Volume 22, Number 9—September 2016
CME ACTIVITY - Research
Fluoroquinolone-Resistant and Extended-Spectrum β-Lactamase–Producing Escherichia coli Infections in Patients with Pyelonephritis, United States1
Cite This Article
Citation for Media
Introduction

Medscape CME Activity
Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented through the joint providership of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.00 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at http://www.medscape.org/journal/eid; and (4) view/print certificate. For CME questions, see this page.
Release date: August 12, 2016; Expiration date: August 12, 2017
Learning Objectives
Upon completion of this activity, participants will be able to:
• Assess recommendations regarding the treatment of pyelonephritis
• Distinguish the rates of fluoroquinolone resistance and extended-spectrum β-lactamase (ESBL) production among patients with pyelonephritis in the current study
• Evaluate risk factors for fluoroquinolone resistance in the current study
• Evaluate risk factors for ESBL production in the current study
CME Editor
Rhonda Ray, PhD, Copyeditor, Emerging Infectious Diseases. Disclosure: Rhonda Ray, PhD, has disclosed no relevant financial relationships.
CME Author
Charles P. Vega, MD, Clinical Professor of Family Medicine, University of California, Irvine. Disclosure: Charles P. Vega, MD, has disclosed the following financial relationships: served as an advisor or consultant for Allergan, Inc.; McNeil Consumer Healthcare; served as a speaker or a member of a speakers bureau for Shire Pharmaceuticals.
Authors
Disclosures: David A. Talan, MD, has disclosed the following relevant financial relationships: served as an advisor or consultant for Allergan, Inc.; Cempra; served as a speaker or a member of a speakers bureau for Allergan, Inc.; Merck; received grants for clinical research from Merck. Sukhjit S. Takhar, MD; Anusha Krishnadasan, PhD; and William R. Mower, MD, PhD, have disclosed no relevant financial relationships. Fredrick M. Abrahamian, DO, has disclosed the following relevant financial relationships: served as an advisor or consultant for Grifols; Cempra; Summitt Therapeutics; Tetraphase Pharmaceutics; Janssen; Seqirus; served as a speaker or a member of a speakers bureau for Merck; Actavis; Medicines Company; received grants for clinical research from Merck; Cempra; owns stock, stock options, or bonds from Gilead. Gregory J. Moran, MD, has disclosed the following relevant financial relationships: received grants for clinical research from Cempra; Allergan.
Abstract
For 2013–2014, we prospectively identified US adults with flank pain, temperature >38.0°C, and a diagnosis of acute pyelonephritis, confirmed by culture. Cultures from 453 (86.9%) of 521 patients grew Escherichia coli. Among E. coli isolates from 272 patients with uncomplicated pyelonephritis and 181 with complicated pyelonephritis, prevalence of fluoroquinolone resistance across study sites was 6.3% (range by site 0.0%–23.1%) and 19.9% (0.0%–50.0%), respectively; prevalence of extended-spectrum β-lactamase (ESBL) production was 2.6% (0.0%–8.3%) and 12.2% (0.0%–17.2%), respectively. Ten (34.5%) of 29 patients with ESBL infection reported no exposure to antimicrobial drugs, healthcare, or travel. Of the 29 patients with ESBL infection and 53 with fluoroquinolone-resistant infection, 22 (75.9%) and 24 (45.3%), respectively, were initially treated with in vitro inactive antimicrobial drugs. Prevalence of fluoroquinolone resistance exceeds treatment guideline thresholds for alternative antimicrobial drug strategies, and community-acquired ESBL-producing E. coli infection has emerged in some US communities.
Escherichia coli, the predominant cause of community-acquired urinary tract infection (UTI) worldwide, is increasingly resistant to available antimicrobial drugs. In the United States, in vitro resistance of E. coli to trimethoprim/sulfamethoxazole (TMP/SMX) became prevalent in the 1990s (1). Over the past decade, fluoroquinolone resistance rates have increased to >10% in some surveys (2,3).
In many parts of the world, E. coli fluoroquinolone resistance rates are >20% among patients with community-acquired uncomplicated UTI and >50% among patients with complicated infections (4). In addition, infections resulting from extended-spectrum β-lactamase (ESBL)–producing E. coli and other Enterobacteriaceae are becoming increasingly common in these same areas and are associated with sequence type (ST) 131, a globally disseminated, multidrug-resistant clone that frequently produces CTX-M-15 ESBL. These E. coli isolates are generally resistant to cephalosporins and often to other antimicrobial drug classes. In North America, ESBL-producing E. coli infections have occurred predominantly in patients with healthcare exposure and have not become prevalent as a cause of community-acquired infections (5–8).
The 2010 international treatment guidelines of the Infectious Disease Society of America (IDSA) recommend for acute uncomplicated pyelonephritis a fluoroquinolone and an initial dose of an agent from another antimicrobial drug class (e.g., ceftriaxone or gentamicin) if the fluoroquinolone resistance rate is >10% (9). For uncomplicated cystitis, the guidelines discourage use of an antimicrobial drug if its resistance rate is >20%. The guidelines do not address a scenario in which ESBL-producing uropathogens have become prevalent among patients with community-acquired infections. Use of antimicrobial drugs for which the uropathogen shows in vitro resistance has been associated with substantially reduced response rates (1,10,11), which can lead to serious consequences, particularly for patients with pyelonephritis. Given rapid changes in global resistance patterns and a lack of recent active and prospective surveillance of community-acquired UTI in the United States, the extent to which the prevalence of fluoroquinolone resistance has increased and multidrug-resistant ESBL-producing strains have emerged in the community is unknown.
We sought to determine the prevalence of E. coli antimicrobial resistance among patients with acute pyelonephritis who sought care at a US emergency department (ED)–based sentinel research network. We focused on fluoroquinolone-resistant and ESBL-producing isolates from these patients and examined risk factors for antimicrobial drug resistance.
Participants
We recruited adults seeking care in EMERGEncy ID NET, a network of 10 university-affiliated urban US EDs (12). All 10 study sites (Technical Appendix) provided institutional review board approval.
We enrolled patients >18 years of age who sought care during July 2013–December 2014 and had flank pain or costovertebral tenderness; temperature >38.0°C (100.4°F) measured by any method (i.e., oral, rectal, or axillary); and a presumptive diagnosis of acute pyelonephritis (i.e., patient received treatment for this infection during ED visit or was prescribed treatment at discharge). All sites conducted an audit to compare characteristics of enrolled and nonenrolled eligible patients to estimate case-finding sensitivity and detect enrollment biases (online Technical Appendix).
Design and Measurements
We conducted a cross-sectional study by using a convenience sample of prospectively identified patients. ED physicians or study coordinators who used standardized forms at the time of care collected the following: demographic characteristics (i.e., age, sex, race, ethnicity); symptom duration; urinary tract abnormalities; UTI within the previous year; concurrent and immunocompromising conditions; antimicrobial drug use within the previous 2 and 60 days; a fluoroquinolone- or ceftriaxone-resistant UTI within the previous 90 days and 1 year; long-term care residence; hospitalization; travel outside North America within the previous 90 days; illness severity; disposition; and treatments provided. The study population consisted of patients with urine specimens that grew a single uropathogen at >104 CFU/mL. We defined a uropathogen as an organism known to be associated with UTI; among possible pathogens, we found E. coli, Klebsiella, Proteus, Pseudomonas, Citrobacter, Enterobacter, and Salmonella species; Staphylococcus aureus or S. saprophyticus; and Enterococcus and Aerococcus species. If a urine specimen grew >1 organism, we considered it to be contaminated and excluded it. We also excluded specimens that grew Lactobacillus, non-saprophyticus coagulase-negative Staphylococcus, or Corynebacteria species; or α- or γ-hemolytic streptococci.
Patients’ urine specimens were collected in sterile containers by mid-stream clean-catch technique (91.4%), urethral catheterization (5.8%), and other techniques (e.g., sample from collection bag [2.7%] or suprapubic aspirate [0.2%]). Laboratories determined MICs by using automated susceptibility testing with VITEK commercial panels (bioMérieux, Marcy l’Etoile, France) at 7 sites; Microscan (Dade Behring, Inc., Sacramento, CA, USA) at 2 sites; and Phoenix Instrument System (Becton Dickinson, Franklin Lakes, NJ, USA) at 1 site, according to manufacturer instructions.
Each site used E. coli antimicrobial drug resistance breakpoints based on MIC breakpoints (µg/mL) of the Clinical and Laboratory Standards Institute (Wayne, PA, USA) as follows: ampicillin >32; TMP/SMX >4/76; cefazolin >8; ceftriaxone >4; cefotaxime >4; ciprofloxacin >4; levofloxacin >8; ertapenem >2; and imipenem >4 (13). We report resistance rates for antimicrobial drugs included in the study site laboratories’ standard susceptibility testing panel. In 2010, the Clinical and Laboratory Standards Institute changed breakpoints for cephalosporins, aztreonam, and carbapenems. We provided Etests (bioMérieux) for ceftriaxone and imipenem to sites that had not yet updated automated susceptibility testing systems to enable these sites to use the new breakpoints. We considered Enterobacteriaceae isolates that were nonsusceptible to ceftriaxone (i.e., MIC >1 µg/mL) to be potentially ESBL producing (13). The sites shipped these isolates to a reference laboratory to confirm speciation, ESBL production, and molecular characterization (online Technical Appendix).
We classified patients as having complicated pyelonephritis if they were pregnant or male or had a current or preexisting functional or anatomic urinary tract abnormality or current immunocompromising condition. Possible preexisting urinary tract abnormalities were history of kidney stones, genitourinary procedures within the past 30 days, prostatic pathology, bladder catheter within the past 30 days, neurogenic urinary retention, ureteral stricture, duplicated collecting system, renal or bladder cancer, renal transplant, ureteral diversion, vesico-ureteral reflux, single kidney, and nephrostomy tubes. Immunocompromising conditions were diabetes, active cancer, systemic corticosteroid use, current use of other immunosuppressants, chronic debilitating condition, chronic renal insufficiency or failure, and HIV infection. We identified current complicating features on the basis of clinical findings or laboratory studies in the ED; possible complications were pregnancy, diabetes, bladder catheter, ureteral stent, percutaneous nephrostomy tube, prostatitis, nephrolithiasis, renal or perirenal abscess, and urinary retention. We recorded history of chronic debilitating illness, such as chronic obstructive pulmonary disease or heart or hepatic failure, but did not assign such illness as a criterion for complicated infection. We classified patients without criteria for complicated pyelonephritis as having uncomplicated pyelonephritis. We defined healthcare-associated infections as those in patients hospitalized or residing in a long-term care facility within the previous 90 days; other patients were classified as having community-acquired infections.
Statistical Analysis
To manage the study data, we used REDCap electronic-data capture tools hosted by the University of California, Los Angeles, CA, USA (14). We used SAS Version 9.3 (Cary, NC, USA) and Microsoft Excel 2013 (Redmond, WA, USA) to analyze data and used descriptive statistics to summarize patient characteristics and resistance prevalence. We calculated relative risks and 95% CIs to determine associations between epidemiologic and clinical characteristics and presence or absence of fluoroquinolone-resistant and ESBL-producing E. coli infections.
Of 817 enrolled patients with acute pyelonephritis, 793 (97.1%) submitted a urine culture. Of those 793 patients, 272 (34.3%) were excluded from analysis: 149 (18.8%) had no culture growth; 74 (9.3%) grew >1 contaminant; 17 (2.1%) grew >1 isolate at <104 CFU/mL; 25 (3.2%) grew >1 organism at 104 CFU/mL; and 7 (0.9%) had no fever. The study population consisted of 521 patients who grew 1 uropathogen at >104 CFU/mL. The case finding audit revealed a 66% enrollment of eligible patients. Enrolled and nonenrolled patients were similar for most characteristics, including E. coli susceptibility rates to TMP/SMX, ceftriaxone, and fluoroquinolones (Technical Appendix).
Among the 521 study patients, median age was 37 (range 18–88, interquartile range 26–52) years; 455 (87.3%) were female (Table 1). Most (446 [85.6%]) patients had a community-acquired infection; 74 (14.2%) had a healthcare-associated infection (70 with hospitalization and 9 with nursing home residence in the previous 90 days). A total of 286 (54.9%) patients had uncomplicated pyelonephritis; 235 (45.1%) had complicated pyelonephritis.
Of types of uropathogens in patients with uncomplicated and complicated pyelonephritis (Table 2), E. coli accounted for infections in 453 (86.9%) patients; 272 (60.0%) of the infections were uncomplicated, and 181 (40.0%) were complicated. Among the 286 patients with uncomplicated infections, E. coli accounted for 95.1%; among the 235 patients with complicated infections, E. coli accounted for 77.0% (Table 2).
E. coli antimicrobial drug resistance rates among patients with complicated pyelonephritis tended to be higher than rates for patients with uncomplicated cases, except for TMP/SMX (Table 3). Among all patients, E. coli resistance rates varied by drug: ampicillin, 57.2% (259/453); TMP/SMX, 36.4% (165/453); gentamicin, 9.9% (43/436); cefazolin, 14.2% (52/367); ceftriaxone, 7.7% (35/453); levofloxacin, 10.2% (33/325); and ciprofloxacin, 12.1% (48/397). Among 53 fluoroquinolone-resistant isolates, 8 (15.1%) were susceptible to ampicillin, 34 (64.2%) to gentamicin, and 27 (50.9%) to ceftriaxone. Of 46 isolates tested for susceptibility to cefazolin, 19 (41.3%) were susceptible; all 48 (100%) isolates tested were susceptible to a carbapenem. E. coli antimicrobial drug resistance rates for patients with uncomplicated and complicated pyelonephritis varied by study site (Technical Appendix Tables 2, 3).
Among patients with uncomplicated pyelonephritis, 17 (6.3%) of 272 E. coli isolates were resistant to fluoroquinolone. The range of prevalence by site was 0.0%–23.1%; for 2 sites, prevalence was >10% (Figure 1; Technical Appendix Tables 2, 3). Among patients with complicated pyelonephritis, 36 (19.9%) of 181 E. coli isolates showed fluoroquinolone resistance. The range of prevalence by site was 0.0%–50.0%; 8 sites had a prevalence >10%, 4 of which had a prevalence >20%. We found fluoroquinolone resistance associated with complicated E. coli infection, prior use of antimicrobial drugs and fluoroquinolone, hospital admission, and prior UTI resulting from a fluoroquinolone- or ceftriaxone-resistant organism (Table 4).
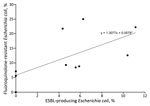
Among patients with uncomplicated pyelonephritis, ESBL production was found in 7 (2.6%) of 272 E. coli isolates; range by study site was 0.0%–8.3% (Figure 2; Technical Appendix Tables 2, 3). Among patients with complicated pyelonephritis, ESBL production was found in 22 (12.2%) of 181 E. coli isolates; range by site was 0.0%–17.2% (Figure 2; Technical Appendix Tables 2, 3). Frequencies of ESBL-producing E. coli isolates were higher among patients with antimicrobial drug resistance risk fac-tors than among those without these factors (Table 5). Nineteen (65.5%) of 29 patients with ESBL-producing E. coli infection had a recognized risk factor for antimicrobial drug resistance (Figure 2; Technical Appendix Tables 2, 3). Sixteen (55.2%) had antimicrobial drug exposure within the previous 60 days. During the previous 90 days, 6 (20.7%) had healthcare-setting exposure and 4 (13.8%) had travel outside North America. We found ESBL-producing E. coli infection associated with complicated infection, prior antimicrobial drug use, travel outside North America, and prior UTI resulting from a fluoroquinolone- or ceftriaxone-resistant organism. Among 37 isolates that grew other Enterobacteriaceae, including Klebsiella pneumoniae, 1 (2.7%) was ESBL producing. Among 29 ESBL-producing E. coli isolates, susceptibility rates to other antimicrobial drugs were 41.4% to TMP/SMX, 18.5% to ciprofloxacin, 21.7% to levofloxacin, 41.4% to gentamicin, and 100% to carbapenem. The prevalence of E. coli fluoroquinolone resistance correlated with the prevalence of ESBL-producing E. coli by site (Figure 3).
We further characterized 26 ESBL-producing E. coli isolates and 1 K. pneumoniae isolate. Among E. coli isolates, PCR identified multiple ESBL types: 22 (84.6%) produced CTX-M-15 (16 [61.5%] produced only CTX-M-15; 6 [23.1%] produced CTX-M-15 and TEM-1); 2 (7.7%) produced CTX-M-27; 1 (3.8%) produced CTX-M-14; and 1 (3.8%) produced CTX-M-14 and TEM-1. The K. pneumoniae isolate produced SHV-1 and CTX-M-15 ESBL types. Sixteen (61.5%) of the E. coli isolates were clonal type O25b-ST131.
Among patients with an E. coli infection, 223 (49.2%) were discharged from the ED and 229 (50.6%) were admitted to the hospital; of these, 13 (5.8%) and 18 (7.9%), respectively, were treated with an antimicrobial drug that lacked in vitro activity against their infection. Of 53 patients with a fluoroquinolone-resistant and 29 with an ESBL-producing infection, 24 (45.3%) and 22 (75.0%), respectively, were initially treated with in vitro–inactive antimicrobial drugs. Among 29 patients with an ESBL infection, 9 (31.0%) were discharged from the ED; an in vitro–inactive antimicrobial drug was initially prescribed to 7 (77.8%). Among 20 (69.0%) hospitalized patients with an ESBL infection, 15 (75.0%) were initially given an in vitro–inactive antimicrobial drug; 1 of those 15 patients was given gentamicin, 13 were given cephalosporin, and 1 was given both.
For 2013–2014, we found that prevalence of E. coli fluoroquinolone resistance was >10% for patients with uncomplicated pyelonephritis at 2 of 10 sites and >20% for patients with complicated infections at 4 of 10 sentinel sites surveyed in the United States. E. coli fluoroquinolone resistance was particularly prevalent in groups with antimicrobial drug resistance risk. These rates exceed thresholds for the 2010 IDSA treatment guidelines, which recommend consideration of an additional antimicrobial drug of a different class and other agents (9). Data from a similar study we conducted during 2000–2004 (15) indicate that, among all healthcare-seeking ED patients with acute pyelonephritis, the prevalence of fluoroquinolone-resistant E. coli increased from 3.9% during 2000–2004 to 11.7% during 2013–2014. During 2000–2004, we found no infections caused by ESBL-producing bacteria. As in other parts of the world, ESBL-producing Enterobacteriaceae are emerging among patients with community-acquired UTI in the United States. For patients with uncomplicated and complicated pyelonephritis caused by E. coli, we found that 2.6% and 12.2%, respectively, had infection caused by an ESBL-producing organism; rates were even higher for patients with risk factors. The globally disseminated, multidrug-resistant clone ST131, which produces CTX-M-15 β-lactamase, accounted for 85.2% of these infections. Of ESBL-infected patients, about one third lacked traditional antimicrobial resistance risk factors (i.e., antimicrobial drug or healthcare-setting exposure or international travel), suggesting that these isolates are now endemic in some US communities. Among ESBL-infected patients, about three quarters were initially treated with an antimicrobial drug lacking in vitro activity, including the sickest patients who required hospitalization. We did not collect outcome data, but lack of in vitro activity of the antimicrobial drug used for treatment has been associated with relatively poor response rates among patients with pyelonephritis (1,10,11).
Previous surveys have suggested that E. coli fluoroquinolone resistance rates are increasing in the United States. Among outpatients seeking care at 30 US centers during 2003–2004, 59 (6.8%) of 862 E. coli isolates were resistant to ciprofloxacin (2). Another analysis of >12 million urine specimens from US outpatient centers found that the E. coli fluoroquinolone resistance rate increased from 3.0% in 2000 to 17.1% in 2010 (3). Such laboratory-based resistance surveillance data may exaggerate the prevalence of resistance because patients for whom cultures are performed would be expected to have received prior therapy and to have had healthcare exposure more frequently than patients without cultures. Isolate-driven studies require retrospective review of records, which have missing and inaccurate data; also, uncertainty may exist regarding whether a specimen is from a patient with an actual clinical infection, rather than being a colonized or contaminated specimen. In routine practice, providers typically estimate local resistance rates on the basis of an antibiogram published by the local hospital laboratory. Resistance rates determined from antibiograms are prone to bias and indicate only whether the specimen was obtained from an outpatient or inpatient location.
In contrast, we conducted syndromic surveillance of patients who sought care at a geographically diverse network of US EDs. We studied acute pyelonephritis because it is a distinct clinical syndrome for which cultures are routinely obtained and because isolates grown are less likely to be contaminants or colonizers, compared with those from patients with suspected cystitis. Historical data were obtained by real-time patient interviews, enabling accurate classification of complicated and uncomplicated pyelonephritis and ascertainment of antimicrobial drug resistance risk factors. To identify biases, we compared characteristics of enrolled and nonenrolled qualifying patients and found their characteristics to be similar, suggesting the validity of our sampling. Consequently, our data and that of other studies indicate that, in some parts of the United States, the rate of E. coli fluoroquinolone resistance among uropathogens is >10% among patients with uncomplicated pyelonephritis and >20% among those with complicated infections. However, variability exists; among patients with uncomplicated pyelonephritis, 3 of 10 sites had fluoroquinolone resistance rates <5%. Consistent with findings of previous investigations, we found E. coli fluoroquinolone resistance associated with complicated infection, prior use of antimicrobial drugs and fluoroquinolone, hospitalization, and prior UTI caused by a fluoroquinolone- or ceftriaxone-resistant organism (16,17).
By using active, prospective surveillance, we found that ESBL-producing E. coli infections have now emerged to a considerable degree among patients with clinically confirmed community-acquired infections in parts of the United States, including among persons lacking commonly recognized antimicrobial drug resistance risk factors. This new observation is not unexpected, given the reported epidemiology of ESBL-producing Enterobacteriaceae infections in communities outside North America. CTX-M enzymes are currently the most prevalent ESBL types worldwide. The ST131 clone is largely responsible for the international epidemic caused by CTX-M-15–producing E. coli, including infections seen in the United States (7,18,19).
A few US laboratory-based surveillance studies have reported community-acquired ESBL infections. Peirano et al. (19) described ESBL-producing E. coli isolates from 30 community-dwelling patients at 5 Chicago-area hospitals during 2008. These ESBL-producing strains represented 2%–8% of E. coli isolates at each hospital. Khawcharoenporn et al. (20) reported that ≈5% of Enterobacteriaceae isolates from ED patients with presumed UTI during 2008–2009 were ESBL-producing, although specific risk data were not provided. Doi et al. (7) reviewed records of patients with cultures that grew ESBL-producing E. coli isolates at 1 hospital in each of 5 US cities during 2009–2010. Among 13,270 E. coli isolates, 523 (3.9%) were ESBL producing. Of the 291 patients infected or colonized with ESBL-producing E. coli as outpatients, infections of 107 (36.8%) were thought to be community associated. Community ESBL-producing E. coli isolates were resistant to multiple agents: 87.5% to ciprofloxacin or levofloxacin and 39.4% to gentamicin. All isolates from that study were susceptible to a carbapenem; we also found that all ESBL-producing isolates in our investigation were susceptible to a carbapenem.
Studies from outside North America have identified several characteristics associated with ESBL infection, such as recurrent and complicated UTI; advanced age; recent hospitalization; use of a β-lactam or fluoroquinolone; travel to Asia, Middle East, or Africa; and fresh water swimming (5,21–23). Banerjee et al. (23) conducted a case–control study among adults with E. coli clinical isolates cultured in the Chicago area and found that ESBL infection was associated with travel to India, ciprofloxacin use, and age. We found ESBL-producing E. coli infection associated with complicated infection, prior antimicrobial drug use, travel outside North America, and prior UTI resulting from fluoroquinolone- or ceftriaxone-resistant infection. The importance of investigating past susceptibility data when considering empirical treatment is highlighted by our observation that both fluoroquinolone resistance and ESBL-production were associated with previous resistant infections.
Our study has limitations. We were unsuccessful in enrolling all consecutive patients, which may have introduced bias in our selection of patients. However, our audit of eligible case-patients showed similarity of enrolled and nonenrolled patients, including their E. coli antimicrobial drug susceptibility rates; furthermore, most (>97%) enrolled patients had urine cultures collected, reducing potential bias. The prevalence of ESBL-producing strains may have been underestimated because we did not use ESBL-selective media and used only ceftriaxone instead of several advanced-generation cephalosporins to screen for presence of ESBL-producing strains. However, this method enabled us to have greater site participation; furthermore, screening isolates with a ceftriaxone MIC >1 µg/mL has been reported to have a sensitivity >98% on the basis of phenotypic testing (24). In addition, some patients may not have had pyelonephritis if a contaminated specimen was misinterpreted as noncontaminated, although >97% of patients had urine collected by a technique that minimizes contamination (i.e., clean catch, urethral catheterization, or suprapubic aspiration), and the diagnosis of pyelonephritis was further supported by clinical assessment. Our definition of confirmed infection as growth of 1 uropathogen at >104 CFU/mL may have missed some cases of pyelonephritis, although only ≈5% grew a single uropathogen at <104 CFU/mL or grew >1 uropathogen at >104 CFU/mL. Our hospitals were large US urban centers and may not represent patients in other settings, emphasizing the importance of local surveillance.
IDSA treatment guidelines for acute uncomplicated pyelonephritis recommend that, if the fluoroquinolone-resistance rate is >10%, then in addition to a fluoroquinolone, an agent of another class (i.e., ceftriaxone or gentamicin) should be administered (9). Our findings indicate that fluoroquinolone resistance rates for E. coli are approaching or exceed this threshold for patients with uncomplicated pyelonephritis in many parts of the United States. For uncomplicated cystitis, the guidelines recommend alternative agents if the resistance rate is >20%, which is the current situation for fluoroquinolones in many settings for patients with complicated pyelonephritis. Unfortunately, we found that only one half to two thirds of fluoroquinolone-resistant E. coli isolates were susceptible to ceftriaxone or gentamicin. Rates of fluoroquinolone-resistant and ESBL-producing E. coli infections correlate to geographic location. Prior exposure to antimicrobial drugs or a healthcare setting, travel outside the United States, and a history of an antimicrobial drug–resistant infection substantially increases the chance that a person will have a current fluoroquinolone-resistant or ESBL-producing E. coli infection. Therefore, in settings with high fluoroquinolone resistance rates, in settings where ESBL-producing Enterobacteriaceae infections have emerged, or among persons with antimicrobial drug resistance risk factors (especially patients with or at risk for severe sepsis), healthcare providers should consider empirical treatment with a carbapenem or another agent found to be consistently active on the basis of the local antibiogram. In this study, ≈50% of patients with pyelonephritis were managed as outpatients. Currently, no oral antimicrobial drugs with consistent in vitro activity are available for empirical treatment of pyelonephritis caused by ESBL-producing E. coli uropathogens. Our findings, including the variability in the prevalence of resistance by site, show that increased local efforts to enhance surveillance for antimicrobial drug resistance are necessary to best inform treatment decisions. Furthermore, availability of new antimicrobial drugs must be expedited.
Dr. Talan is professor of medicine in residence (emeritus) at the David Geffen School of Medicine at UCLA in Los Angeles. His research focuses on emergency department–based surveillance and epidemiologic research of emerging infections and clinical investigations of acute infectious diseases.
Acknowledgments
We thank Robert Badal, Brian Johnson, and Krystyna Kazmierczak at the International Health Management Associates, Inc. (IMHA). We also thank Ellen Jo Baron, Kavitha Pathmarajah, Britany Zeglin, Mary Mulrow, Shelley Fuentes, Laurie Kemble, Danielle Beckham, Niccole Neal, Ada Rubin, Sarah Usher, Stephen Peterson, Lila Steinberg, Christine Sayegh, Kimberly Dehnkamp, Kamil Narayan, Silas Bussman, Richard Dwyer, and the residents and staff at the participating emergency departments.
We also thank the site investigators of the EMERGEncy ID NET Study Group: Fredrick M. Abrahamian, Olive View–University of California, Los Angeles Medical Center, Sylmar, CA, USA; Johanna Moore, Hennepin County Medical Center, Minneapolis, MN, USA; Jon Femling, University of New Mexico Health Sciences Center, Albuquerque, NM, USA; William K. Chiang, Bellevue Hospital Center, New York, NY, USA; Frank LoVecchio, Maricopa Medical Center, Phoenix, AZ, USA; Jon Jui, Oregon Health & Science University, Portland, OR, USA; Manish Garg, Temple University School of Medicine, Philadelphia, PA, USA; Mark T. Steele, University of Missouri–Kansas City, Kansas City, MO, USA; Sukhjit S. Takhar, Brigham and Women’s Hospital, Boston, MA, USA; and Richard Rothman, The Johns Hopkins Hospital, Baltimore, MD, USA.
This research was supported by a grant from the US Centers for Disease Control and Prevention, Cooperative Agreement Number U01CK000176. Confirmation of ESBL production and molecular characterization was completed by the IHMA laboratory and funded by Merck & Co., Inc.
References
- Talan DA, Stamm WE, Hooton TM, Moran GJ, Burke T, Iravani A, Comparison of ciprofloxacin (7 days) and trimethoprim-sulfamethoxazole (14 days) for acute uncomplicated pyelonephritis in women: a randomized trial. JAMA. 2000;283:1583–90. DOIPubMedGoogle Scholar
- Zhanel GG, Hisanaga TL, Laing NM, DeCorby MR, Nichol KA, Weshnoweski B, ; NAUTICA Group. Antibiotic resistance in Escherichia coli outpatient urinary isolates: final results from the North American Urinary Tract Infection Collaborative Alliance (NAUTICA). Int J Antimicrob Agents. 2006;27:468–75. DOIPubMedGoogle Scholar
- Sanchez GV, Master RN, Karlowsky JA, Bordon JM. In vitro antimicrobial resistance of urinary Escherichia coli isolates among U.S. outpatients from 2000 to 2010. Antimicrob Agents Chemother. 2012;56:2181–3. DOIPubMedGoogle Scholar
- Dalhoff A. Global fluoroquinolone resistance epidemiology and implications for clinical use. Interdiscip Perspect Infect Dis. 2012;2012:976273. PubMedGoogle Scholar
- Johnson SW, Anderson DJ, May DB, Drew RH. Utility of a clinical risk factor scoring model in predicting infection with extended-spectrum β-lactamase-producing enterobacteriaceae on hospital admission. Infect Control Hosp Epidemiol. 2013;34:385–92. DOIPubMedGoogle Scholar
- Hayakawa K, Gattu S, Marchaim D, Bhargava A, Palla M, Alshabani K, Epidemiology and risk factors for isolation of Escherichia coli producing CTX-M-type extended-spectrum β-lactamase in a large U.S. Medical Center. Antimicrob Agents Chemother. 2013;57:4010–8. DOIPubMedGoogle Scholar
- Doi Y, Park YS, Rivera JI, Adams-Haduch JM, Hingwe A, Sordillo EM, Community-associated extended-spectrum β-lactamase-producing Escherichia coli infection in the United States. Clin Infect Dis. 2013;56:641–8. DOIPubMedGoogle Scholar
- Peirano G, van der Bij AK, Gregson DB, Pitout JD. Molecular epidemiology over an 11-year period (2000 to 2010) of extended-spectrum β-lactamase-producing Escherichia coli causing bacteremia in a centralized Canadian region. J Clin Microbiol. 2012;50:294–9. DOIPubMedGoogle Scholar
- Gupta K, Hooton TM, Naber KG, Wullt B, Colgan R, Miller LG, ; Infectious Diseases Society of America; European Society for Microbiology and Infectious Diseases. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: a 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011;52:e103–20. DOIPubMedGoogle Scholar
- Shin J, Kim J, Wie SH, Cho YK, Lim SK, Shin SY, Fluoroquinolone resistance in uncomplicated acute pyelonephritis: epidemiology and clinical impact. Microb Drug Resist. 2012;18:169–75. DOIPubMedGoogle Scholar
- Lee S, Song Y, Cho SH, Kwon KT. Impact of extended-spectrum beta-lactamase on acute pyelonephritis treated with empirical ceftriaxone. Microb Drug Resist. 2014;20:39–44. DOIPubMedGoogle Scholar
- Talan DA, Moran GJ, Mower W, Newdow M, Ong S, Slutsker L, The EMERGEncy ID NET Study Group. EMERGEncy ID NET: an emergency department-based emerging infections sentinel network. Ann Emerg Med. 1998;32:703–11. DOIPubMedGoogle Scholar
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; twenty-second informational supplement. CLSI document M100–S22. Wayne (PA): The Institute; 2012.
- Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81. DOIPubMedGoogle Scholar
- Talan DA, Krishnadasan A, Abrahamian FM, Stamm WE, Moran GJ; EMERGEncy ID NET Study Group. Prevalence and risk factor analysis of trimethoprim-sulfamethoxazole- and fluoroquinolone-resistant Escherichia coli infection among emergency department patients with pyelonephritis. Clin Infect Dis. 2008;47:1150–8. DOIPubMedGoogle Scholar
- Bedoin M, Cazorla C, Lucht F, Berthelot P, Boyer M, Carricajo A, Risk factors for quinolone-resistance in women presenting with Escherichia coli acute pyelonephritis. Med Mal Infect. 2014;44:206–16. DOIPubMedGoogle Scholar
- van der Starre WE, van Nieuwkoop C, Paltansing S, van’t Wout JW, Groeneveld GH, Becker MJ, Risk factors for fluoroquinolone-resistant Escherichia coli in adults with community-onset febrile urinary tract infection. J Antimicrob Chemother. 2011;66:650–6. DOIPubMedGoogle Scholar
- Johnson JR, Urban C, Weissman SJ, Jorgensen JH, Lewis JS II, Hansen G, ; AMERECUS Investigators. Molecular epidemiological analysis of Escherichia coli sequence type ST131 (O25:H4) and blaCTX-M-15 among extended-spectrum-β-lactamase-producing E. coli from the United States, 2000 to 2009. Antimicrob Agents Chemother. 2012;56:2364–70. DOIPubMedGoogle Scholar
- Peirano G, Costello M, Pitout JD. Molecular characteristics of extended-spectrum beta-lactamase-producing Escherichia coli from the Chicago area: high prevalence of ST131 producing CTX-M-15 in community hospitals. Int J Antimicrob Agents. 2010;36:19–23. DOIPubMedGoogle Scholar
- Khawcharoenporn T, Vasoo S, Singh K. Urinary tract infections due to multidrug-resistant Enterobacteriaceae: prevalence and risk factors in a Chicago emergency department. Emerg Med Int. 2013;2013:258517.
- Søraas A, Sundsfjord A, Sandven I, Brunborg C, Jenum PA. Risk factors for community-acquired urinary tract infections caused by ESBL-producing enterobacteriaceae—a case–control study in a low prevalence country. PLoS One. 2013;8:e69581. DOIPubMedGoogle Scholar
- Azap OK, Arslan H, Serefhanoğlu K, Colakoğlu S, Erdoğan H, Timurkaynak F, Risk factors for extended-spectrum beta-lactamase positivity in uropathogenic Escherichia coli isolated from community-acquired urinary tract infections. Clin Microbiol Infect. 2010;16:147–51. DOIPubMedGoogle Scholar
- Banerjee R, Strahilevitz J, Johnson JR, Nagwekar PP, Schora DM, Shevrin I, Predictors and molecular epidemiology of community-onset extended-spectrum β-lactamase–producing Escherichia coli infection in a Midwestern community. Infect Control Hosp Epidemiol. 2013;34:947–53. DOIPubMedGoogle Scholar
- Huang Y, Carroll KC, Cosgrove SE, Tamma PD. Determining the optimal ceftriaxone MIC for triggering extended-spectrum β-lactamase confirmatory testing. J Clin Microbiol. 2014;52:2228–30. DOIPubMedGoogle Scholar
Figures
Tables
Follow Up
Medscape CME Quiz
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to http://www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the “Register” link on the right hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/about-ama/awards/ama-physicians-recognition-award.page. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title:
Fluoroquinolone-Resistant and Extended-Spectrum β-Lactamase–Producing Escherichia coli infections in Patients with Pyelonephritis, United States
CME Questions
1. You are seeing a 37-year-old woman with a 5-day history of fever, dysuria, and flank pain. You suspect that the patient has pyelonephritis. What should you consider the Infectious Diseases Society of America (IDSA) 2010 recommendations regarding the treatment of uncomplicated pyelonephritis as you recommend treatment for this patient?
A. Fluoroquinolones are not recommended as initial treatment for pyelonephritis
B. Ceftriaxone or gentamicin may be added to initial treatment
C. Trimethoprim/sulfamethoxazole is a good choice for patients with extended-spectrum β-lactamase (ESBL) bacterial infections
D. Fluoroquinolones should be avoided only if the local resistance rate is in excess of 5%
2. The patient's initial urine culture grows Escherichia coli. What were the approximate rates of fluoroquinolone resistance and ESBL-producing E. coli among patients with uncomplicated pyelonephritis in the current study?
A. Less than 1% for both
B. An estimated 6% of isolates were resistant to fluoroquinolones; nearly 3% were ESBL producers
C. Rate of fluoroquinolone resistance among isolates was 15%; nearly half were ESBL producers
D. Nearly 40% of isolates were resistant to fluoroquinolones; 1% were ESBL producers
3. Which of the following variables was not a significant risk factor for E. coli fluoroquinolone resistance in the current study?
A. Residence in a long-term care facility within 90 days
B. Recent hospital admission
C. Complicated vs. uncomplicated pyelonephritis
D. Previous urinary tract infection with fluoroquinolone- or ceftriaxone-resistant organism
4. Which of the following variables was the strongest risk factor for ESBL-producing E. coli in the current study?
A. Fluoroquinolone resistance of E. coli
B. Age older than 65 years
C. Residence in a long-term care facility within 90 days
D. Intravenous antibiotic administration within the past 30 days
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
1Preliminary results of this research were presented at the 2014 IDWeek Meeting, Philadelphia, PA, USA, October 8–12, 2014.
Related Links
Table of Contents – Volume 22, Number 9—September 2016
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
David A. Talan, Olive View-UCLA Medical Center, 14445 Olive View Dr, North Annex, Sylmar, CA 91342, USA
Top