Volume 25, Number 5—May 2019
Dispatch
Fatal Meningitis in Patient with X-Linked Chronic Granulomatous Disease Caused by Virulent Granulibacter bethesdensis
Cite This Article
Citation for Media
Abstract
Granulibacter bethesdensis is a pathogen reported to cause recurrent lymphadenitis exclusively in persons with chronic granulomatous disease. We report a case of fatal meningitis caused by a highly virulent G. bethesdensis strain in an adolescent in Europe who had chronic granulomatous disease.
Chronic granulomatous disease (CGD) is a primary immunodeficiency characterized by a deficient nicotinamide adenine dinucleotide phosphate oxidative burst that impairs phagocyte superoxide formation and killing of certain pathogens. Mutations can occur in any of the 5 subunits of nicotinamide adenine dinucleotide phosphate oxidase. Most cases are inherited as X-linked defects (gp91phox), but they also can occur in an autosomal recessive manner (1). Increased susceptibility develops to recurrent infections of the skin, lymph nodes, lungs, and other organs (2), mostly caused by bacteria and fungi, including Staphylococcus aureus, Serratia marcescens, Burkholderia cepacia, Salmonella spp., Nocardia spp., and Aspergillus spp. (2). Emerging organisms, such as Granulibacter bethesdensis and other methylotrophs, occur almost exclusively in CGD patients (3,4).
G. bethesdensis was first described in 2006, when it was isolated in a CGD patient with lymphadenitis (4). It is a gram-negative, aerobic, oxidase-negative, catalase-positive, nonmotile coccobacillus to rod-shaped bacterium belonging to the Acetobacteraceae family (5,6). G. bethesdensis was the first of these Acetobacteraceae family bacteria with proven pathogenicity in humans, causing invasive disease in CGD patients and mice (4). It has been mostly linked to indolent nonfatal lymphadenitis and deep neck infections in patients in North America. The infection can recur over several years by reactivation of the same strain or reinfection with different strains (3,7–9). The first fatal infection was reported in a 10-year-old boy from Spain, who died of fulminant sepsis (10). In vitro, G. bethesdensis shows extensive resistance to various antimicrobial drugs, although its slow growth makes susceptibility testing difficult. Ceftriaxone, aminoglycosides, doxycycline, and trimethoprim/sulfamethoxazole showed activity in vitro (7).
We report a case of G. bethesdensis meningitis in a patient with X-linked CGD. We also report animal data comparing this G. bethesdensis strain with the strain recovered from recurrent lymphadenitis in a US CGD patient.
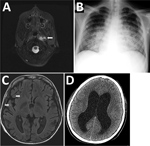
The patient was a 16-year-old boy whose X-linked CGD (CYBB exon 13 deletion) was diagnosed when he was 2 years old. His disease had been well-controlled with cotrimoxazole, itraconazole, and interferon-γ. In September 2014, he was hospitalized with a deep cervical abscess (Figure 1, panel A) and received a 5-week course of intravenous ciprofloxacin, doxycycline, and ceftriaxone that resulted in complete clinical and radiologic resolution, followed by 6 weeks of oral amoxicillin/clavulanic acid, doxycycline, and ciprofloxacin along with his usual prophylaxis. No pathogen was identified despite blood cultures, bronchoalveolar lavage, and lymph node biopsy cultures and broad-range bacterial PCR.
After this regimen was completed, the boy was readmitted for 8 weeks with pneumonia with pleural effusion (Figure 1, panel B). Full 16S rRNA gene sequencing (≈1,500 bp) identified Cupriavidus spp. in pleural fluid. He received meropenem, amikacin, ciprofloxacin, teicoplanin, doxycycline, and voriconazole, and his condition improved. However, 2 weeks later, fever returned, along with splenomegaly, hemodynamic instability, pancytopenia, hypofibrinogenemia, hyperferritinemia, and elevated soluble CD25. He received intravenous immunoglobulin and dexamethasone for this inflammatory condition and fully recovered. Neck and lung computed tomography images and positron emission tomography performed 1 month later showed no signs of active infection.
Nevertheless, a few days later, the patient sought care for altered mental status, hallucinations, aggressiveness, and respiratory instability requiring admission to the pediatric intensive care unit. He had extensive bilateral pneumonia and multiple intraparenchymal brain abscesses (Figure 1, panel C). Meropenem, ciprofloxacin, amikacin, doxycycline, teicoplanin, and voriconazole were started; results of cerebrospinal fluid (CSF) and lung biopsy samples were unremarkable. Teicoplanin was switched to linezolid and voriconazole to caspofungin and liposomal amphotericin B because of toxicity concerns. Four weeks later, he was discharged from the intensive care unit. One month later, fever, vomiting, and focal neurologic deficits developed. CSF showed pleocytosis and hypoglycorrhachia with elevated protein levels. Cerebral imaging confirmed leptomeningitis. Isoniazid, clarithromycin, and rifampin were added to his treatment regimen, but his neurologic status continued to deteriorate. Obstructive hydrocephalus (Figure 1, panel D) and multiorgan failure developed, and he died 3 months later.
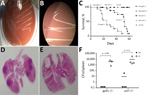
CSF cultures yielded yellow-brown, shiny, small colonies (2–4 mm) on chocolate agar after 4 day’s incubation. (Figure 2, panels A, B). Full 16S RNA sequencing (≈1,500 bp) showed 99.7% match to the type strain of G. bethesdensis CGDNIH1T (ATCC BAA-1260T, DSM 17861T) from North America and 100% match to a previously reported G. bethesdensis strain from Spain. Nonstandardized susceptibility test using Etest, performed in Mueller Hinton agar supplemented with 5% sheep blood agar with an overnight air incubation at 37°C, showed resistance to doxycycline (MIC 24 mg/L) and ceftriaxone (MIC >32 mg/L).
We used mouse models of CGD to determine whether differences existed in immune response, pathogenicity, or severity of disease between the European (CSF strain) and the US (type strain) strains. We intraperitoneally infected gp91phox−/− mice with 107 CFU of G. bethesdensis type strain and p47phox−/− mice intraperitoneally with 107 CFU of CSF strain and monitored moribundity during infection. We euthanized gp91phox−/− mice 4 weeks and p47phox−/− mice 8 weeks after infection and collected brain, spleen, lung, lymph nodes, and blood for culture, bacterial enumeration, and histopathologic examination. Plasma cytokines were assayed.
Although we observed differences between gp91phox−/− and p47phox−/− mice, both CGD mice models showed high rates of death when infected with CSF strain. No deaths occurred in mice infected with type strain, nor did CSF strain cause disease in wild-type mice (Figure 2, panel C). CSF strain–infected mice showed more severe pathologic organ changes than did type strain–infected mice (Figure 2, panels D, E; Appendix Figure 1). We performed quantitative cultures to assess bacterial load in the spleens of inoculated mice. CSF strain CFUs were 100–1,000 times higher than those of type strain 4 and 8 weeks after infection (Table; Figure 2, panel F). In addition, infection with CSF strain yielded 100–1,000 times higher CFUs in spleens of CGD mice than in wild-type mice 4 and 8 weeks after infection. (Appendix Figure 2). CSF strain showed faster growth on solid and in liquid media and a higher optimal growth temperature (37°C) than previously described North America lymph node isolates (Appendix Figure 3). CGD mice infected with CSF strain showed higher plasma interleukin-1β, tumor necrosis factor–α, and interleukin-6 than those infected with type strain 4 and 8 weeks after infection, which correlated with differences in tissue bacterial load. Cytokine levels did not increase in wild-type mice infected with CSF strain (Appendix Figure 4).
G. bethesdensis is an emerging pathogen shown to cause infection exclusively in CGD patients and has a spectrum of disease severity ranging from chronic and recurrent infections to fulminant sepsis, central nervous system infection, and death (3,7,10). Until recently, all reported North America cases were nonfatal chronic infections; 1 case from Europe (Spain) was fatal. Recently, Mayer et al. reported a X-linked CGD patient in the United States who died of fulminant infection with an organism with 100% identity to 500 bp of G. bethesdensis 16S (11). Unfortunately, that G. bethesdensis isolate was not available for analysis and comparison with other G. bethesdensis strains. The previous strain from Europe was highly resistant to antimicrobial agents, including colistin, most β-lactams, and quinolones (10).
We found that a CSF G. bethesdensis strain, showing an identical 16S sequence to a previously described fulminant strain from Europe, was more virulent and lethal in a mouse model than the G. bethesdensis US type strain and more virulent in gp91phox−/− than in p47 phox−/− mice. A fatal case of G. bethesdensis infection in the United States suggests that heterogeneity might exist among North America G. bethesdensis strains. Bacterial genome sequencing may identify discrete virulence factors. G. bethesdensis must be included as a cause of fatal disseminated infection in CGD.
Dr. Rebelo is a pediatrics resident in Hospital Dona Estefânia, Centro Hospitalar de Lisboa Central. Her primary research interests are clinical immunology, primary immunodeficiencies, and infectious diseases
Acknowledgment
This work was supported through the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health.
References
- Holland SM. Chronic granulomatous disease. [viii.]. Hematol Oncol Clin North Am. 2013;27:89–99, viii. DOIPubMedGoogle Scholar
- Gennery A. Recent advances in understanding and treating chronic granulomatous disease. F1000 Res. 2017;6:1427. DOIPubMedGoogle Scholar
- Falcone EL, Petts JR, Fasano MB, Ford B, Nauseef WM, Neves JF, et al. Methylotroph infections and chronic granulomatous disease. Emerg Infect Dis. 2016;22:404–9. DOIPubMedGoogle Scholar
- Greenberg DE, Ding L, Zelazny AM, Stock F, Wong A, Anderson VL, et al. A novel bacterium associated with lymphadenitis in a patient with chronic granulomatous disease. PLoS Pathog. 2006;2:e28. DOIPubMedGoogle Scholar
- Greenberg DE, Porcella SF, Stock F, Wong A, Conville PS, Murray PR, et al. Granulibacter bethesdensis gen. nov., sp. nov., a distinctive pathogenic acetic acid bacterium in the family Acetobacteraceae. Int J Syst Evol Microbiol. 2006;56:2609–16. DOIPubMedGoogle Scholar
- Greenberg DE, Porcella SF, Zelazny AM, Virtaneva K, Sturdevant DE, Kupko JJ III, et al. Genome sequence analysis of the emerging human pathogenic acetic acid bacterium Granulibacter bethesdensis. J Bacteriol. 2007;189:8727–36. DOIPubMedGoogle Scholar
- Greenberg DE, Shoffner AR, Zelazny AM, Fenster ME, Zarember KA, Stock F, et al. Recurrent Granulibacter bethesdensis infections and chronic granulomatous disease. Emerg Infect Dis. 2010;16:1341–8. DOIPubMedGoogle Scholar
- Zarember KA, Marshall-Batty KR, Cruz AR, Chu J, Fenster ME, Shoffner AR, et al. Innate immunity against Granulibacter bethesdensis, an emerging gram-negative bacterial pathogen. Infect Immun. 2012;80:975–81. DOIPubMedGoogle Scholar
- Chu J, Song HH, Zarember KA, Mills TA, Gallin JI. Persistence of the bacterial pathogen Granulibacter bethesdensis in chronic granulomatous disease monocytes and macrophages lacking a functional NADPH oxidase. J Immunol. 2013;191:3297–307. DOIPubMedGoogle Scholar
- López FC, de Luna FF, Delgado MC, de la Rosa II, Valdezate S, Nieto JA, et al. Granulibacter bethesdensis isolated in a child patient with chronic granulomatous disease. J Infect. 2008;57:275–7. DOIPubMedGoogle Scholar
- Mayer EFF, Gialanella P, Munjal I, Cunningham-Rundles C, Dara J. Fulminant sepsis due to Granulibacter bethesdensis in a 4-year-old boy with X-linked chronic granulomatous disease. Pediatr Infect Dis J. 2017;36:1165–6. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: March 29, 2019
1These authors contributed equally to this article.
2These authors contributed equally to this article.
Table of Contents – Volume 25, Number 5—May 2019
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
João Farela Neves, Primary Immunodeficiencies Unit, Hospital Dona Estefânia, Rua Jacinta Marto, 1145, Lisbon, Portugal
Top