Volume 23, Number 1—January 2017
CME ACTIVITY - Research
Analysis of Anthrax Immune Globulin Intravenous with Antimicrobial Treatment in Injection Drug Users, Scotland, 2009–2010
Figure 2
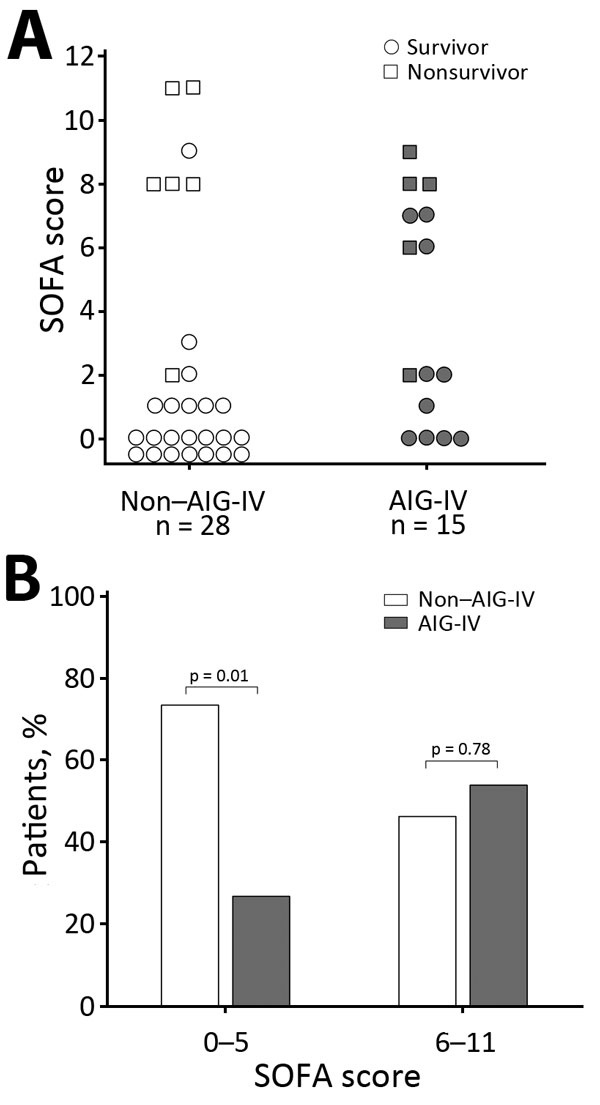
Figure 2. SOFA scores of injection drug users who did and did not receive AIG-IV, Scotland, UK, 2009–2010. A) Individual SOFA scores for patients who did or did not receive AIG-IV and did or did not survive. B) Proportion of patients who did or did not receive AIG-IV for those who had admission SOFA scores of 0–5 and a low risk for death and for those with SOFA scores of 6–11 and a higher risk for death. AIG-IV, anthrax immune globulin intravenous; SOFA, sequential organ failure.
1These authors contributed equally to this article.
Page created: December 14, 2016
Page updated: December 14, 2016
Page reviewed: December 14, 2016
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.