Volume 26, Number 12—December 2020
Dispatch
Characterization and Source Investigation of Multidrug-Resistant Salmonella Anatum from a Sustained Outbreak, Taiwan
Cite This Article
Citation for Media
Abstract
An ongoing outbreak of multidrug-resistant Salmonella enterica serovar Anatum began in Taiwan in 2015. Pork and poultry were identified as vehicles for transmission. Contaminated meat contributed to the high rate of infections among children. Nearly identical Salmonella Anatum strains have been identified in the United Kingdom, the United States, and the Philippines.
Nontyphoidal Salmonella (NTS) is a major cause for foodborne diseases worldwide. In Taiwan, the ambient climate and flourishing pig-raising industry makes NTS infections rampant. As in other countries, salmonellosis was primarily caused by Salmonella enterica serovars Enteritidis and Typhimurium in Taiwan (1), but rare serovars such as Salmonella Goldcoast have appeared in recent years (2). Recommended antimicrobial treatment options for salmonellosis include fluoroquinolones and extended-spectrum cephalosporins (1). However, resistance to these antibiotics has been emerging in many countries, leading to increased disease prevalence, disease severity, and death and the requirement of last-line antimicrobial drugs (e.g., carbapenems) (3–5).
Since 2015, northern Taiwan has seen an increase in Salmonella infections, caused by previously rare Salmonella Anatum. The infections were also reported in central Taiwan, indicating that this outbreak had already prevailed throughout the entire island (6). Co-resistance to ceftriaxone and ciprofloxacin are the main feature of the outbreak clone. Evidence from epidemiologic, laboratory, and supply-chain investigations identified raw pork and poultry as the vehicle for spread of this strain. More important, genomic comparisons against the global public database indicated that this clone has appeared in Europe, Asia, and America. Given the increasing globalization of foodstuffs, these findings prompt an urgent global sharing of whole-genome sequencing (WGS) data to facilitate disease surveillance and early recognition of international foodborne outbreaks (7,8).
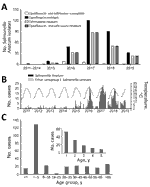
Chang Gung Memorial Hospital is a main referral hospital for cities in northern Taiwan, including Taipei, New Taipei, and Taoyuan. The population in this region is »7 million. In 2012, the hospital’s clinical microbiology laboratory launched a program to monitor the NTS serovars causing human infections. All Salmonella isolates from patients were collected and serotyped. Before 2015, very few Salmonella Anatum isolates were recovered, and most were susceptible to antimicrobial agents. Since then, an increase has been observed, peaking in 2017 (Figure 1, panel A). As of June 2019, a total of 319 nonrepetitive isolates have been identified; of these, 197 (61.8%) isolates were ceftriaxone-resistant (MIC >2 μg/mL), 301 (94.4%) were ciprofloxacin-resistant (MIC >0.12 μg/mL), and 197 (61.8%) were resistant to both. In addition, 292 (91.5%) isolates were resistant to chloramphenic, and 295 (92.5%) were resistant to trimethoprim/sulfamethoxazole. A positive correlation was found between higher temperatures and the infections (r = 0.4; p<0.05) (Figure 1, panel B); however, no notable effects on Salmonella Anatum infections have been associated with precipitation or humidity (r<0.3; p>0.05).
Detailed methods are described in the Appendix. We first reviewed the clinical and laboratory characteristics of 278 patients from 2015–2018. Most patients had acute gastroenteritis, whereas a few (14/278, 5%) had invasive diseases, such as bacteremia and sepsis. In terms of age distribution, the highest number of cases were in young children (Figure 1, panel C). Pediatric patients (n = 169) had significantly higher rates than adult patients (n = 109) for hospitalization (79.2% vs. 55.0%; p<0.05), diarrhea (89.9% vs. 68.8%; p<0.05), and fever (89.2% vs. 58.1%; p<0.05).
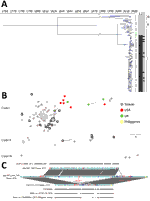
Multilocus sequence typing indicated that the entire collection of clinical Salmonella Anatum isolates belonged to sequence type 64. We randomly selected 54 clinical isolates for WGS (Appendix). Both core genome multilocus sequence typing and whole-genome single-nucleotide polymorphism analyses, performed by using the BacWGSTdb database (9), further divided these isolates into 3 clades (Figure 2, panel A, B). Clades I and II were more closely related to each other; their most recent common ancestor occurred >21 years ago. Clade III was more distantly connected to these 2 clades. Typing based on PCR assay was performed on the unsequenced isolates. Clade I accounted for 95.6% (305/319) of all isolates, suggesting it was the cause of the outbreak. The isolates resistant to ceftriaxone, ciprofloxacin, or both clustered within clade I, whereas the isolates of clades II and III were more susceptible. Most of the clade I isolates harbored a 90-kb IncA/C plasmid carrying blaDHA-1 (encoding a class C β-lactamase) and qnrB (conferring resistance to quinolones). A conjugation assay demonstrated that this plasmid conferred ceftriaxone and ciprofloxacin resistance. In addition, 31 (9.7%) clinical isolates carried blaCMY-2, which was located within a >100-kb IncI1 plasmid and also encoded a class C β-lactamase. These 31 isolates carried blaDHA-1 simultaneously. In 11 of them, the blaDHA-1–carrying and blaCMY-2–carrying plasmids were fused into 1 large plasmid (Figure 2, panel C).
By comparing these findings against sequences in GenBank, we found nearly identical genomic sequences for isolates in the United Kingdom, the United States, and the Philippines. The collection time for these isolates also occurred during 2015–2019, which nearly coincided with the outbreak in Taiwan. These international Salmonella Anatum isolates also carried the 90-kb IncA/C plasmid (Figure 2, panel A, C); therefore, they were likely ceftriaxone- and ciprofloxacin-resistant concomitantly. The only distinction of these international isolates was their lack of the blaCMY-2–carrying plasmid. Accordingly, we speculated that the Salmonella Anatum clone had arrived in Taiwan through food trade and later acquired the blaCMY-2–carrying plasmid.
To trace the source of Salmonella Anatum, we investigated food samples from supermarkets and traditional markets of 8 districts with high density of Salmonella patients in New Taipei City and Taoyuan City, Taiwan (Appendix). A total of 11 Salmonella Anatum isolates were collected from pork, 4 from poultry, and 1 from beef in these regions (Appendix). WGS showed that they all belonged to clades I and II, providing strong evidence that raw meats were the outbreak vehicle. All 16 isolates harbored the blaDHA-1–carrying IncA/C plasmid. Other Salmonella serovars also were detected in this investigation. The overall Salmonella isolation rate from retail meats was significantly higher in traditional markets than in the supermarkets (p<0.001) (Appendix). In Taiwan, pork in the supermarkets is usually provided through the cold transportation chain, whereas for traditional markets pork is usually provided through the traditional chain, with notable differences. Temperatures were much lower in the cutting factory and butcher shop in the cold chain than in the traditional chain (Appendix). Furthermore, pork was wrapped by plastic tissue and bags in the cold chain, but the traditional chain did not do any wrapping or packaging during transportation.
To clarify the contradictory findings that most infections occurred in young children even though pork is not a major food for infants, we conducted a questionnaire survey among parents of 20 infants (<1 year of age) with NTS infections and 80 parents of infants without (controls) (Appendix). Parents of the infected infants more often touched, rinsed, and cooked meat before feeding other foods to their infants (Appendix). Moreover, these parents were more willing to purchase meat from traditional markets rather than supermarkets. A possibility is that they bought meat from the traditional markets, then their frequent rinsing flushed the Salmonella on the surface of the meats, cutting boards and knives, and sinks, and finally onto fresh vegetables, fruit, and other ready-to-eat foods that were cross-contaminated and reached the infants through parents or other caregivers. This transmission mode is of particular importance in infants and has already been reported for other bacterial pathogens such as Yersinia enterocolitica (10).
Our study sought to describe an outbreak in Taiwan caused by a multidrug-resistant Salmonella Anatum clone. The questionnaire and supply-chain investigations we conducted found that the infection cases were closely associated with improper packaging during transportation and unhygienic food handling in the customers’ kitchen. The high similarity of genomic sequence between the Taiwan isolates and international isolates indicates the global dissemination of this clone and highlights the public health value of multicountry sharing of epidemiologic, trace-back, microbiologic, genomic, and food trade data.
Dr. Feng is an associate professor at the Institute for Translational Medicine, Zhejiang University School of Medicine, Hangzhou, China. His research interests are the epidemiology, genomics, and drug-resistance mechanisms of nontyphoidal Salmonella.
Acknowledgment
This study was financially supported by the National Natural Science Foundation, China (grant no. 31670132); the Chang Gung Memorial Hospital, Taiwan (grant nos. CRRPG3F0084, CMRPG3D1721-3, CMRPG3G1451-3, and CMRPG3E1371-3); and the Ministry of Health and Welfare, Taiwan (grant nos. MOHW106-CDC-C-114-113702, MOHW107-CDC-C-114-123505, and MOHW108-CDC-C-114-133505).
References
- Chen HM, Wang Y, Su LH, Chiu CH. Nontyphoid salmonella infection: microbiology, clinical features, and antimicrobial therapy. Pediatr Neonatol. 2013;54:147–52. DOIPubMedGoogle Scholar
- Feng Y, Chang YJ, Fang SH, Su LH, Li HC, Yang HP, et al. Emergence and evolution of high-level cephalosporin-resistant Salmonella Goldcoast in Northern Taiwan. Open Forum Infect Dis. 2019;6:
ofz447 . DOIPubMedGoogle Scholar - Qi X, Li P, Xu X, Yuan Y, Bu S, Lin D. Epidemiological and molecular investigations on Salmonella responsible for gastrointestinal infections in the southwest of Shanghai from 1998 to 2017. Front Microbiol. 2019;10:2025. DOIPubMedGoogle Scholar
- Jean SS, Lu MC, Shi ZY, Tseng SH, Wu TS, Lu PL, et al. In vitro activity of ceftazidime-avibactam, ceftolozane-tazobactam, and other comparable agents against clinically important Gram-negative bacilli: results from the 2017 Surveillance of Multicenter Antimicrobial Resistance in Taiwan (SMART). Infect Drug Resist. 2018;11:1983–92. DOIPubMedGoogle Scholar
- Zhan Z, Xu X, Gu Z, Meng J, Wufuer X, Wang M, et al. Molecular epidemiology and antimicrobial resistance of invasive non-typhoidal Salmonella in China, 2007-2016. Infect Drug Resist. 2019;12:2885–97. DOIPubMedGoogle Scholar
- Chiou CS, Hong YP, Liao YS, Wang YW, Tu YH, Chen BH, et al. New multidrug-resistant Salmonella enterica serovar Anatum clone, Taiwan, 2015–2017. Emerg Infect Dis. 2019;25:144–7. DOIPubMedGoogle Scholar
- Ruan Z, Yu Y, Feng Y. The global dissemination of bacterial infections necessitates the study of reverse genomic epidemiology. Brief Bioinform. 2020;21:741–50. DOIPubMedGoogle Scholar
- Nadon C, Van Walle I, Gerner-Smidt P, Campos J, Chinen I, Concepcion-Acevedo J, et al.; FWD-NEXT Expert Panel. PulseNet International: Vision for the implementation of whole genome sequencing (WGS) for global food-borne disease surveillance. Euro Surveill. 2017;22:30544. DOIPubMedGoogle Scholar
- Ruan Z, Feng Y. BacWGSTdb, a database for genotyping and source tracking bacterial pathogens. Nucleic Acids Res. 2016;44(D1):D682–7. DOIPubMedGoogle Scholar
- Lee LA, Gerber AR, Lonsway DR, Smith JD, Carter GP, Puhr ND, et al. Yersinia enterocolitica O:3 infections in infants and children, associated with the household preparation of chitterlings. N Engl J Med. 1990;322:984–7. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: August 18, 2020
Table of Contents – Volume 26, Number 12—December 2020
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Cheng-Hsun Chiu, Department of Pediatrics, Chang Gung Memorial Hospital, No. 5, Fu-Hsin St, Kweishan 333, Taoyuan, Taiwan
Top