Volume 23, Number 12—December 2017
CME ACTIVITY - Research
Group B Streptococcus Infections Caused by Improper Sourcing and Handling of Fish for Raw Consumption, Singapore, 2015–2016
Cite This Article
Citation for Media
Introduction

In support of improving patient care, this activity has been planned and implemented by Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.00 AMA PRA Category 1 Credit(s)™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at http://www.medscape.org/journal/eid; and (4) view/print certificate. For CME questions, see page XXX.
Release date: November 15, 2017; Expiration date: November 15, 2018
Learning Objectives
Upon completion of this activity, participants will be able to:
• Evaluate clinical and epidemiological findings of a human group B Streptococcus outbreak in Singapore, based on a microbial study of raw fish and human samples
• Determine the pathogenic potential of fish and human samples after a human group B Streptococcus outbreak in Singapore, based on a microbial study of these samples
• Assess the public health implications of a human group B Streptococcus outbreak in Singapore, based on a microbial study of raw fish and human samples.
CME Editor
Thomas J. Gryczan, MS, Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: Thomas J. Gryczan, MS, has disclosed no relevant financial relationships.
CME Author
Laurie Barclay, MD, freelance writer and reviewer, Medscape, LLC. Disclosure: Laurie Barclay, MD, has disclosed the following relevant financial relationships: owns stock, stock options, or bonds from Alnylam; Biogen; Pfizer.
Authors
Disclosures: Man Ling Chau, BSc; Swaine L. Chen, MD, PhD; Min Yap, BSc; Sri Harminda Pahm Hartantyo, PhD; Paul King Tiong Chiew, DVM, MVPM; Charlene Judith Fernandez, BS, DVM, MPH, CPH; Wai Kwan Wong, BSc; Rockey Kwonghung Fong, BVSc; Wei Ling Tan, BSc; Brian Zi Yan Tan, PhD; Youming Ng, BSc; Kyaw Thu Aung, MBBS, MSc, PGDip; Kurosh S. Mehershahi, PhD; Christopher Goh, MSc; Joanne Su Lin Kang, PhD; Timothy Barkham, MBBS, MSc, FRCPath; Adeline Oi Kheng Leong, MSc; Ramona Alikiiteaga Gutiérrez, DVM, PhD; and Lee Ching Ng, PhD, have disclosed no relevant financial relationships.
Abstract
We assessed microbial safety and quality of raw fish sold in Singapore during 2015–2016 to complement epidemiologic findings for an outbreak of infection with group B Streptococcus serotype III sequence type (ST) 283 associated with raw fish consumption. Fish-associated group B Streptococcus ST283 strains included strains nearly identical (0–2 single-nucleotide polymorphisms) with the human outbreak strain, as well as strains in another distinct ST283 clade (57–71 single-nucleotide polymorphisms). Our investigations highlight the risk for contamination of freshwater fish (which are handled and distributed separately from saltwater fish sold as sashimi) and the need for improved hygienic handling of all fish for raw consumption. These results have led to updated policy and guidelines regarding the sale of ready-to-eat raw fish dishes in Singapore.
A major outbreak of group B Streptococcus (GBS) infection associated with consumption of a Chinese-style raw fish dish (yusheng) occurred in Singapore during 2015 and involved 238 persons during the first half of the year (1). The Yusheng was typically made from sliced Asian bighead carp (Hypophthalmichthys nobilis) and snakehead (Channa spp.) and served as a side dish with porridge by food stalls within larger eating establishments. Persons with severe clinical cases had meningoencephalitis, bacteremia, and septic arthritis (2–4). GBS, or Streptococcus agalactiae, was identified as the causative agent (2,3).
GBS is found in ≈30% of healthy adults (5) and is a member of the human commensal gastrointestinal and genitourinary flora (4). GBS is a common cause of neonatal sepsis, is acquired by newborns from the vaginal flora of the mother, and is an increasingly common pathogen among vulnerable populations (6). The incidence of invasive disease in adults, particularly older adults, has been increasing (7,8). GBS is also a fish and bovine pathogen (9). Although GBS has been shown to colonize the gastrointestinal tract of humans linked to fish consumption (9), foodborne transmission leading to invasive disease has not been reported. Local epidemiologic investigations conducted separately (2,3) identified a single strain of GBS serotype III sequence type (ST) 283 as the causative agent of the outbreak in Singapore during 2015. GBS ST283 had previously been isolated from tilapia in Thailand (10) and in adult human cases in Hong Kong (11). However, GBS ST283 has not been reported to colonize the human gastrointestinal tract, although to date only 1 study of fish mongers and fish handlers has specifically looked for colonization by this strain (12).
We investigated microbial safety and quality of fish sold in the Singapore market during and after the outbreak during 2015 to trace the source of GBS ST283 and provide risk assessment data to support outbreak control and prevention measures. Shortly after identification of GBS ST283 as the cause of the outbreak, these data supported implementing a ban on the sale of ready-to-eat (RTE) dishes containing raw freshwater fish, as well as imposing additional requirements for sale of RTE raw fish dishes made with saltwater fish (13). We report the results of our analysis, which might assist the review of guidelines for handling of fish meant for raw consumption in Singapore and other countries. This report offers unique food and environmental insights into the investigation of this outbreak and complements published epidemiologic findings (2,3).
Collection of Fish and Fish Tank Water Samples
We collected samples of fish commonly used for raw consumption (n = 997) and fish tank water for holding live freshwater fish (n = 102) along the supply chain in Singapore during August 2015–January 2016 (Technical Appendix Table 1). We tested samples for GBS, Aeromonas spp., Listeria monocytogenes, Salmonella spp., Vibrio cholerae, and V. parahaemolyticus, and determined Escherichia coli counts, Staphylococcus aureus counts, and standard plate counts (SPCs) (Technical Appendix). We characterized selected species to determine their virulence potential (Technical Appendix).
Statistical Analysis
We evaluated significant differences (p<0.05) between bacterial counts (log10 CFU/g) and presence of specific foodborne bacteria by using Kruskal-Wallis, Mann-Whitney, χ2, and Fisher exact tests as appropriate. We performed analysis by using SPSS version 24.0 software (IBM, Armonk, NY, USA).
Raw Fish Samples from Food Stalls and Restaurants/Snack Bars
Although raw freshwater and saltwater fish were served as RTE food at food stalls, only raw saltwater fish were reportedly served at restaurants/snack bars. PCR positivity rates were 43.5% (20/46) for GBS and 23.9% (11/46) for GBS serotype III in sliced fish samples from food stalls. Fish sampled from restaurants/snack bars had significantly lower rates (p<0.05) of 9.2% (26/282) for GBS and 0.7% (2/282) for GBS serotype III (Table). Among the 20 GBS PCR-positive samples from food stalls, 5 yielded isolates; these isolates were of serotype II ST652, serotype III ST283, serotype III ST335, and serotype V ST1 (Technical Appendix Table 3). The GBS ST283 isolated was from a RTE sliced fish sample sold as grass carp collected from a food stall linked to a human case, as described (12). We did not detect GBS ST283 in samples from restaurant/snack bars; however, we did find a range of other GBS, including serotypes Ia ST7, Ia ST103, Ia ST485, III ST651, III ST861, V ST1, V ST24, VI ST167, and VII ST1.
We found Salmonella serogroup B ST29 (serovar Stanley) (n = 2); V. parahaemolyticus (negative for tdh, trh1, and trh2 genes) (n = 1); and non–O1 V. cholerae (n = 1) in freshwater fish samples from food stalls. We also isolated V. cholerae from saltwater fish samples, 1 from a food stall and 1 from a restaurant. We detected L. monocytogenes in 5 samples from restaurants/snack bars.
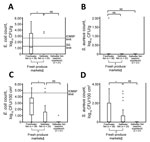
SPCs of most RTE raw freshwater (71.4%, 5/7) and saltwater (85.7%, 18/21) fish samples from food stalls exceeded the regulatory limit for RTE food (5 log10 CFU/g) in Singapore (14). We observed no difference in SPCs for fish slices intended for raw consumption and cooking purposes (Figure 1). We also found that 24.8% (70/282) of saltwater fish samples from restaurants/snack bars did not comply with regulatory limits for SPCs, E. coli counts (1.3 log10 CFU/g), or both (14). These results showed the poor quality of RTE raw freshwater and saltwater fish sold at food stalls in comparison to those sold at restaurants and snack bars.
Comparison of Freshwater and Saltwater Fish Samples from Fresh Produce Markets
Fish sold at food stalls were typically procured from local fresh produce markets. For the 62 samples of whole fish and fish parts we collected from these markets, we detected GBS ST283 in 28.2% (11/39) of the freshwater fish (Table), which included fish sold as tilapia, Asian bighead carp, grass carp, snakehead-haruan, snakehead-toman, and silver carp (Technical Appendix Table 3). However, we did not detect GBS ST283 in saltwater fish. Other GBS strains detected among these fish include serotypes Ia ST7, Ia ST23, Ia ST24, and II ST28 (Technical Appendix Table 3).
We detected Aeromonas spp. (48.4%, 30/62), S. aureus (27.4%, 17/62), non–O1 V. cholerae (12.9%, 8/62) and V. parahaemolyticus (negative for tdh, trh1, and trh2 genes) (6.4%, 4/62) in fish samples from fresh produce markets. There was no difference in positivity rates of these organisms between freshwater and saltwater fish. We did not detect L. monocytogenes or Salmonella spp. in any fish samples collected from fresh produce markets.
Approximately 42% (15/36) of freshwater fish muscle samples had SPCs or E. coli counts, or both, exceeding regulatory limits for RTE food in Singapore (14). Positivity rates for GBS, GBS serotype III, and E. coli, as well as SPCs for saltwater fish, were significantly lower (p<0.05) (Figure 1; Table). E. coli and S. aureus counts for freshwater fish surfaces were significantly higher (p<0.05) than those for saltwater fish (Figure 2).
We collected 4 fish tank water samples from wet markets and supermarkets. One water sample and the live freshwater fish the tank contained were positive for GBS by PCR and non–O1 V. cholerae by culture; the associated fish was positive for GBS ST283 by culture. Two other fish tank water samples and the live fish the tanks contained were positive for E. coli, S. aureus, or both. The level of E. coli detected in each positive fish tank water sample was 1.3 log10 CFU/500 mL, which was greater than the 1 log10 CFU/500 mL coliform (which includes E. coli) limit set by the British Columbia Centre for Disease Control (15).
Whole Freshwater Fish and Fish Tank Water from Ports
We tested for GBS only in whole fish and fish tank water samples collected from ports. We detected GBS ST283 in 1% (6/586) of freshwater fish samples; positive samples were from Asian bighead carps imported from and farmed in Malaysia. For 98 fish tank water samples collected from ports, 55.1% (54/98) were positive for GBS, and 6.1% (6/98) were positive for GBS ST283. Three of the GBS ST283–positive fish were kept in fish tank water that was also positive for GBS ST283.
Comparison of Saltwater Fish from Fresh Produce Markets and Sashimi Suppliers
Our data indicate the risk for contamination of fish sold at local fresh produce markets, although saltwater fish samples from fresh produce markets had lower rates of contamination than freshwater fish samples. The SPCs and the positivity rates for E. coli in saltwater fish samples from sashimi suppliers were significantly lower (p<0.05) than those for saltwater fish samples from fresh produce markets (Figure 3; Table), which suggested that the microbial quality of fish could be managed by improvements in handling throughout distribution channels. None of the saltwater fish muscle samples from sashimi suppliers exceeded the Singapore SPC (5 log10 CFU/g) and E. coli (1.3 log10 CFU/g) limits for RTE food (14). We did not detect GBS, S. aureus, V. cholerae, and V. parahaemolyticus in any fish samples collected from sashimi suppliers. However, we detected L. monocytogenes in 1 salmon sample.
Characterization of GBS Isolates
We detected 6 GBS serotypes (Ia, II, III, V, VI, and VII) and 13 STs (1, 7, 23, 24, 28, 103, 167, 283, 335, 485, 651, 652, and 861) in fish (Technical Appendix Table 3). Although most strains were within clonal complexes (1, 10, 17, 19, and 23) associated with human carriage and diseases (16), a total of 20 isolates from 7 sashimi samples (SGEHI2015-IV45, SGEHI2015-IV72, SGEHI2015-IV74, SGEHI2015-IV89, SGEHI2015-IV100, SGEHI2015-IV211, and SGEHI2015-IV232) did not belong to these clonal complexes. These strains had few closely related strains in the public genomic databases, and the closely related strains are mostly from animals (Technical Appendix Figure 2).
We found GBS ST283 only among freshwater fish and water for holding freshwater fish. Genomic analyses indicated that GBS ST283 isolated from fish clustered in 2 clades (Figure 4). The first clade included 12 isolates from 6 fish from a food stall, a fresh produce market and a port, and 4 fish tank water samples from a port. Genome sequencing showed that these 12 isolates were nearly identical (0–2 SNPs and 0, 1, and 12 indels all in homopolymeric runs of >4 nt) compared with the 2.1-Mbp genome of the reference human outbreak strain, SG-M1, isolated from a meningitis patient during the GBS outbreak in Singapore during 2015 (12,17). Isolates that clustered into the second clade were 20 isolates from 12 fish and 2 fish tank water samples and did not include any human isolates either from this outbreak or from previous reports of human GBS infecting isolates. Sequences of these isolates showed higher intraclade diversity (57–71 SNPs and 11–33 indels) when compared with the SG-M1 genome (Figure 4).
Characterization of S. aureus, V. cholerae, and V. parahaemolyticus Isolates
We characterized 18 S. aureus isolates from 17 fish. All except 1 were obtained from fish surfaces. We detected >1 enterotoxin gene in two thirds of these isolates and the sec gene in 55.6% (10/18) of the isolates. Other enterotoxin genes (sea, seg, seh, sei, and sel) were detected at much lower rates (5.6% [1/18] to 11.1% [2/18]). We detected 4 enterotoxin genes (sec, seg, sei, and sel) in a S. aureus isolate obtained from the surface of a wolf herring sample collected from a port. We did not detect virulence genes (ctxA, ctxB, and tcpA) in any of the 16 non–O1 V. cholerae isolates from 9 fish and 1 fish tank water samples and did not detect virulence genes (tdh, trh1, and trh2) in any of the 6 V. parahaemolyticus isolates from 5 fish samples.
We found GBS ST283, the causative strain of a severe foodborne outbreak in Singapore, in the local freshwater fish supply chain that stretches from food stalls to local fresh produce markets and back to ports. Patients with GBS ST283 infections during this outbreak were more likely to show development of meningoencephalitis, septic arthritis, and spinal infection than were persons with non–GBS ST283 infections (12). Although this study suggested Malaysia as a source of the strain, the finding of the same ST in Hong Kong and Thailand (10,11) suggested that GBS ST283 is generally prevalent throughout the region.
Our analysis shows that there are at least 2 clades of GBS ST283 strains among fish in local markets. Fish and water strains from 1 clade were nearly identical to clinical strains from this outbreak (Figure 4). The small variability of 0–2 SNPs and 0–12 indels between fish and water strains and the reference human outbreak strain (SG-M1) is equivalent to variability observed in 131 clinical strains from the same outbreak reported elsewhere (0–5 SNPs from the SG-M1 reference) (12). Strains from a second clade of GBS ST283 had a difference of 57–71 SNPs and 11–33 indels when compared with the SG-M1 genome. Other GBS ST283 isolates, many collected in Hong Kong <17 years before this outbreak (11) are also different from the SG-M1 strain (<129 SNPs) (Figure 4). We found no human-infecting isolate from Singapore or elsewhere within the second fish-associated GBS ST283 clade.
A major issue is whether all GBS ST283 strains are capable of causing invasive human disease by the foodborne route. If strains from the fish-associated clade are not pathogenic to humans, they could be used as effective controls for identifying the genetic basis of pathogenicity of the first clade and the cause of its emergence, which resulted in outbreak in Singapore in 2015. If these strains are pathogenic to humans, then broader tracking of the prevalence of GBS ST283 would be warranted.
In contrast to GBS strains that are known to cause disease outbreaks in fish (10,18), the live and whole fish from which GBS ST283 was recovered in this study did not have defects, such as corneal opacity and exophthalmia (18), which suggests that this ST might not be pathogenic for freshwater fish. The closest GBS fish pathogens with published genomes, GD201008–001 (19) and HN016 (20), are serotype Ia ST7 strains that are distant (>4,000 SNPs) from all ST283 strains that our group and others have identified (12).
Detection of 6 GBS serotypes and 13 STs showed the diversity of GBS strains in fish. Although the sample size in this study was small and our results might not represent the distribution of GBS in all fish species, our findings provide valuable data for characterizing the public health risk from consuming raw fish. No baseline information on GBS in fish was publicly available before this outbreak because fish were not a recognized source or a recognized route of transmission of GBS. Further work on GBS STs other than ST283 is underway to investigate the role of fish as a source of GBS disease in humans.
Several GBS strains from sashimi had relatively few closely related strains in the public genomic databases (Technical Appendix Figure 2), which suggests that the GBS population associated with saltwater fish could be different from that associated with freshwater fish and humans. Another reason for this observation is that GBS from food and environmental sources are relatively undersampled in the genomic databases than those from humans.
We detected GBS serotypes Ia ST23 and Ia ST7, which are associated with human carriage (10), in fish samples. Although GBS ST7 has been described as a fish pathogen, the presence of GBS serotype Ia ST23 has not been reported in fish (10). GBS serotypes Ia ST23, and Ia ST7 and E. coli, which are all associated with human gut flora, suggest possible contamination of fish by effluent water.
The intentional introduction of animal feces into fish ponds as part of integrated farming (21,22) might further contribute to the complex flow of pathogens between animals and humans. Such findings point to areas for research to clarify the diversity and role of GBS strains in affecting animal and human health. For instance, GBS ST861, which was isolated from salmon in this study (Technical Appendix Table 3), was also isolated from a clinical case in the same year in Singapore on the basis of metadata available in the PubMLST S. agalactiae database (http://pubmlst.org/sagalactiae/).
In addition to the finding of GBS ST283 in freshwater fish, detection of high SPCs and E. coli and S. aureus counts indicates the hazard of using such fish for preparing raw RTE dishes. Because E. coli is not part of the intestinal flora of cold-blooded animals (23), its presence suggests contamination from polluted water, unhygienic handling, or temperature abuse after harvesting. Similarly, because S. aureus is not part of usual fish flora, its presence on fish surfaces suggests possible transfer of human skin flora caused by unhygienic handling (24). We detected V. parahaemolyticus, an organism known to grow well in seawater but lyse rapidly in freshwater (24), in freshwater fish samples from fresh produce markets (5.1%, 2/39). This finding was not surprising because freshwater and saltwater fish are typically sold, handled, stored, and degutted within the same confined areas in fresh produce markets. Thus, despite lower SPCs and positive rates for E. coli in saltwater fish than in freshwater fish from fresh produce markets, saltwater fish procured from such environments are prone to cross-contamination.
Fish used by food stalls were generally obtained from such markets. Moreover, microbial counts for sliced fish samples from eating establishments indicated that most food stalls were not able to prepare RTE raw fish dishes of acceptable hygiene quality. Poor practices observed included use of common chopping boards, knives, or slicers for preparing fish slices meant for raw consumption and cooking. If fish slices are contaminated, rinsing with water cannot improve their quality (Technical Appendix).
In contrast to the quality of saltwater fish samples from fresh produce markets, all saltwater fish samples from sashimi suppliers complied with local SPCs (5 log10 CFU/g) and E. coli (1.3 log10 CFU/g) limits for ready-to-eat food (14); all samples were negative for GBS, S. aureus, Salmonella spp., and V. parahaemolyticus. The compliance rate among restaurants/snack bars was higher because such premises are more likely to procure fish from sashimi suppliers that harvest fish from cleaner waters and adhere to stricter cold chain management practices. However, some saltwater fish samples from sashimi suppliers and restaurants were found to contain Aeromonas spp. (47.6%, 10/21) and L. monocytogenes (2.0%, 6/303), whose psychrotrophic nature has posed a challenge to the fish industry. L. monocytogenes is also a concern in chilled RTE food because of its ubiquity and persistence in food-processing environments (25).
Food and environmental findings of our study were consistent with epidemiologic findings for this outbreak (2,3). Multivariate analyses of a case–control study showed that persons who had consumed yusheng were more likely to acquire GBS ST283 infections than those who had not consumed yusheng (2). However, there was no strong association between GBS ST283 infections and consumption of sashimi and sushi (2).
Findings of this study have led to implementation of new policies in Singapore. These new policies included banning the use of freshwater fish in RTE dishes and requiring procurement of saltwater fish from suppliers for raw fish approved by the Agri-Food and Veterinary Authority of Singapore. Food stalls and food establishments providing catering services were required to stop selling RTE raw fish dishes until they complied with practices required for preparing RTE raw saltwater fish dishes.
The number of RTE fish samples collected from food stalls was limited because eating establishments were advised to stop the sale of RTE raw fish dishes containing Asian bighead carp and snakehead during July 24–December 5, 2015, while the outbreak investigation was underway (1). Sampling was not random because it was part of an outbreak investigation, but it was biased toward fish species and food stalls implicated in the outbreak. Thus, contamination rates might not reflect contamination rates of all fish species sold for raw consumption in the market. Similarly, testing of samples from ports and retail outlets was performed by using different protocols, which limited comparisons that could be made.
In conclusion, we detected GBS ST283, which caused a severe foodborne outbreak in Singapore in 2015, in freshwater fish, not only in food stalls and fish markets, but also in ports from which fish are imported. Comparison of human and fish isolates showed as few as 0–2 SNPs between human and fish isolates of GBS ST283 on a background of a diversity of GBS and other bacteria in freshwater fish. These data indicate the risk for contamination of raw freshwater fish and underscore the need for proper sourcing and handling of all fish for raw consumption. To control the outbreak, a ban on the sale of RTE raw freshwater fish dishes was implemented, and additional requirements were imposed for the sale of RTE raw fish dishes made with saltwater fish (13). Our study complements the epidemiologic findings for this outbreak (2,3) and illustrates the need for public health authorities and industries to remain vigilant regarding emerging pathogens.
Ms. Chau is research group leader of the Applied Food Hygiene Epidemiology Group, Food and Environmental Hygiene Research Program, Environmental Health Institute, National Environment Agency, Singapore. Her research interests include microbiology, food hygiene and safety, and foodborne diseases.
Acknowledgments
We thank the Agri-Food and Veterinary Authority of Singapore, the National Environment Agency, and the Ministry of Health of Singapore for providing operational support and Lyn Gilbert and Fanrong Kong for providing the GBS serotype VIII reference strain.
Analyses of fish and fish tank water samples was supported by the National Environment Agency and the Agri-Food and Veterinary Authority of Singapore. Whole-genome sequencing and analysis was supported by the Genome Institute of Singapore, the Agency for Science, Technology, and Research, and the Singapore Ministry of Health National Medical Research Council (NMRC/CIRG/1357/2013).
References
- Joint news release between the Ministry of Health Singapore (MOH) and the Agri-Food and Veterinary Authority of Singapore (AVA) and the National Environment Agency (NEA). Update on investigation into group B Streptococcus cases. July 24, 2015 [cited 2016 Aug 10]. http://www.nea.gov.sg/corporate-functions/newsroom/news-releases/year/2015/month/7/category/food-hygiene/update-on-investigation-into-group-b-streptococcus-cases
- Tan S, Lin Y, Foo K, Koh HF, Tow C, Zhang Y, et al. Group B Streptococcus serotype III sequence type 283 bacteremia associated with consumption of raw fish, Singapore. Emerg Infect Dis. 2016;22:1970–3. DOIPubMedGoogle Scholar
- Rajendram P, Mar Kyaw W, Leo YS, Ho H, Chen WK, Lin R, et al. Group B Streptococcus sequence type 283 disease linked to consumption of raw fish, Singapore. Emerg Infect Dis. 2016;22:1974–7. DOIPubMedGoogle Scholar
- Tan K, Wijaya L, Chiew HJ, Sitoh YY, Shafi H, Chen RC, et al. Diffusion-weighted MRI abnormalities in an outbreak of Streptococcus agalactiae Serotype III, multilocus sequence type 283 meningitis. J Magn Reson Imaging. 2017;45:507–14. DOIPubMedGoogle Scholar
- Manning SD, Neighbors K, Tallman PA, Gillespie B, Marrs CF, Borchardt SM, et al. Prevalence of group B streptococcus colonization and potential for transmission by casual contact in healthy young men and women. Clin Infect Dis. 2004;39:380–8. DOIPubMedGoogle Scholar
- Public Health Agency of Canada. Streptococcus agalactiae: pathogen safety data sheet—infectious substances. April 30, 2012 [cited 2016 Aug 10]. http://www.phac-aspc.gc.ca/lab-bio/res/psds-ftss/streptococcus-agalactiae-eng.php
- Ballard MS, Schønheyder HC, Knudsen JD, Lyytikäinen O, Dryden M, Kennedy KJ, et al.; International Bacteremia Surveillance Collaborative. The changing epidemiology of group B streptococcus bloodstream infection: a multi-national population-based assessment. Infect Dis (Lond). 2016;48:386–91. DOIPubMedGoogle Scholar
- Phares CR, Lynfield R, Farley MM, Mohle-Boetani J, Harrison LH, Petit S, et al.; Active Bacterial Core surveillance/Emerging Infections Program Network. Epidemiology of invasive group B streptococcal disease in the United States, 1999-2005. JAMA. 2008;299:2056–65. DOIPubMedGoogle Scholar
- Foxman B, Gillespie BW, Manning SD, Marrs CF. Risk factors for group B streptococcal colonization: potential for different transmission systems by capsular type. Ann Epidemiol. 2007;17:854–62. DOIPubMedGoogle Scholar
- Delannoy CM, Crumlish M, Fontaine MC, Pollock J, Foster G, Dagleish MP, et al. Human Streptococcus agalactiae strains in aquatic mammals and fish. BMC Microbiol. 2013;13:41. DOIPubMedGoogle Scholar
- Ip M, Cheuk ES, Tsui MH, Kong F, Leung TN, Gilbert GL. Identification of a Streptococcus agalactiae serotype III subtype 4 clone in association with adult invasive disease in Hong Kong. J Clin Microbiol. 2006;44:4252–4. DOIPubMedGoogle Scholar
- Kalimuddin S, Chen SL, Lim CTK, Koh TH, Tan TY, Kam M, et al. Singapore Group B Streptococcus Consortium. 2015 epidemic of severe Streptococcus agalactiae ST283 infections in Singapore associated with the consumption of raw freshwater fish: a detailed analysis of clinical, epidemiological and bacterial sequencing data. Clin Infect Dis. 2017;64(Suppl_2):S145–52.
- Joint news release between the Agri-Food and Veterinary Authority of Singapore (AVA) and the Ministry of Health Singapore (MOH) and the National Environment Agency (NEA). Freshwater fish banned from ready-to-eat raw fish dishes. December 5, 2015 [cited 2016 Aug 10]. http://www.nea.gov.sg/corporate-functions/newsroom/news-releases/freshwater-fish-banned-from-ready-to-eat-raw-fish-dishes
- Agri-Food and Veterinary Authority of Singapore. Sale of food act, chapter 283, section 56 (1), food regulations. December 20, 2016 [cited 2017 Jan 9]. http://www.ava.gov.sg/legislation
- British Columbia Centre for Disease Control. Food processing plants: guideline for live retail fish holding system. November 2013 [cited 2016 Aug 17]. http://www.bccdc.ca/resource-gallery/Documents/Educational%20Materials/EH/FPS/Food/RetailFishHoldingTankGuidelines_Nov2013trs.pdf
- Da Cunha V, Davies MR, Douarre PE, Rosinski-Chupin I, Margarit I, Spinali S, et al.; DEVANI Consortium. Streptococcus agalactiae clones infecting humans were selected and fixed through the extensive use of tetracycline. Nat Commun. 2014;5:4544. DOIPubMedGoogle Scholar
- Mehershahi KS, Hsu LY, Koh TH, Chen SL. Complete genome sequence of Streptococcus agalactiae serotype III, multilocus sequence type 283 strain SG-M1. Genome Announc. 2015;3:e01188–15.PubMedGoogle Scholar
- Abuseliana AF, Daud HH, Aziz SA, Bejo SK, Alsaid M. Pathogenicity of Streptococcus agalactiae isolated from a fish farm in Selangor to juvenile red tilapia (Oreochromis sp.). J Anim Vet Adv. 2011;10:914–9. DOIGoogle Scholar
- Liu G, Zhang W, Lu C. Complete genome sequence of Streptococcus agalactiae GD201008-001, isolated in China from tilapia with meningoencephalitis. J Bacteriol. 2012;194:6653. DOIPubMedGoogle Scholar
- Wang R, Li L, Huang Y, Luo F, Liang W, Gan X, et al. Comparative genome analysis identifies two large deletions in the genome of highly-passaged attenuated Streptococcus agalactiae strain YM001 compared to the parental pathogenic strain HN016. BMC Genomics. 2015;16:897. DOIPubMedGoogle Scholar
- Martins C, Eding EH, Verdegem MC, Heinsbroek LT, Schneider O, Blancheton J-P, et al. New developments in recirculating aquaculture systems in Europe: a perspective on environmental sustainability. Aquacult Eng. 2010;43:83–93. DOIGoogle Scholar
- Little D, Edwards P. Integrated livestock-fish farming systems. Rome: Food and Agriculture Organization of the United Nations; 2003 [cited 2017 Aug 15]. http://www.fao.org/docrep/006/y5098e/y5098e00.htm
- International Commission on Microbiological Specifications for Foods. Micro-organisms in foods 2: sampling for microbiological analysis; principles and specific applications. 2nd ed. 1986 [cited 2016 Aug 11]. http://www.icmsf.org/pdf/icmsf2.pdf
- Food and Drug Administration. Bad bug book: foodborne pathogenic microorganisms and natural toxins. 2nd ed. Silver Spring (MD): The Administration; 2012.
- Chau ML, Aung KT, Hapuarachchi HC, Lee PSV, Lim PY, Kang JSL, et al. Microbial survey of ready-to-eat salad ingredients sold at retail reveals the occurrence and the persistence of Listeria monocytogenes Sequence Types 2 and 87 in pre-packed smoked salmon. BMC Microbiol. 2017;17:46. DOIPubMedGoogle Scholar
Figures
Table
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to http://www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers.
You must be a registered user on http://www.medscape.org. If you are not registered on http://www.medscape.org, please click on the “Register” link on the right hand side of the website.
Only one answer is correct for each question. Once you successfully answer all post-test questions, you will be able to view and/or print your certificate. For questions regarding this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@medscape.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please go to https://www.ama-assn.org. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the AMA PRA CME credit certificate, and present it to your national medical association for review.
Article Title:
Group B Streptococcus Infections Caused by Improper Sourcing
and Handling of Fish for Raw Consumption, Singapore, 2015–2016
CME Questions
1. You are advising a public health department in Singapore regarding the potential for future human group B Streptococcus (GBS) outbreak. On the basis of the microbial study of raw fish and human samples by Chau and colleagues, which one of the following statements about the clinical and epidemiological findings of a human GBS outbreak in Singapore is correct?
A. The outbreak was associated with consumption of raw saltwater fish
B. The source of the outbreak was limited to restaurants
C. The causative agent was Streptococcus viridans
D. Severe clinical cases included meningoencephalitis, bacteremia, spinal infection and septic arthritis
2. According to the microbial study of raw fish and human samples by Chau and colleagues, which one of the following statements about the pathogenic potential of fish and human samples after a human GBS outbreak in Singapore is correct?
A. Fish-associated GBS ST283 strains included only strains nearly identical to the human outbreak strain
B. The study proves that all GBS ST283 strains are capable of causing invasive human disease through the foodborne route
C. The live and whole fish from which GBS ST283 was recovered did not show corneal opacity or exophthalmia, suggesting this strain may not be pathogenic to freshwater fish
D. The human outbreak strain is highly similar to known GBS fish pathogens with published genomes
3. According to the microbial study of raw fish and human samples by Chau and colleagues, which one of the following statements about public health implications of a human GBS outbreak in Singapore is correct?
A. Both saltwater and freshwater fish are suitable for raw consumption if proper sourcing and handling standards are followed
B. To control the outbreak, the sale of ready-to-eat (RTE) raw freshwater fish dishes was banned, and additional requirements were imposed for sale of RTE raw fish dishes using saltwater fish
C. GBS ST283 was the only hazard identified in freshwater fish used to prepare raw RTE dishes
D. Most food stalls tested were able to prepare RTE raw fish dishes of acceptable hygiene quality
1These authors contributed equally to this article.
Related Links
Table of Contents – Volume 23, Number 12—December 2017
| EID Search Options |
|---|
|
|
|
|
|
|



Please use the form below to submit correspondence to the authors or contact them at the following address:
Lee C. Ng, Environmental Health Institute, National Environment Agency, 11 Biopolis Way, #06-05/08, Helios Block, Singapore 138667, Singapore
Top