Volume 24, Number 7—July 2018
Perspective
Effects of Sexual Network Connectivity and Antimicrobial Drug Use on Antimicrobial Resistance in Neisseria gonorrhoeae
Connectivity–AMR Thesis—Combination of High Prevalence of Antimicrobial Use and Network Connectivity as Cause of Resistance
Effects of N. gonorrhoeae Screening on AMR
Future Evaluation of the Connectivity–AMR Hypothesis
Allodemics of Resistance in MSM
Conclusions
Cite This Article
Cite This Article
Citation for Media
Abstract
Contemporary strategies to curtail the emergence of antimicrobial resistance in Neisseria gonorrhoeae include screening for and treating asymptomatic infections in high-prevalence populations in whom antimicrobial drug–resistant infections have typically emerged. We argue that antimicrobial resistance in these groups is driven by a combination of dense sexual network connectivity and antimicrobial drug exposure (for example, through screen-and-treat strategies for asymptomatic N. gonorrhoeae infection). Sexual network connectivity sustains a high-equilibrium prevalence of N. gonorrhoeae and increases likelihood of reinfection, whereas antimicrobial drug exposure results in selection pressure for reinfecting N. gonorrhoeae strains to acquire antimicrobial resistance genes from commensal pharyngeal or rectal flora. We propose study designs to test this hypothesis.
The rapid emergence of antimicrobial resistance (AMR) in Neisseria gonorrhoeae has led to fears that gonorrhea may soon become untreatable (1). An incompletely explained feature of the emergence of AMR in N. gonorrhoeae is its repeated emergence in core groups (2). As Lewis noted, AMR first emerged in core groups of sex workers in East Asia and elsewhere from the 1960s onward (2). In the past 3 decades, however, AMR has repeatedly emerged in men who have sex with men (MSM) (2,3). In both the United States and the United Kingdom, N. gonorrhoeae resistant to several antimicrobial drugs emerged in MSM years ahead of men who have sex with women (MSW) (Figure 1).
Several explanations have been proposed for this observation (4). Excess use of antimicrobial drugs is a possibility. One study found that MSM with a diagnosis of gonorrhea were more likely to report recent antimicrobial drug use than were MSW. After controlling for antimicrobial drug use, however, MSM remained at significantly higher risk for N. gonorrhoeae with resistance to all classes of antimicrobial drugs tested (p<0.001 for all) (3). Higher prevalence of HIV, which has been associated with various types of AMR in some studies, could also cause elevated resistance rates in MSM groups (5).
We focus on the emergence of N. gonorrhoeae AMR in MSM and hypothesize that the combination of high sexual network connectivity and excess antimicrobial drug use plays an important role in AMR genesis. This connectivity–AMR hypothesis proceeds in 2 steps: first, that high-equilibrium prevalence of N. gonorrhoeae in contemporary MSM populations is a function of densely connected sexual networks; and second, that extensive antimicrobial drug use (such as with STI screening and treatment) may temporarily reduce N. gonorrhoeae prevalence in this setting but produce selection pressure for N. gonorrhoeae to acquire AMR.
STIs are transmitted along sexual networks, and as a result, the equilibrium prevalences of these STIs are determined by structural characteristics of these networks (6,7). These characteristics include the number of partners per unit time, prevalence of concurrent partnering, size of core groups, type of sex, size of sexual network, length of gaps between partnerships, degree and type of homophily (preference for partners with similarities to oneself), and relationships between core and noncore groups (7). Combinations of these attributes should result in higher network connectivity in some populations than in others (6,7). Studies have found a correlation between markers of network connectivity and the prevalence of various major STIs (7), including N. gonorrhoeae (8,9). STI prevalence can also be influenced by other risk factors that can affect the probability of transmission per contact (such as male circumcision, condom use, and presence of other STIs) or the duration of infectivity (such as STI early detection and treatment efficacy) (10). Although the relative contributions of these risk factors to STI prevalence vary considerably among populations, a consistent feature of contemporary STI epidemics in MSM populations in numerous countries is their association with dense sexual networks (11,12). For instance, nationally representative data from the United Kingdom, United States, and Australia reveal that MSM report considerably more sexual partners per unit of time than do heterosexual men (Table 1). An example of the prevalence of multiple partnering in MSM is given by >180,000 participants in the European Men Who Have Sex with Men Internet Survey (13); 67% of respondents reported a nonsteady partner in the past year, and 37.7% of respondents reported >10 partners in the past year. These high rates of partner change, combined with high rates of partner concurrency (14) and other determinants of network connectivity, translate into dense networks (7). Network connectivity is particularly dense in preexposure prophylaxis (PrEP) cohorts, in which the median number of sex partners typically exceeds 10 per 90 days. In the iPrEx study, for example, participants reported a mean of 18 partners (SD ± 35) in the preceding 90 days (15). The resulting dense network typically sustains equilibrium prevalences of both N. gonorrhoeae and Chlamydia trachomatis at >10% (15). In comparison, the prevalence of N. gonorrhoeae in the general heterosexual population in the United Kingdom is estimated at <0.1% and of C. trachomatis at 1.3% (16).
Connectivity–AMR Thesis—Combination of High Prevalence of Antimicrobial Use and Network Connectivity as Cause of Resistance
In the absence of an antimicrobial selection pressure, we would not expect a high prevalence of N. gonorrhoeae to lead to AMR (17,18). Under such pressure, however, N. gonorrhoeae has developed AMR to each antimicrobial therapy introduced to treat infections, often within as few as 3 years (1,19,20). This effect is similar to the rapid development of AMR observed in a range of other bacteria (21). Individual-level studies have also found recent antimicrobial drug use to be a risk factor for AMR in N. gonorrhoeae (3,22,23). We would thus expect higher rates of antimicrobial drug use to be a risk factor for the emergence of AMR. Four mechanisms have been proposed to explain this antimicrobial drug–induced selection of AMR in bacteria for which, as for N. gonorrhoeae, horizontal gene transfer is a major mechanism of AMR acquisition (Table 2) (1,24). We argue that high network connectivity coupled with antimicrobial exposure constitutes an emergent fifth pathway to AMR in N. gonorrhoeae.
Figure 2 is a schematic representation of a PrEP MSM cohort with a dense sexual network and quarterly N. gonorrhoeae and C. trachomatis screening. In the absence of a global screen-and-treat strategy that leads to N. gonorrhoeae extinction, a typical local screen-and-treat approach induces a temporary decline in N. gonorrhoeae prevalence. Without altering the underlying determinant of high N. gonorrhoeae prevalence (network connectivity), N. gonorrhoeae tends to return to its high-equilibrium prevalence. Moreover, this strategy also increases the prevalence of the genes that encode AMR in N. gonorrhoeae. On the basis of studies using macrolides for other indications, ≈90% of patients treated with ceftriaxone and azithromycin, the currently recommended therapy for N. gonorrhoeae infection, would be expected to acquire macrolide resistance that can persist for up to 4 years in commensal pharyngeal and colonic bacteria (25,26). Recently treated patients are also at high risk for early reinfection because it is unlikely that their whole local sexual network has been effectively screened via partner tracing (27).
N. gonorrhoeae has a highly developed system of transformation to take up DNA from its environment, particularly from other Neisseria spp. (28), which, along with other mechanisms, may lead to AMR acquisition (1). Studies have established that transformation is a method by which N. gonorrhoeae acquired resistance to cefixime from commensal pharyngeal Neisseria species (19,24).
Further examples of the connectivity–AMR mechanism come from various branches of high-density animal husbandry in which antimicrobial drug–based strategies used alone to combat epidemics have led to the induction of AMR (29). Norwegian salmon farms, for example, contain roughly 200,000 salmon per pen (with population densities of <25 kg/m3) and consequently are prone to outbreaks of various bacterial, viral, and parasitic diseases (29,30). Initially, these epizootic infections were controlled predominantly with prophylactic and therapeutic antimicrobial drugs, but the bacterial and ectodermal pathogens rapidly developed resistance (29). Consequently, zoo sanitation (increased separation and fallowing of the fish) and vaccination were introduced, which allowed a decrease of antimicrobials used from 48 tons to 1 ton annually while reducing the number and severity of outbreaks and increasing the total salmon harvest (29). Studies from other sites have linked declines in antimicrobial drug use to declines in AMR in salmon-associated infections (31).
We acknowledge that there is conflicting evidence of whether an excess use of antimicrobial drugs results in AMR. A recent ecologic analysis from the United States, for example, found no association between antimicrobial prescribing and gonococcal AMR in 23 STI clinics (32).
Theoretically, a sufficiently intense and synchronized global screen-and-treat program could lead to the extinction of N. gonorrhoeae. However, if the screening program falls short of complete eradication, and if a combination of high network connectivity and antimicrobial drug exposure is responsible for AMR, then paradoxically the more effective the screening program is at decreasing prevalence, the greater this AMR selection pressure would be. This conclusion is at odds with current initiatives to enhance N. gonorrhoeae screening in MSM and other high–N. gonorrhoeae prevalence populations. Screening for N. gonorrhoeae every 3–12 months is typically recommended in clinical guidelines for sexually active MSM (33). A notable exception to these guidelines is that from the US Preventive Service Task Force, which concluded that the absence of randomized controlled trials (RCTs) evaluating the merits of screening in men precluded recommendations on the matter (34). In MSM-PrEP programs, screening is recommended every 3–6 months (35). Longitudinal analyses of PrEP studies typically show high N. gonorrhoeae prevalences that do not decline despite frequent screening (15,35,36). A recent PrEP study found that the prevalence of N. gonorrhoeae remained static in the pharynx and rectum and increased in the urethra despite quarterly screening (35). Modeling studies have found that increasing screening intensity in MSM populations results in either a modest (37) or dramatic (38) reduction in N. gonorrhoeae prevalence.
Some authors have gone further and argued that screening is an important component for containing AMR emergence in N. gonorrhoeae (2,20). A recent paper on this topic for example outlined the argument as follows: “Gonococcal AMR will only be effectively mitigated when the global gonorrhea burden is reduced. Increased detection and effective treatment of asymptomatic gonorrhea in general and pharyngeal gonorrhea in particular are critical, because these infections are potential gonococcal reservoirs in which AMR (especially extended spectrum cephalosporin AMR) can emerge. Oropharyngeal infections are prevalent, mostly asymptomatic, and more difficult to treat; accordingly, screening and treatment in high-risk patients are important” (20). For similar reasons, the World Health Organization (WHO) has made the early detection and treatment of asymptomatic N. gonorrhoeae a key component of its plan to reduce the prevalence of N. gonorrhoeae infection by 90% by 2030 as well as N. gonorrhoeae AMR (39).
Increasing screening of high-risk patients to combat AMR is diametrically opposed to our connectivity–AMR thesis. Given the stakes involved (including untreatable infections), establishing the validity of the connectivity–AMR hypothesis in general and the place of screening in high-prevalence populations specifically is imperative. Part of the answer lies in accurately describing the mechanisms underpinning AMR in N. gonorrhoeae compared with other organisms. In some pathogens, such as Mycobacterium tuberculosis, resistance emerges primarily through mutations during treatment in hosts (18). For these pathogens, screening and treating infected persons is crucial for containing the spread of AMR (18). For other bacteria, such as Streptococcus pneumonia, Enterococcus spp., Staphylococcus aureus, and N. gonorrhoeae, horizontal gene transfer is the predominant means of acquisition of AMR (40). For these bacteria, AMR is driven predominantly by indirect population-level mechanisms of selection (Table 2) (18). Although screening for these organisms may reduce prevalence, it may also increase AMR by these indirect mechanisms. Two types of study could assess the connectivity–AMR thesis and the net benefits and harms of N. gonorrhoeae screening programs in MSM: RCTs and modeling studies.
Community RCTs in high-connectivity populations, including MSM who are taking PrEP, could assess the effect of N. gonorrhoeae screening and treatment (vs. no screening and limiting therapy to patients with symptomatic N. gonorrhoeae infection) on several parameters: prevalence of N. gonorrhoeae infection; susceptibility to other STIs, including HIV; effect on adaptive immunity to N. gonorrhoeae; effect on individual and population resistome and microbiome; and emergence of AMR. Researchers could increase the probability of reducing N. gonorrhoeae prevalence in these studies by including aggressive contact tracing strategies, such as by using sexual networking apps. A practical challenge would be the large cohort size required to demonstrate a difference in the probability of AMR between the screening and no-screening arms of the study because AMR emergence is a rare event. Nonetheless, establishing whether N. gonorrhoeae screening reduces infection prevalence in dense networks and at what cost to the resistome (individual and population) would be informative. If screening is found to have little or no effect on N. gonorrhoeae prevalence but a large effect on the population resistome, it may cause a reevaluation of screening policies. Researchers could also assess the significance of altered resistomes to AMR in N. gonorrhoeae in vitro by assessing whether N. gonorrhoeae is able to acquire AMR via transformation with DNA extracts from posttreatment microbiomes. Such studies could also provide the probabilities of resistome alteration following specific therapies (including the decay curves of these alterations). These data could then be used to construct more realistic models of AMR induction in N. gonorrhoeae (41).
Recent modeling studies have found that screening high-connectivity MSM populations could reduce N. gonorrhoeae prevalence by ≈50% (38), but at the expense of an 11-fold increase in antimicrobial drug exposure (37). Few studies have evaluated the effect of screening on the emergence of AMR. One such study found evidence of a screening paradox: although screening the core group was crucial to reduce prevalence of N. gonorrhoeae, this strategy involved the highest risk of inducing AMR (42). However, that study used a compartmental model of the underlying sexual network and examined only 1 type of N. gonorrhoeae AMR, chromosomally mediated AMR (42). Future models that evaluate the probability of AMR emergence should use individual-based models that can model AMR via horizontal gene transfer. These models could assess if the combination of high connectivity and antimicrobial exposure more likely to produce and disseminate AMR than the combination of low connectivity and high antimicrobial exposure or of high connectivity and low antimicrobial exposure. Using models could also help establish the level of intensity required of screen-and-treat programs for highly connected sexual networks to reduce the prevalence of N. gonorrhoeae to a level with minimal risk for reinfection during the period when the resistomes of treated persons are altered. Our discussion has focused on MSM populations taking PrEP, but similar arguments would apply to other segments of the MSM sexual network, such as HIV-infected MSM who are excluded from PrEP programs and MSM without HIV infection who are not taking PrEP. Modeling studies could explore how differential network connectivity and antimicrobial drug exposure in different sections of MSM sexual networks may interact to produce AMR.
The connectivity–AMR theory makes 2 other predictions regarding AMR in MSM. The first is that N. gonorrhoeae will become resistant to the full range of antimicrobial drugs to which the population is exposed. In accordance with the connectivity–AMR theory, any widely used antimicrobial drug that reduces N. gonorrhoeae prevalence in MSM populations is at risk for AMR. Although not all studies have reached the same conclusion, the data from the national N. gonorrhoeae surveillance projects in the United States and United Kingdom have generally found this to be true (3,43). In the United States, for example, MSM were statistically more likely to have AMR N. gonorrhoeae for all classes of antimicrobial drugs tested (3).
The second prediction is that AMR in other bacterial STIs will be likely to emerge or to become more prevalent in MSM. Although the link is not as clearly established as with N. gonorrhoeae, there is some evidence that this is the case. For example, in the United States and Australia, macrolide resistance in Treponema pallidum first emerged in predominantly MSM populations at roughly the same time as azithromycin resistance in N. gonorrhoeae (44,45). In addition, the prevalence of macrolide resistance in Mycoplasma genitalium in MSM in 1 Australia study was found to be approximately double that of heterosexual men (46). Outbreaks have occurred in MSM of macrolide- or quinolone-resistant sexually transmissible enteric organisms Shigella spp. (47) and Campylobacter spp. (48). One phylogenetic analysis of Shigella flexneri infections from 29 countries concluded that the 3a serotype had emerged and acquired multiple AMR mutations while circulating sexually in international MSM sexual networks characterized by high rates of reinfection with this same serotype (47). Outbreaks of sexually transmitted methicillin-resistant Staphylococcus aureus have also been described in MSM (49).
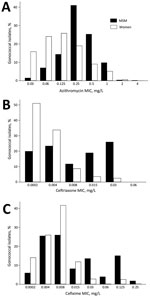
The variations in population shifts of N. gonorrhoeae MICs to various antimicrobial drugs by sexual orientation are also compatible with the connectivity–AMR thesis. The earliest available data for N. gonorrhoeae sensitivity by sexual orientation from the United Kingdom reveal that MSM have a higher proportion of N. gonorrhoeae with high MICs than women do (Figure 3). Furthermore, the evolution of N. gonorrhoeae MIC distributions in MSM from 2010–2015 reveals a right shifting of the whole distribution curve, indicating a reduction in the proportion of MSM with low MICs (Figure 4).
Considered together, these findings support the hypothesis that the problem of AMR in MSM could constructively be viewed as an allodemic of AMR. Baquero et al. first introduced this term to describe how the spread of extended spectrum β lactamase (ESBL)–producing bacteria in a hospital in Spain was best described by an increase in ESBL production in a range of bacteria (an allodemic) rather than epidemics of single species or clones (50). They argued that appreciating this polyclonal spread of resistance as an allodemic enabled them to address the underlying environmental determinant of AMR: excess use of antimicrobial drugs that induce ESBL production in multiple bacteria species rather than traditional approaches targeting individual clones or species (50). The problem of polyclonal AMR in MSM may likewise benefit from efforts to address the underpinning environmental determinants.
Although we have focused our discussion on the connectivity–AMR thesis in MSM, similar considerations would also apply to other high connectivity populations. The emergence of N. gonorrhoeae AMR in sex workers, for example, has been linked to extensive antimicrobial drug use (2). Various studies have also concluded that high rates of STIs in various populations in sub-Saharan Africa are underpinned by dense sexual networks (7). In keeping with WHO directives, interventions are being planned to detect and treat asymptomatic STIs in South Africa and elsewhere. If the connectivity–AMR thesis applies to these populations, then due caution should be exercised if screening and antimicrobial drug use are used to reduce STI prevalence. In high-connectivity populations, particular consideration should be given to the use of nonantimicrobial STI therapies such as local disinfectants (e.g., for pharyngeal STIs), bacteriophage therapy, and vaccines. If antimicrobial drugs are used, research is required to guide their selection on the basis of efficacy and resistogenicity of therapies. Genotypic resistance profiling before therapy could also be considered. If STI prevention and control programs are unable to attain the level of screen-and-treat coverage required to eradicate STIs (or make negligible the risk for reinfection during the period of posttreatment resistome alteration), then they should prioritize STI reduction strategies that minimize the risk for AMR selection. These strategies would include methods to fragment sexual network connectivity (e.g., through decreasing rates of partner change) and treat STIs with nonantimicrobial therapies (e.g., bacteriophages and antiseptics).
Dr. Kenyon is a professor and head of the STI/HIV unit of the Institute of Tropical Medicine. His research interests include AMR in STIs and the effects of sexual networks.
Dr. Schwartz is an assistant professor at the University of Alberta in the Department of Medicine, Division of Infectious Diseases. His research interests include invasive fungal disease and global health.
References
- Unemo M, Del Rio C, Shafer WM. Antimicrobial resistance expressed by Neisseria gonorrhoeae: a major global public health problem in the 21st century. Microbiol Spectr. 2016;4:4.PubMedGoogle Scholar
- Lewis DA. The role of core groups in the emergence and dissemination of antimicrobial-resistant N gonorrhoeae. [Erratum in: Sex Transm Infect 2014;90:400.]. Sex Transm Infect. 2013;89(Suppl 4):iv47–51. DOIPubMedGoogle Scholar
- Kirkcaldy RD, Zaidi A, Hook EW III, Holmes KK, Soge O, del Rio C, et al. Neisseria gonorrhoeae antimicrobial resistance among men who have sex with men and men who have sex exclusively with women: the Gonococcal Isolate Surveillance Project, 2005-2010. [Erratum in: Ann Intern Med. 2013;159:372.]. Ann Intern Med. 2013;158:321–8. DOIPubMedGoogle Scholar
- Kenyon C, Osbak K. Certain attributes of the sexual ecosystem of high-risk MSM have resulted in an altered microbiome with an enhanced propensity to generate and transmit antibiotic resistance. Med Hypotheses. 2014;83:196–202. DOIPubMedGoogle Scholar
- Popovich KJ, Hota B, Aroutcheva A, Kurien L, Patel J, Lyles-Banks R, et al. Community-associated methicillin-resistant Staphylococcus aureus colonization burden in HIV-infected patients. Clin Infect Dis. 2013;56:1067–74. DOIPubMedGoogle Scholar
- Aral SO, Leichliter JS, Blanchard JF. Overview: the role of emergent properties of complex systems in the epidemiology and prevention of sexually transmitted infections including HIV infection. Sex Transm Infect. 2010;86(Suppl 3):iii1–3. DOIPubMedGoogle Scholar
- Morris M, Goodreau S, Moody J. Sexual networks, concurrency, and STD/HIV. In: Holmes KK, editor. Sexually transmitted diseases. 4th ed. New York: McGraw-Hill Medical; 2008. p. 109–127
- Ghani AC, Swinton J, Garnett GP. The role of sexual partnership networks in the epidemiology of gonorrhea. Sex Transm Dis. 1997;24:45–56. DOIPubMedGoogle Scholar
- Kenyon C. Strong associations between national prevalence of various STIs suggests sexual network connectivity is a common underpinning risk factor. BMC Infect Dis. 2017;17:682. DOIPubMedGoogle Scholar
- Aral SO. Determinants of STD epidemics: implications for phase appropriate intervention strategies. Sex Transm Infect. 2002;78(Suppl 1):i3–13. DOIPubMedGoogle Scholar
- Fenton KA, Imrie J. Increasing rates of sexually transmitted diseases in homosexual men in Western europe and the United States: why? Infect Dis Clin North Am. 2005;19:311–31. DOIPubMedGoogle Scholar
- Truong HM, Kellogg T, Klausner JD, Katz MH, Dilley J, Knapper K, et al. Increases in sexually transmitted infections and sexual risk behaviour without a concurrent increase in HIV incidence among men who have sex with men in San Francisco: a suggestion of HIV serosorting? [Erratum in: Sex Transm Infect. 2007;83:76]. Sex Transm Infect. 2006;82:461–6. DOIPubMedGoogle Scholar
- The EMIS Network. EMIS 2010: The European Men-Who-Have-Sex-With-Men Internet Survey. Findings from 38 countries. Stockholm: European Centre for Disease Prevention and Control; 2013.
- Glick SN, Morris M, Foxman B, Aral SO, Manhart LE, Holmes KK, et al. A comparison of sexual behavior patterns among men who have sex with men and heterosexual men and women. J Acquir Immune Defic Syndr. 2012;60:83–90. DOIPubMedGoogle Scholar
- Grant RM, Lama JR, Anderson PL, McMahan V, Liu AY, Vargas L, et al.; iPrEx Study Team. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587–99. DOIPubMedGoogle Scholar
- Sonnenberg P, Clifton S, Beddows S, Field N, Soldan K, Tanton C, et al. Prevalence, risk factors, and uptake of interventions for sexually transmitted infections in Britain: findings from the National Surveys of Sexual Attitudes and Lifestyles (Natsal). Lancet. 2013;382:1795–806. DOIPubMedGoogle Scholar
- Cantas L, Shah SQ, Cavaco LM, Manaia CM, Walsh F, Popowska M, et al. A brief multi-disciplinary review on antimicrobial resistance in medicine and its linkage to the global environmental microbiota. Front Microbiol. 2013;4:96. DOIPubMedGoogle Scholar
- Lipsitch M, Samore MH. Antimicrobial use and antimicrobial resistance: a population perspective. Emerg Infect Dis. 2002;8:347–54. DOIPubMedGoogle Scholar
- Unemo M, Shafer WM. Antimicrobial resistance in Neisseria gonorrhoeae in the 21st century: past, evolution, and future. Clin Microbiol Rev. 2014;27:587–613. DOIPubMedGoogle Scholar
- Wi T, Lahra MM, Ndowa F, Bala M, Dillon JR, Ramon-Pardo P, et al. Antimicrobial resistance in Neisseria gonorrhoeae: Global surveillance and a call for international collaborative action. PLoS Med. 2017;14:e1002344. DOIPubMedGoogle Scholar
- Goossens H, Ferech M, Vander Stichele R, Elseviers M; ESAC Project Group. Outpatient antibiotic use in Europe and association with resistance: a cross-national database study. Lancet. 2005;365:579–87. DOIPubMedGoogle Scholar
- Bauer HM, Mark KE, Samuel M, Wang SA, Weismuller P, Moore D, et al. Prevalence of and associated risk factors for fluoroquinolone-resistant Neisseria gonorrhoeae in California, 2000-2003. Clin Infect Dis. 2005;41:795–803. DOIPubMedGoogle Scholar
- Wind CM, de Vries E, Schim van der Loeff MF, van Rooijen MS, van Dam AP, Demczuk WHB, et al. Decreased azithromycin susceptibility of Neisseria gonorrhoeae isolates in patients recently treated with azithromycin. Clin Infect Dis. 2017;65:37–45. DOIPubMedGoogle Scholar
- Ito M, Deguchi T, Mizutani KS, Yasuda M, Yokoi S, Ito S, et al. Emergence and spread of Neisseria gonorrhoeae clinical isolates harboring mosaic-like structure of penicillin-binding protein 2 in Central Japan. Antimicrob Agents Chemother. 2005;49:137–43. DOIPubMedGoogle Scholar
- Malhotra-Kumar S, Lammens C, Coenen S, Van Herck K, Goossens H. Effect of azithromycin and clarithromycin therapy on pharyngeal carriage of macrolide-resistant streptococci in healthy volunteers: a randomised, double-blind, placebo-controlled study. Lancet. 2007;369:482–90. DOIPubMedGoogle Scholar
- Jakobsson HE, Jernberg C, Andersson AF, Sjölund-Karlsson M, Jansson JK, Engstrand L. Short-term antibiotic treatment has differing long-term impacts on the human throat and gut microbiome. PLoS One. 2010;5:e9836. DOIPubMedGoogle Scholar
- Fung M, Scott KC, Kent CK, Klausner JD. Chlamydial and gonococcal reinfection among men: a systematic review of data to evaluate the need for retesting. Sex Transm Infect. 2007;83:304–9. DOIPubMedGoogle Scholar
- Hamilton HL, Dillard JP. Natural transformation of Neisseria gonorrhoeae: from DNA donation to homologous recombination. Mol Microbiol. 2006;59:376–85. DOIPubMedGoogle Scholar
- Midtlyng PJ, Grave K, Horsberg TE. What has been done to minimize the use of antibacterial and antiparasitic drugs in Norwegian aquaculture? Aquacult Res. 2011;42:28–34. DOIGoogle Scholar
- Liu YJ, Rosten TW, Henriksen K, Hognes ES, Summerfelt S, Vinci B. Comparative economic performance and carbon footprint of two farming models for producing Atlantic salmon (Salmo salar): Land-based closed containment system in freshwater and open net pen in seawater. Aquacult Eng. 2016;71:1–12. DOIGoogle Scholar
- Cabello FC, Godfrey HP, Tomova A, Ivanova L, Dölz H, Millanao A, et al. Antimicrobial use in aquaculture re-examined: its relevance to antimicrobial resistance and to animal and human health. Environ Microbiol. 2013;15:1917–42. DOIPubMedGoogle Scholar
- Kirkcaldy RD, Bartoces MG, Soge OO, Riedel S, Kubin G, Del Rio C, et al. Antimicrobial drug prescription and Neisseria gonorrhoeae susceptibility, United States, 2005–2013. Emerg Infect Dis. 2017;23:1657–63. DOIPubMedGoogle Scholar
- Workowski KA. Centers for Disease Control and Prevention sexually transmitted diseases treatment guidelines. Clin Infect Dis. 2015;61(Suppl 8):S759–62. DOIPubMedGoogle Scholar
- LeFevre ML; U.S. Preventive Services Task Force. Screening for chlamydia and gonorrhea: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161:902–10. DOIPubMedGoogle Scholar
- Marcus JL, Hurley LB, Hare CB, Nguyen DP, Phengrasamy T, Silverberg MJ, et al. Preexposure prophylaxis for HIV prevention in a large integrated health care system: adherence, renal safety, and discontinuation. J Acquir Immune Defic Syndr. 2016;73:540–6. DOIPubMedGoogle Scholar
- Tsoumanis A, Hens N, Kenyon C. Do screening programmes for chlamydia and gonorrhea in men who have sex with men reduce the prevalence of these infections? A systematic review of observational studies. Sex Transm Dis. 2018. In press. DOIGoogle Scholar
- Buyze J, Vanden Berghe W, Hens N, Kenyon C. Current levels of gonorrhoea screening in MSM in Belgium may have little effect on prevalence: a modelling study. Epidemiol Infect. 2018;146:333–8. DOIPubMedGoogle Scholar
- Jenness SM, Weiss KM, Goodreau SM, Gift T, Chesson H, Hoover KW, et al. Incidence of gonorrhea and chlamydia following HIV preexposure prophylaxis among men who have sex with men: a modeling study. Clin Infect Dis. 2017;65:712–8. DOIPubMedGoogle Scholar
- World Health Organization. Global action plan to control the spread and impact of antimicrobial resistance in Neisseria gonorrhoeae. Geneva: The Organization; 2012.
- von Wintersdorff CJH, Penders J, van Niekerk JM, Mills ND, Majumder S, van Alphen LB, et al. Dissemination of antimicrobial resistance in microbial ecosystems through horizontal gene transfer. Front Microbiol. 2016;7:173. DOIPubMedGoogle Scholar
- Fingerhuth SM, Bonhoeffer S, Low N, Althaus CL. Antibiotic-resistant Neisseria gonorrhoeae spread faster with more treatment, not more sexual partners. PLoS Pathog. 2016;12:e1005611. DOIPubMedGoogle Scholar
- Chan CH, McCabe CJ, Fisman DN. Core groups, antimicrobial resistance and rebound in gonorrhoea in North America. Sex Transm Infect. 2012;88:200–4. DOIPubMedGoogle Scholar
- Public Health England. Surveillance of antimicrobial resistance in Neisseria gonorrhoeae: key findings from the Gonococcal Resistance to Antimicrobials Surveillance Programme (GRASP) and related surveillance data. London: Public Health England; 2015.
- Workgroup AGP; A2058G Prevalence Workgroup. Prevalence of the 23S rRNA A2058G point mutation and molecular subtypes in Treponema pallidum in the United States, 2007 to 2009. Sex Transm Dis. 2012;39:794–8.PubMedGoogle Scholar
- Read P, Jeoffreys N, Tagg K, Guy RJ, Gilbert GL, Donovan B. Azithromycin-resistant syphilis-causing strains in Sydney, Australia: prevalence and risk factors. J Clin Microbiol. 2014;52:2776–81. DOIPubMedGoogle Scholar
- Read TR, Fairley CK, Tabrizi SN, Bissessor M, Vodstrcil L, Chow EP, et al. Azithromycin 1.5g over 5 days compared to 1g single dose in urethral Mycoplasma genitalium: impact on treatment outcome and resistance. Clin Infect Dis. 2017;64:250–6. DOIPubMedGoogle Scholar
- Baker KS, Dallman TJ, Ashton PM, Day M, Hughes G, Crook PD, et al. Intercontinental dissemination of azithromycin-resistant shigellosis through sexual transmission: a cross-sectional study. Lancet Infect Dis. 2015;15:913–21. DOIPubMedGoogle Scholar
- Gaudreau C, Rodrigues-Coutlée S, Pilon PA, Coutlée F, Bekal S. Long-lasting outbreak of erythromycin- and ciprofloxacin-resistant Campylobacter jejuni subspecies jejuni from 2003 to 2013 in men who have sex with men, Quebec, Canada. Clin Infect Dis. 2015;61:1549–52. DOIPubMedGoogle Scholar
- Diep BA, Chambers HF, Graber CJ, Szumowski JD, Miller LG, Han LL, et al. Emergence of multidrug-resistant, community-associated, methicillin-resistant Staphylococcus aureus clone USA300 in men who have sex with men. Ann Intern Med. 2008;148:249–57. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 24, Number 7—July 2018
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Chris R. Kenyon, Institute of Tropical Medicine, Nationalestraat 155, Antwerpen 2000, Belgium
Top