Volume 22, Number 1—January 2016
Dispatch
Increase in Sexually Transmitted Infections among Men Who Have Sex with Men, England, 2014
Cite This Article
Citation for Media
Abstract
Surveillance data from sexual health clinics indicate recent increases in sexually transmitted infections, particularly among men who have sex with men. The largest annual increase in syphilis diagnoses in a decade was reported in 2014. Less condom use may be the primary reason for these increases.
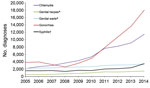
Sexually transmitted infections (STIs) are a major public health concern; they can facilitate the transmission of HIV and are associated with severe disease. Treatment for some STIs, especially gonorrhea, has been compromised by antimicrobial drug resistance (1). In 2014 in England, there were 439,243 diagnoses of STIs. Although this number reflects a very small decline (0.3%) relative to 2013, numbers of diagnoses of syphilis and gonorrhea rose substantially, by 33% (from 3,236 to 4,317) and 19% (from 29,419 to 34,958), respectively (2). This number of syphilis diagnoses is the highest reported in England since 1949, and the number of gonorrhea diagnoses is the highest reported since 1986. These increases resulted almost entirely from increased diagnoses among men who have sex with men (MSM), among whom diagnoses of syphilis and gonorrhea increased 46% (from 2,375 to 3,477) and 32% (from 13,629 to 18,029), respectively (Figure 1), resulting in the highest number of diagnoses of these STIs since reporting among MSM began in 1994. We explored the epidemiology of these and other STIs among MSM and describe recent trends.
In England, surveillance for STIs is conducted through mandatory reporting in sexual health clinics (SHCs) (genitourinary medicine [GUM] and integrated GUM/sexual and reproductive health clinics) by using the GUM clinic activity dataset version 2 (GUMCADv2) (3). SHCs provide free and open access services and, since 2012, all (216 in 2014) have submitted data to Public Health England. Although STIs are not notifiable diseases in the United Kingdom, GUMCADv2 is a comprehensive, patient-level dataset of all attendances and laboratory-confirmed STIs at SHCs. Information about the sexual orientation of each attendee is collected through GUMCADv2 (completion >90% since 2011); this information was also collected by the preceding system, the KC60 aggregate return (3). We reviewed the most recent GUMCADv2 data, extracted on April 28, 2015, to assess trends in laboratory-confirmed gonorrhea, infectious (primary/secondary/early latent) syphilis, chlamydia, genital herpes (first episode), and genital warts (first episode). Only 1 diagnosis of each STI was counted within a 6-week period; this restriction also applies to instances of infection at multiple anatomic sites (3). These data represent the number of diagnoses reported, not the number of persons in whom the infections were diagnosed. Descriptive analyses by demographic characteristics and tests for linear trend were performed; p values <5% were considered significant.
Since 2013, syphilis diagnoses among MSM increased by 46% (from 2,375 to 3,477); this increase is the largest year-on-year increase in syphilis diagnoses among MSM since 2005 (Figure 1; Table 1). Relative to 2013, in 2014 the number of syphilis diagnoses among heterosexual men and men of unknown sexual orientation decreased by 0.2% (from 578 to 577), with a larger decrease in women (7.1%, from 283 to 263) (Table 1).
From 2013 to 2014, diagnoses of gonorrhea among MSM increased 32% (from 13,629 to 18,029) (Figure 1; Table 1). Although this increase was relatively large, it is consistent with the increasing trend from 2005 (3,817 diagnoses; p = 0.015). Compared with the increased diagnoses in MSM since 2013, in 2014 the increases in gonorrhea diagnoses among heterosexual men and men of unknown sexual orientation (5.2%, from 8,122 to 8,546) and women (9.3%, from 7,664 to 8,379) were smaller (Table 1).
Relative to 2013, in 2014, diagnoses further increased for chlamydia (25.8%), genital herpes (10.1%), and genital warts (9.5%) among MSM, consistent with the increasing trend since 2005 (Figure 1; Table 1). Diagnoses of chlamydia decreased among heterosexual men and men of unknown sexual orientation (0.4%, from 44,512 to 44,339) and increased among women (4.9%, from 48,642 to 51,045). Genital herpes diagnoses decreased by 4.8% (from 10,938 to 10,415) and 0.9% (from 20,069 to 19,883) and genital warts by 5.2% (from 37,872 to 35,893) and 4.8% (from 32,834 to 31,251) among heterosexual men and men of unknown sexual orientation and among women, respectively.
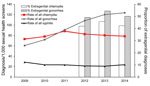
Although the number of sexual health screen (tests for chlamydia, gonorrhea, HIV, and syphilis) among MSM increased 29% from 2013 to 2014, the rates (diagnoses/1,000 screenings) of syphilis and gonorrhea increased 13.8% (from 20.9 to 23.8) and 2.9% (from 120.2 to 123.6), respectively. The rate (diagnosis/1,000 screenings) of chlamydia decreased 2.2% (from 80.4 to 78.6) (Figure 2), and the proportion of diagnoses of extragenital chlamydia and gonorrhea also decreased (Figure 2).
In 2014, the median age for MSM in whom these STIs were diagnosed ranged from 28 years (genital warts) to 36 years (syphilis) (Table 2). More than three quarters of MSM in whom STIs were diagnosed were white, 60.3%–71.4% were born in the United Kingdom, and 12.6%–18.4% were born in other European countries (Table 2). MSM in whom bacterial STIs were diagnosed were more likely to be HIV positive and live in London than were MSM in whom genital warts and herpes were diagnosed (Table 2).
We report the continuing increase of diagnoses of all STIs among MSM, particularly for syphilis, for which the largest number of cases was recorded since 1994. Previously reported trends (4) markedly worsened in 2014. Similar levels of syphilis diagnoses among men (reporting for MSM began in 1994) were last reported in the late 1970s and were followed by a precipitous decline after the emergence of HIV in the United Kingdom in the 1980s (5). In 2014, most MSM in whom STIs were diagnosed lived in London, and an average of 16% were born in Europe outside the United Kingdom. Given such a mobile population, the potential for spread of these infections to MSM in other major cities is clear (6).
Diagnoses of STIs among HIV-positive MSM since 2009 have steadily increased; the STI rate is 2–4 times that among MSM who are HIV negative or of unknown HIV status (7). Sex without condom use, associated with increasing HIV seroadaptive behaviors (i.e., seeking out partners according to their HIV serostatus for unprotected sex) and use of geospatial social networking applications, may facilitate STI transmission in concentrated sexual networks (8,9). Given the risk for emergence of strains of Neisseria gonorrhoeae that are resistant to first-line antimicrobial drugs (1), the increase in gonorrhea diagnoses, especially among MSM, is concerning.
In response to gonorrhea testing guidance published in 2010 (10), use of highly sensitive nucleic acid amplification tests for screening of extragenital sites in MSM has occurred more frequently (11); this change may account for part of the increase in gonorrhea diagnoses in the earlier part of the decade. However, the most recent update contained no changes in syphilis testing guidelines (12), so recent increases are unlikely to result from changes in testing practice. Further, although more MSM were tested for STIs in 2014 compared with 2013, the rate of chlamydia and gonorrhea diagnoses remained relatively stable, while that of syphilis increased.
Given the comprehensive coverage of national STI surveillance and that SHCs in England are open access and free, most syphilis and gonorrhea cases are probably captured in GUMCADv2 (13). A key limitation of GUMCADv2 is that it does not collect any behavioral data; however, an enhancement to collect data on behavior, including recreational drug use and unprotected anal intercourse, is being piloted (https://www.gov.uk/guidance/genitourinary-medicine-clinic-activity-dataset-gumcadv3-pilot).
A focus on biomedical interventions, such as preexposure prophylaxis, for the control of HIV among MSM may have unintended consequences for transmission of other STIs, which highlights a need to ensure that robust STI prevention and control measures are in place (14). These measures should include promoting condom use; increased screening (in the United Kingdom, quarterly HIV/STI testing of MSM who engage in condomless sex with new partners is recommended); ensuring that services are easily accessible; and promoting other risk reduction strategies to improve the health and well-being of MSM (15).
Dr. Mohammed is a principal STI surveillance scientist at Public Health England. His team manages the national surveillance system for STIs in England, and he is the lead on a pilot of enhanced surveillance for behavioral and partner notification outcomes at sexual health clinics.
References
- Ison CA, Town K, Obi C, Chisholm S, Hughes G, Livermore DM, Decreased susceptibility to cephalosporins among gonococci: data from the Gonococcal Resistance to Antimicrobials Surveillance Programme (GRASP) in England and Wales, 2007–2011. Lancet Infect Dis. 2013;13:762–8. DOIPubMedGoogle Scholar
- Public Health England. Sexually transmitted infections and chlamydia screening in England, 2014 [cited 2015 June 23]. https://www.gov.uk/government/statistics/sexually-transmitted-infections-stis-annual-data-tables
- Savage EJ, Mohammed H, Leong G, Duffell S, Hughes G. Improving surveillance of sexually transmitted infections using mandatory electronic clinical reporting: the genitourinary medicine clinic activity dataset, England, 2009 to 2013. Euro Surveill. 2014;19:20981. DOIPubMedGoogle Scholar
- Savage EJ, Marsh K, Duffell S, Ison CA, Zaman A, Hughes G. Rapid increase in gonorrhoea and syphilis diagnoses in England in 2011. Euro Surveill. 2012;17:20224.PubMedGoogle Scholar
- Doherty L, Fenton KA, Jones J, Paine TC, Higgins SP, Williams D, Syphilis: old problem, new strategy. BMJ. 2002;325:153–6. DOIPubMedGoogle Scholar
- Vanden Berghe W, Nöstlinger C, Hospers H, Laga M. International mobility, sexual behaviour and HIV-related characteristics of men who have sex with men residing in Belgium. BMC Public Health. 2013;13:968. DOIPubMedGoogle Scholar
- Malek R, Mitchell H, Furegato M, Simms I, Mohammed H, Nardone A, Contribution of transmission in HIV-positive men who have sex with men to evolving epidemics of sexually transmitted infections in England: an analysis using multiple data sources, 2009–2013. Euro Surveill. 2015;20:21093. DOIPubMedGoogle Scholar
- Aghaizu A, Nardone A, Copas A, Mercey D, Wayal S, Parsons V, Understanding continuing high HIV incidence: sexual behavioural trends among MSM in London, 2000–2013. Sex Transm Infect. 2015;91(Suppl 1):A2.
- Gilbart VL, Simms I, Jenkins C, Furegato M, Gobin M, Oliver I, Sex, drugs and smart phone applications: findings from semistructured interviews with men who have sex with men diagnosed with Shigella flexneri 3a in England and Wales. Sex Transm Infect. 2015 April 28 [Epub ahead of print]. PMID: 25921020
- Bignell C, Fitzgerald M; Guideline Development Group, British Association for Sexual Health and HIV UK. UK national guideline for the management of gonorrhoea in adults, 2011. Int J STD AIDS. 2011;22:541–7.unknown DOIPubMedGoogle Scholar
- Kingston M, French P, Goh B, Goold P, Higgins S, Sukthankar A, UK national guidelines on the management of syphilis 2008. Int J STD AIDS. 2008;19:729–40. DOIPubMedGoogle Scholar
- Wetten S, Mohammed H, Yung M, Mercer CH, Cassell JA, Hughes G. Diagnosis and treatment of chlamydia and gonorrhoea in general practice in England 2000–2011: a population-based study using data from the UK Clinical Practice Research Datalink. BMJ Open. 2015;5:e007776. DOIPubMedGoogle Scholar
- Nardone A, Semaille C. We need to improve screening for sexually transmitted infections in men who have sex with men in Europe. Sex Transm Infect. 2013;89:539–40. DOIPubMedGoogle Scholar
- Public Health England. Promoting the health and wellbeing of gay, bisexual and other men who have sex with men: action plan [cited 2015 Jun 4]. https://www.gov.uk/government/publications/promoting-the-health-and-wellbeing-of-gay-bisexual-and-other-men-who-have-sex-with-men
Figures
Tables
Cite This ArticleTable of Contents – Volume 22, Number 1—January 2016
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Hamish Mohammed, Department of HIV and STIs, Public Health England, 61 Colindale Ave, London, NW9 5EQ, UK
Top