Volume 26, Number 12—December 2020
Research Letter
SARS-CoV-2 in Quarantined Domestic Cats from COVID-19 Households or Close Contacts, Hong Kong, China
Cite This Article
Citation for Media
Abstract
We tested 50 cats from coronavirus disease households or close contacts in Hong Kong, China, for severe acute respiratory syndrome coronavirus 2 RNA in respiratory and fecal samples. We found 6 cases of apparent human-to-feline transmission involving healthy cats. Virus genomes sequenced from 1 cat and its owner were identical.
Naturally occurring human-to-animal transmission of severe acute respiratory syndrome (SARS) coronavirus was reported during 2003 when viral RNA was detected in oropharyngeal and rectal swab specimens from healthy domestic cats in a housing estate at the center of a large SARS cluster in Hong Kong, China; infections were confirmed serologically (1). Susceptibility of cats to infection with this virus and transmission between cats were demonstrated experimentally (2). Pulmonary pathologic changes, similar to those for humans with SARS, developed in infected cats, but the cats remained asymptomatic (2,3).
These findings informed the current precautionary strategy of the Agriculture, Fisheries and Conservation Department of Hong Kong to quarantine mammalian pets from households with confirmed human coronavirus disease (COVID-19) or their close contacts (defined as a person who had face-to-face contact for >15 min with a person who had confirmed SARS coronavirus 2 [SARS-CoV-2] infection) in a holding facility, when alternative care was unavailable. Pets are swabbed for SARS-CoV-2 testing and confined until reverse transcription PCR (RT-PCR) results are negative on 2 consecutive occasions. Findings for 2 infected dogs have been reported (4). We report testing results for cats.
Swab (nasal, oral, rectal) specimens and feces collected from nonsedated cats after admission were tested for SARS-CoV-2 RNA at the Agriculture, Fisheries and Conservation Department Veterinary Laboratory by using a commercial RT-PCR targeting the partial envelope and RNA-dependent RNA polymerase genes (Molbiol Lightmix; TIB MOLBIOL, https://www.tib-molbiol.com). This PCR does not show cross-reactivity with these genes from enteric coronavirus (5).
For samples with positive or equivocal results, confirmatory quantitative RT-PCRs targeting nonstructural protein 4, nonstructural protein 16, nucleoprotein, and membrane genes were performed at the World Health Organization COVID-19 Reference Laboratory at the University of Hong Kong (4). Animals with positive results were evaluated by repeated sampling to monitor viral shedding by RT-PCR. Serologic analysis was used selectively.
We sampled 50 cats during February 11–August 11, 2020. Time from onset of COVID-19 symptoms in owners to first sampling of their cats was available for 21 owners of 35 cats and ranged from 3 to 15 (median 8, interquartile range 4) days. SARS-CoV-2 RNA was detected in samples from 6 (12%) of 50 cats (Table, https://wwwnc.cdc.gov/EID/article/26/12/20-2786-T1.htm).
The first positive case (cat 1) was from a household that had 3 persons with confirmed cases of COVID-19; their symptoms (fever, cough, or shortness of breath) started on March 20, 29, and 30, 2020. Their 7-year old, female, domestic shorthair cat was examined by a veterinarian at admission on day 1 (March 30), and reported to be clinically healthy. Nasal, oral, and rectal swab specimens collected on day 1 were positive for SARS-CoV-2 RNA; viral nucleoprotein gene copy number were log10 6.3/mL, log10 5.6/mL, and 3.2 log10/mL, respectively.
Attempts to culture virus from day 1 samples on Vero E6 (ATCC CRL-1586) cells as described (4) were unsuccessful. Viral RNA was detected in oral swab specimens for 8 days and nasal swab specimens for 11 days, but rectal swab specimens were negative after day 1 (Table).
We performed serologic analysis to detect neutralizing antibodies by using a 90% plaque reduction neutralization test for SARS-CoV-2 (4). The result was positive for the only serum sample collected (on day 19) and had titer >1:320 (4).
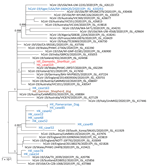
Viral genomes from cat 1 and 1 owner were sequenced by using a MiSeq Sequencing Platform (Illumina, https://www.illumina.com) after reverse transcription of viral RNA and multiple, overlapping, »2-kb PCRs that targeted the viral genome (4). Both genome sequences (29,830 nt sequenced; 99.8% of the genome) were identical (Figure) and deposited in GenBank (accession no. MT628701).
Four of the other 5 positive cats were from confirmed COVID-19–infected households, and 1 indoor-only cat belonged to a close contact who was not confirmed to be infected. Time from onset of COVID-19 symptoms in owners to animal sampling was known for 3 cats (5, 11, and 8 days); 1 had equivocal envelope gene PCR results but was positive by a novel surrogate virus neutralization test (6). Signs of disease did not develop in any infected cats, consistent with experimental feline infections, which are also usually subclinical (7; A. Bosco-Lauth et al., unpub. data).
COVID-19–like signs have been reported in domestic cats naturally infected with SARS-CoV-2 in other countries (8). In addition, 4 tigers and 3 lions with respiratory signs in a zoo in New York, New York, USA, were confirmed to be shedding SARS-CoV-2 in feces (S.L. Bartlett et al., unpub. data). Susceptibility to SARS-CoV-2 might differ between felid species.
SARS-CoV-2 RNA persisted longest in nasal secretions in 1 case for 11 days at low levels. Viral RNA was detected in nasal washes from kittens experimentally inoculated with SARS-CoV-2 for 8–9 days, after which sampling was stopped (7). Cats acquiring infection from being cohoused with experimentally infected cats shed virus in respiratory secretions longer (7 days) than directly inoculated cats (5 days) (8).
Although feline-to-human transmission is theoretically possible, we did not find any evidence of this transmission. The timeline of infection in cat 1 and the finding of an identical SARS-CoV-2 genome sequence in a human from the same household is consistent with human-to-animal transmission. In support of these findings, the cat had no outdoor access.
More widespread serologic surveillance of cats in contact with COVID-19 patients is warranted to determine the prevalence of human-to-cat transmission. Some infected cats might have stopped shedding virus before being quarantined because viral shedding periods as short as 3 days have been reported in experimentally infected cats (7).
Dr. Barrs is a chair professor of companion animal health and disease at the City University of Hong Kong, Hong Kong, China. Her primary research interests are One Health infectious diseases; viral diseases of cats, dogs, and wildlife; and shared environmental pathogens of public health concern, especially cryptic species of Aspergillus.
Acknowledgments
We thank Dominic N.C. Tsang for providing the human specimen.
M.P. was supported by the US National Institutes of Health (contract no. HHSN272201400006C).
References
- World Health Organization. Consensus document on the epidemiology of severe acute respiratory syndrome (SARS). Department of Communicable Disease Surveillance and Response; 2003. WHO/CDS/CSR/GAR/2003.11:1–47 [cited 2020 Sep 10]. https://www.who.int/csr/sars/en/WHOconsensus.pdf
- Martina BE, Haagmans BL, Kuiken T, Fouchier RA, Rimmelzwaan GF, Van Amerongen G, et al. Virology: SARS virus infection of cats and ferrets. Nature. 2003;425:915. DOIPubMedGoogle Scholar
- van den Brand JM, Haagmans BL, Leijten L, van Riel D, Martina BE, Osterhaus AD, et al. Pathology of experimental SARS coronavirus infection in cats and ferrets. Vet Pathol. 2008;45:551–62. DOIPubMedGoogle Scholar
- Sit THC, Brackman CJ, Ip SM, Tam KWS, Law PYT, To EMW, et al. Infection of dogs with SARS-CoV-2. Nature. 2020; (
May ):14; [Epub ahead of print].PubMedGoogle Scholar - Corman VM, Landt O, Kaiser M, Molenkamp R, Meijer A, Chu DK, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020;25:
2000045 . DOIPubMedGoogle Scholar - Tan CW, Chia WN, Qin X, Liu P, Chen MI, Tiu C, et al. A SARS-CoV-2 surrogate virus neutralization test based on antibody-mediated blockage of ACE2-spike protein-protein interaction. Nat Biotechnol. 2020;38:1073–8. DOIPubMedGoogle Scholar
- Shi J, Wen Z, Zhong G, Yang H, Wang C, Huang B, et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS-coronavirus 2. Science. 2020;368:1016–20. DOIPubMedGoogle Scholar
- World Organisation for Animal Health. Questions and answers on COVID-19, Jun 4, 2020 [cited 2020 Jun 8]. https://www.oie.int/scientific-expertise/specific-information-and-recommendations/questions-and-answers-on-2019novel-coronavirus/
Figure
Table
Cite This ArticleOriginal Publication Date: September 30, 2020
1These authors contributed equally to this article.
Table of Contents – Volume 26, Number 12—December 2020
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Vanessa R. Barrs, Department of Veterinary Clinical Sciences, Jockey Club College of Veterinary Medicine, City University of Hong Kong, Kowloon Tong, Hong Kong, China
Top