Volume 21, Number 5—May 2015
Research
Novel Thogotovirus Associated with Febrile Illness and Death, United States, 2014
Cite This Article
Citation for Media
Abstract
A previously healthy man from eastern Kansas, USA, sought medical care in late spring because of a history of tick bite, fever, and fatigue. The patient had thrombocytopenia and leukopenia and was given doxycycline for a presumed tickborne illness. His condition did not improve. Multiorgan failure developed, and he died 11 days after illness onset from cardiopulmonary arrest. Molecular and serologic testing results for known tickborne pathogens were negative. However, testing of a specimen for antibodies against Heartland virus by using plaque reduction neutralization indicated the presence of another virus. Next-generation sequencing and phylogenetic analysis identified the virus as a novel member of the genus Thogotovirus.
The genus Thogotovirus (family Orthomyxoviridae) contains >6 distinct viruses, including Araguari, Aransas Bay, Dhori, Jos, Thogoto, and Upolu viruses (1–3). These viruses have been primarily associated with either hard or soft ticks and have a wide geographic distribution (1–8). The only virus in this genus known to occur in the United States is Aransas Bay virus, which was isolated from soft ticks (Ornithodoros spp.) collected from a seabird nest off the coast of Texas (3).
Two viruses in the genus Thogotovirus (Thogoto and Dhori viruses) are currently known to cause human infection and disease. Antibodies against Thogoto virus have been identified in humans living in parts of Europe, Asia, and Africa (1,4,6,8). Two persons from Nigeria infected with this virus were identified in 1966. The first patient was a man with a febrile illness in whom neuromyelitis optica later developed. The second patient was a 14-year-old boy in whom meningitis developed and who died 6 days later because of complications of sickle cell disease (9).
Antibodies against Dhori virus in humans have been reported in a similar distribution as those against Thogoto virus (1,6,8,10). Five patients with disease have been described after accidental laboratory exposure to Dhori virus; encephalitis developed in 2 of these patients (11). We report a novel Thogotovirus associated with a febrile illness and death that occurred in a man in the United States in 2014.
The patient was a previously healthy man >50 years of age from Bourbon County, Kansas, USA. While working outdoors on his property in late spring 2014, the patient had several tick bites and found an engorged tick on his shoulder several days before he became ill with nausea, weakness, and diarrhea. The following day, a fever, anorexia, chills, headache, myalgia, and arthralgia developed. On the third day of illness, the patient went to his primary care physician, who empirically prescribed doxycycline for a presumed tickborne illness because of his history of tick bites, symptoms, and no reported travel outside the immediate area. The following morning, the patient’s wife found him obtunded (experiencing reduced consciousness) but arousable. He was taken by ambulance to a local hospital.
At the hospital, he had a temperature of 37.3°C, a pulse rate of 84 beats/min, and an increased blood pressure of 151/65 mm Hg. The patient had a papular rash on his trunk, but otherwise results of his physical examination were unremarkable. Initial laboratory findings showed leukopenia (2,200 cells/μL), lymphopenia (absolute lymphocyte count 550 cells/μL), thrombocytopenia (72,000 cells/μL), mild hyponatremia (sodium 133 mmol/L), hypokalemia (potassium 3.0 mmol/L), a creatinine level (0.8 mg/dL) within the reference range (0.6 mg/dL–1.2 mg/dL), a slightly increased level of blood urea nitrogen (25 μg/dL), and increased levels of aspartate aminotransferase (138 U/L) and alanine aminotransferase (86 U/L). He was admitted because of the principal problems of dehydration, syncope, and possible tickborne illness. He was given an intravenous (IV) fluid bolus, then maintenance fluids, and doxycycline (200 mg IV every 12 h for the first 24 h, then 100 mg IV every 12 h).
Despite doxycycline therapy, the patient continued to report malaise and anorexia, and periodic fevers (maximum temperature 38.8°C) developed. At day 8 postillness onset, the patient was transferred to a tertiary care center for further evaluation and management. Patient samples collected before transfer showed no serologic evidence of Rocky Mountain spotted fever, Lyme disease, or ehrlichiosis.
At initial assessment at the tertiary care center, the patient was febrile (temperature 39.4°C) and had a nontender left axillary lymphadenopathy; a diffuse maculopapular rash on his chest, abdomen, and back; petechiae on his soft palate and lower extremities; and bibasilar crackles in the lung fields. Laboratory testing continued to show mild leukopenia (3,600 cells/μL) but also showed worsening thrombocytopenia (34,000 cells/μL). His renal function was normal, but his aspartate aminotransferase level had increased to 119 U/L. Doxycycline treatment (100 mg IV every 12 h) was continued, and the patient was evaluated further for a potential etiology of his illness.
Hematologic results suggested that his persistent thrombocytopenia and leukopenia were secondary to acute bone marrow suppression. A chest, abdomen, and pelvis computed tomography scan with contrast showed trace pleural effusions, bibasilar atelectasis, and multiple prominent abdominal lymph nodes. At day 9 postillness onset, he remained lucid and interactive, but he continued to have episodic high fever (temperature >39°C) and progressive dyspnea developed, which resulted in a need for supplemental oxygen. A chest radiograph showed new findings of pulmonary venous congestion and interstitial edema, suggestive of progressive heart failure or fluid overload, and an echocardiogram showed global hypokinesis.
Because of increasing supplemental oxygen needs and progressive lactic acidosis, he was transferred to the intensive care unit and given broad-spectrum antimicrobial drugs on day 10 of his illness. His renal function began to deteriorate and his aminotransferase levels continued to increase. The patient was intubated because of acute respiratory distress syndrome and was given 3 vasopressor medications because of shock. The patient subsequently had sustained ventricular tachycardia with persistent hypotension and eventual pulseless electrical activity with refractory shock. After multiple resuscitations, the decision was made to withdraw further care, and he died shortly after being extubated, 11 days after first becoming ill. An autopsy was not performed.
Results of comprehensive evaluations for tickborne diseases, including serologic testing for Rocky Mountain spotted fever, tularemia, brucella, babesiosis, and Q fever; molecular testing for Ehrlichia spp. and Anaplasma phagocytophilum; and blood thin smears for Babesia spp. were negative. Results of evaluations for fungal pathogens (Aspergillus spp. galactomannan, antibodies against Histoplasma spp., and Histoplasma spp. antigen in serum and urine) were negative. Evaluations for cytomegalovirus, Epstein-Barr virus, and parvovirus showed past infection. Test results for hepatitis B and C viruses, West Nile virus, and HIV were also negative. Blood, sputum, and urine bacterial cultures were negative. A whole blood specimen collected 9 days after illness onset was sent to the Centers for Disease Control and Prevention (CDC) (Fort Collins, CO, USA) for Heartland virus testing as part of an active institutional review board-approved protocol.
Clinical Specimen Handling and Evaluation
At CDC, EDTA-treated blood, along with serum separated from that blood, were tested for Heartland viral RNA and neutralizing antibodies by real-time reverse transcription PCR (RT-PCR) and plaque reduction neutralization test (PRNT) with 6-well plates with confluent Vero E6 monolayers, according to protocols described elsewhere (12,13). Standard virus isolation methods were also used. In brief, 200 μL of undiluted and 1:10 dilutions of blood or serum specimens were inoculated onto confluent Vero cells in T25 flasks. Inoculated flasks were then incubated at 37°C and reviewed for cytopathic effect daily.
Viral Genome Sequencing, RT-PCR, and Phylogenetic Analyses
Supernatants collected from standard virus isolation cell cultures were subjected to next-generation sequencing (NGS) methods by using the Ion Torrent PGM sequencer (Life Technologies, Grand Island, NY, USA) and methods as described (14). After novel viral sequences were identified by NGS, a real-time RT-PCR was designed to target the newly derived sequences and applied to blood and serum samples by using methods described (15). Phylogenetic analyses were conducted on deduced amino acid sequences from multiple genomic segments of selected viruses of the same viral family by using MEGA 5.05 software (http://www.megasoftware.net/) as described (16).
Isolation and Identification of Virus in Blood and Serum
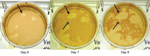
Blood and serum showed negative results for Heartland viral RNA and antibodies against this virus. However, heterologous viral (non–Heartland viral) plaques were noted in PRNT cell culture wells, which indicated the presence of another virus (Figure 1). Standard virus isolation methods showed a substantial cytopathic effect at day 3 postinoculation in cells that were inoculated with blood or serum specimens. These findings were confirmed by repeated isolation attempts.
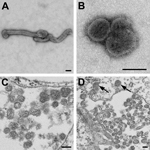
Negative stain and thin-section electron microscopy showed pleomorphic viral particles consistent with viruses in the family Orthomyxoviridae (Figure 2). NGS methods applied to cell culture supernatants from multiple isolations showed the presence of novel orthomyxoviral RNA. We observed ≈70% overall average nucleotide sequence percentage identity with Dhori virus in multiple genomic segments. Blood and serum samples were verified as the source of the novel virus by real-time RT-PCR–based detection of viral RNA in these samples.
Phylogenetic Analyses
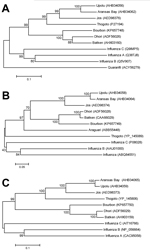
Three phylogenies, each generated by a neighbor-joining method applied with 2,000 bootstrap replicates for grouping analysis, were chosen as representative of overall genetic relationships of selected viruses (Figure 3). The novel virus was found to group with strong support along with Dhori virus, and the closely related Batken virus, in all trees.
Using traditional techniques (i.e., PRNT and culturing on animal cells) in combination with NGS, we isolated a novel virus from a blood sample collected 9 days after illness onset from a previously healthy man. It is likely that this novel Thogotovirus, which we are proposing to call Bourbon virus after the county of residence of the patient, was the cause of his illness. Although it is unclear what role the virus played in the death of the patient, the high level of viremia, as shown by multiple isolations from the blood of the patient 2 days before his death, suggests that this might have contributed to the death of the patient.
The patient had a history of tick exposure, as well as symptoms and laboratory findings (i.e., leukopenia and thrombocytopenia) consistent with a tickborne illness. Several tickborne pathogens, such as Ehrlichia chaffensis, Rickettsia, and Heartland virus, are present in eastern Kansas and adjacent areas (17–19). However, the patient did not respond to doxycycline therapy initiated 3 days after illness onset and had negative results for these and other tickborne pathogens.
Of the 7 symptomatic human infections that have been associated with viruses in the genus Thogotovirus, most case-patients have had neurologic findings (e.g., meningitis, encephalitis) without any described abnormalities in blood counts (9,11). Although cerebrospinal fluid was not tested for the patient reported, his clinical signs and symptoms were not suggestive of neurologic infection. Furthermore, the patient did not have any respiratory symptoms that would be expected with other viruses that are known human pathogens in the large family of Orthomyxoviridae, such as influenza virus (1).
Phylogenetic analyses indicated that Bourbon virus is most closely related to Dhori and Batken viruses. However, the branch lengths suggest a relatively distant evolutionary distinction of Bourbon virus from Dhori and Batken viruses, which have only been described in the Eastern Hemisphere. Dhori, Batken, and Thogoto viruses have been identified in various hard tick species (1). However, Batken virus also has been identified in mosquitoes (1). It is currently unknown how Bourbon virus is transmitted to humans. However, illness onset of the patient in late spring and a history of finding an embedded tick before becoming ill support the notion that Bourbon virus might be transmitted by ticks. Therefore, to potentially prevent Bourbon virus disease, as well as other tickborne diseases, persons should be advised to avoid tick bites by using an insect repellent registered with the US Environmental Protection Agency to be effective against ticks, wearing long sleeves and pants, avoiding bushy and wooded areas, and performing tick checks after spending time outdoors.
The discovery of Bourbon virus, in addition to recent discoveries of tick-associated Heartland and severe fever with thrombocytopenia syndrome viruses (19,20), suggests that the public health burden of these pathogens has been underestimated. As nonselective molecular methods of pathogen identification (i.e., NGS sequencing) become more widely used, ideally in combination with classical microbiologic techniques, it is anticipated that similar discoveries will be made in the future.
It is currently not known how many human infections and disease cases might be attributable to this novel pathogen. On the basis of limited information for our case-patient, health care providers might consider Bourbon virus as a potential infectious etiology in patients in whom fever, leukopenia, and thrombocytopenia develop without a more likely explanation and who have shown negative results for other tickborne diseases (e.g., ehrlichiosis, anaplasmosis, or Heartland virus disease) or have not responded to doxycycline therapy. Work is planned to identify additional human infections with this novel virus, as well as to explore its potential geographic distribution. Also, more comprehensive virologic characterizations and field work are ongoing to better understand the biology of, and to identify potential vectors and reservoirs for, Bourbon virus. These data will be critical to further characterize the epidemiology and illness caused by Bourbon virus and to implement potential prevention and control measures.
Ms. Kosoy is a microbiologist at the Arboviral Diseases Branch, Division of Vector-Borne Diseases, Centers for Disease Control and Prevention, Fort Collins, Colorado. Her research interests are diagnosis of arboviral diseases and biology of arboviruses.
Acknowledgment
We thank Jason Velez for preparing cells; Kristen Burkhalter, Jan Pohl, Roger Nasci, Nicky Sulaiman, and Emily Pau for facilitation of development of the rapid Bourbon real-time RT-PCR; Amanda Panella for assistance with handling the samples; and Robert S. Lanciotti, Ingrid Rabe, and Marc Fischer for their input on diagnostic evaluation, and review of the manuscript.
References
- McCauley JW, Hongo S, Kaverin NV, Kochs G, Lamb RA, Matrosovich MN, Family Orthomyxoviridae. In: King AM, Adams MJ, Carstens EB, Lefkowitz EJ, editors. Virus taxonomy: classification and nomenclature of viruses. Ninth report of the International Committee of Taxonomy of Viruses. New York: Elsevier Inc.; 2012. p. 749–61.
- Briese T, Chowdhary R, Travassos da Rosa A, Hutchison SK, Popov V, Street C, Upolu virus and Aransas Bay virus, two presumptive bunyaviruses, are novel members of the family Orthomyxoviridae. J Virol. 2014;88:5298–309 . DOIPubMedGoogle Scholar
- Bussetti AV, Palacios G, Travassos da Rosa A, Savji N, Jain K, Guzman H, Genomic and antigenic characterization of Jos virus. J Gen Virol. 2012;93:293–8. DOIPubMedGoogle Scholar
- Kuno G, Chang GJ, Tsuchiya KR, Miller BR. Phylogeny of Thogoto virus. Virus Genes. 2001;23:211–4. DOIPubMedGoogle Scholar
- Da Silva EV, Da Rosa AP, Nunes MR, Diniz JA, Tesh RB, Cruz AC, Araguari virus, a new member of the family Orthomyxoviridae: serologic, ultrastructural, and molecular characterization. Am J Trop Med Hyg. 2005;73:1050–8 .PubMedGoogle Scholar
- Filipe AR, Calisher CH, Lazuick J. Antibodies to Congo-Crimean haemorrhagic fever, Dhori, Thogoto and Bhanja viruses in southern Portugal. Acta Virol. 1985;29:324–8 .PubMedGoogle Scholar
- Sang R, Onyango C, Gachoya J, Mabinda E, Konongoi S, Ofula V, Tickborne arbovirus surveillance in market livestock, Nairobi, Kenya. Emerg Infect Dis. 2006;12:1074–80. DOIPubMedGoogle Scholar
- Hubálek Z, Rudolf I. Tick-borne viruses in Europe. Parasitol Res. 2012;111:9–36. DOIPubMedGoogle Scholar
- Moore DL, Causey OR, Carey DE, Reddy S, Cooke AR, Akinkugbe FM, Arthropod-borne viral infections of man in Nigeria, 1964–1970. Ann Trop Med Parasitol. 1975;69:49–64 .PubMedGoogle Scholar
- Sokhey J, Dandawate CN, Gogate SS, Ghosh SN, Gupta NP. Serological studies on Dhori virus. Indian J Med Res. 1977;66:726–31 .PubMedGoogle Scholar
- Butenko AM, Leshchinskaia EV, Semashko IV, Donets MA, Mart’ianova LI. Dhori virus—a causative agent of human disease. 5 cases of laboratory infection [in Russian]. Vopr Virusol. 1987;32:724–9 .PubMedGoogle Scholar
- Muehlenbachs A, Fata C, Lambert AJ, Paddock CD, Velez JO, Blau DM, Heartland virus–associated death in Tennessee. Clin Infect Dis. 2014;59:845–50. DOIPubMedGoogle Scholar
- Beaty BJ, Calisher CH, Shope RE. Arboviruses. In: Lennette EH, Lennette DA, Lennette ET, editors. Diagnostic procedures for viral, rickettsial and chlamydial Infections. 7th ed. Washington (DC): American Public Health Association; 1995. p. 189–212.
- Huhtamo E, Lambert AJ, Costantino S, Servino L, Krizmancic L, Boldorini R, Isolation and full genomic characterization of Batai virus from mosquitoes, Italy 2009. J Gen Virol. 2013;94:1242–8. DOIPubMedGoogle Scholar
- Lambert AJ, Martin DA, Lanciotti RS. Detection of North American eastern and western equine encephalitis viruses by nucleic acid amplification assays. J Clin Microbiol. 2003;41:379–85. DOIPubMedGoogle Scholar
- Lambert AJ, Lanciotti RS. Molecular characterization of medically important viruses of the genus Orthobunyavirus. J Gen Virol. 2008;89:2580–5. DOIPubMedGoogle Scholar
- Chapman AS, Murphy SM, Demma LJ, Holman RC, Curns AT, McQuiston JH, Rocky Mountain spotted fever in the United States, 1997–2002. Vector Borne Zoonotic Dis. 2006;6:170–8. DOIPubMedGoogle Scholar
- Pastula DM, Turabelidze G, Yates KF, Jones TF, Lambert AJ, Panella AJ, Notes from the field: Heartland virus disease—United States, 2012–2013. MMWR Morb Mortal Wkly Rep. 2014;63:270–1 .PubMedGoogle Scholar
- McMullan LK, Folk SM, Kelly AJ, MacNeil A, Goldsmith CS, Metcalfe MG, A new phlebovirus associated with severe febrile illness in Missouri. N Engl J Med. 2012;367:834–41. DOIPubMedGoogle Scholar
- Yu XJ, Liang MF, Zhang SY, Liu Y, Li JD, Sun YL, Fever with thrombocytopenia associated with a novel bunyavirus in China. N Engl J Med. 2011;364:1523–32. DOIPubMedGoogle Scholar
Figures
Cite This ArticleTable of Contents – Volume 21, Number 5—May 2015
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
J. Erin Staples, Arboviral Diseases Branch, Division of Vector-Borne Diseases, Centers for Disease Control and Prevention, 3156 Rampart Rd, Mailstop P02, Fort Collins, CO 80521, USA
Top