mol Ecol Notes. 2002;2:618–20. DOIGoogle Scholar
- Felsenstein J. PHYLIP - Phylogeny Inference Package (version 3.2). Cladistics. 1989;5:164–6.
- Mobyle Portal. Institut Pasteur [cited 2013 Aug 27]. http://mobyle.pasteur.fr/cgi-bin/portal.py?#forms:neighbor
- Pritchard JK, Stephens M, Donnelly P. Inference of population structure using multilocus genotype data. Genetics. 2000;155:945–59 .PubMedGoogle Scholar
- Hubisz MJ, Falush D, Stephens M, Pritchard JK. Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour. 2009;9:1322–32.
- Evanno G, Regnaut S, Goudet J. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol. 2005;14:2611–20. DOIPubMedGoogle Scholar
- Taylor SM, Antonia A, Feng G, Mwapasa V, Chaluluka E, Molyneux M, Adaptive evolution and fixation of drug-resistant Plasmodium falciparum genotypes in pregnancy-associated malaria: 9-year results from the QuEERPAM study. Infect Genet Evol. 2012;12:282–90. DOIPubMedGoogle Scholar
- Minja DT, Schmiegelow C, Mmbando B, Bostrom S, Oesterholt M, Magistrado P, Plasmodium falciparum mutant haplotype infection during pregnancy associated with reduced birthweight, Tanzania. Emerg Infect Dis. 2013;19:1446–54. DOIPubMedGoogle Scholar
- Slatkin M. A measure of population subdivision based on microsatellite allele frequencies. Genetics. 1995;139:457–62 .PubMedGoogle Scholar
- Gutman J, Mwandama D, Wiegand RE, Ali D, Mathanga DP, Skarbinski J. Effectiveness of intermittent preventive treatment with sulfadoxine-pyrimethamine during pregnancy on maternal and birth outcomes in machinga district, Malawi. J Infect Dis. 2013;208:907–16. DOIPubMedGoogle Scholar
- McCollum AM, Poe AC, Hamel M, Huber C, Zhou Z, Shi YP, Antifolate resistance in Plasmodium falciparum: multiple origins and identification of novel dhfr alleles. J Infect Dis. 2006;194:189–97. DOIPubMedGoogle Scholar
- Naidoo I, Roper C. Mapping ‘partially resistant’, ‘fully resistant’, and ‘super resistant’ malaria. Trends Parasitol. 2013;29:505–15. DOIPubMedGoogle Scholar
1Current affiliation: Duke University School of Medicine, Durham, North Carolina, USA.