Volume 26, Number 5—May 2020
Research Letter
Novel Ehrlichia Strain Infecting Cattle Tick Amblyomma neumanni, Argentina, 2018
Cite This Article
Citation for Media
Abstract
In 2018, we detected a novel Ehrlichia strain infecting Amblyomma neumanni ticks in Argentina. The novel strain is phylogenetically related to the ruminant pathogen E. ruminantium and represents a potential risk for veterinary and public health because A. neumanni ticks parasitize domestic and wild ruminants and bite humans.
Ehrlichia spp. are intracellular gram-negative bacteria relevant to human and animal health; they infect monocytes, neutrophils, or endothelial cells, depending on the species involved (1). The genus Ehrlichia (Rickettsiales: Anaplasmataceae) comprises 6 formally recognized tick-transmitted species: E. canis, E. muris, E. chaffeensis, E. ewingii, E. minasensis, and E. ruminantium (2,3). Recently, other Ehrlichia species have been reported and different strains of putative novel Ehrlichia species have been molecularly detected, but their taxonomic positions are still not clearly defined (4–6). Current knowledge about this group of pathogens suggests that a large number of Ehrlichia species might be not yet described.
Amblyomma neumanni ticks are relevant to human and veterinary medicine because in all stages they commonly parasitize wild and domestic ruminants and other large mammals, including humans (7). Moreover, A. neumanni ticks can reportedly be infected with Rickettsia bellii and Rickettsia amblyommatis and are potential vectors of the cattle pathogen Anaplasma marginale (7). To determine the presence of tickborne bacteria of the genus Ehrlichia in questing A. neumanni ticks in northwestern Argentina, we performed phylogenetic analyses on Anaplasmataceae-positive tick DNA samples. All procedures were approved by the Ethics and Biosafety Committee of the Facultad de Ciencias Veterinarias, Universidad Nacional del Litoral, Esperanza, Argentina.
During May 2018 (late autumn), we collected free-living ticks by dragging and by using dry ice-baited traps in Dean Funes (30°22′S, 64°21′W) and San José de la Dormida (30°21′S, 63°58′W), Córdoba Province, Argentina. Both sites are located in the Chaco Seco ecoregion. We identified all ticks by using standard taxonomic keys (7) and individually processed them for DNA extraction by using a boiling method (8). We screened DNA extracts for Anaplasmataceae by real-time PCR targeting the 16S rRNA gene, as previously described (9). We further tested samples positive for Anaplasmataceae by amplification of Ehrlichia genes dsb and groESL, as described elsewhere (5). We sequenced all amplicons and performed phylogenetic analyses with the maximum-likelihood method.
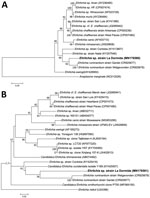
We collected 229 ticks from Dean Funes (70 adults, 159 nymphs) and 62 from San José de la Dormida (24 adults, 38 nymphs) and identified all ticks as A. neumanni. Only 1 adult tick from San José de la Dormida was positive for Anaplasmataceae by PCR. Further analysis of that sample resulted in 2 sequences of 355 bp (dsb) and 784 bp (groESL). Phylogenetic analysis of the dsb sequence (GenBank accession no. MN176580) showed that the A. neumanni tick was infected with a species of Ehrlichia, which we named Ehrlichia sp. strain La Dormida, closely related to E. ruminantium (82.0%, GenBank accession no. CR925677) (Figure, panel A). Other ehrlichiae from South America included in the analysis, such as Ehrlichia sp. strain Córdoba from Argentina (78.0%, GenBank accession no. KY413807) and Ehrlichia sp. strain Natal from Brazil (79.3%, GenBank accession no. KY207546), were placed in a clade sister to the group formed by Ehrlichia sp. strain La Dormida and E. ruminantium. Furthermore, the phylogenetic analysis performed by using the groESL sequence (GenBank accession no. MN176581) confirmed these results (Figure, panel B).
Several recent studies conducted in South America reported finding novel ehrlichial agents infecting jaguars, horses, crab-eating foxes, opossums, sloths, and peccaries (6). Unfortunately, only short dsb sequences are available for those ehrlichiae from South America. Phylogenetic analysis including these sequences (210 positions included in the final dataset) placed them all together in a clade sister to the group formed by Ehrlichia sp. strain La Dormida and E. ruminantium (Appendix Figure).
Ehrlichia sp. strain La Dormida, associated with A. neumanni ticks, circulates in rural areas of northwestern Argentina. In our phylogenetic analyses, Ehrlichia sp. strain La Dormida genotype was unique and well separated from all other available ehrlichial sequences, suggesting that it could represent a distinct species yet to be properly characterized. In addition, these analyses positioned Ehrlichia sp. strain La Dormida in a separate group together with E. ruminantium. The species E. ruminantium is native to the Africa continent, where it is the etiologic agent of heartwater, a tickborne disease of major economic relevance with regard to domestic ruminants throughout sub-Saharan Africa (10). Besides E. ruminantium, the only other species of Ehrlichia known to naturally infect and cause clinical manifestations of ehrlichiosis in cattle is E. minasensis (3). However, E. minasensis is phylogenetically distant from E. ruminantium and closely related to E. canis genotypes (3).
We report A. neumanni ticks as a potential vector of Ehrlichia sp. strain La Dormida. Because it is assumed that bacteria of the genus Ehrlichia are not transmitted transovarially in ticks (1), infection with Ehrlichia must be acquired during feeding of immature ticks, which then pass the infection to adults by transstadial transmission. Regarding E. ruminantium, wild African ruminants are reservoirs of the bacteria (10). The novel Ehrlichia sp. strain La Dormida is phylogenetically related to the ruminant pathogen E. ruminantium and represents a potential risk for veterinary and public health because A. neumanni ticks parasitize domestic and wild ruminants and bite humans.
Dr. Fargnoli is a postdoctoral student at the Instituto de Ciencias Veterinarias del Litoral in Esperanza, Santa Fe, Argentina. Her research interests focus on the ecology of tickborne diseases.
Acknowledgments
We thank Federico Ruiz and Anibal Gomez for their support with field work and Federico Prandi.
This study was supported by Agencia Nacional de Promoción Científica y Tecnológica (PICT-2015-1084) and Agencia Santafesina de Ciencia, Tecnología e Innovación (IO-2017-00125).
References
- Brouqui P, Matsumoto K. Bacteriology and phylogeny of Anaplasmataceae. In: Raoul, D, Parola P, editors. Rickettsial diseases. New York; Informa; 2007. p. 179–198.
- Dumler JS, Barbet AF, Bekker CP, Dasch GA, Palmer GH, Ray SC, et al. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int J Syst Evol Microbiol. 2001;51:2145–65. DOIPubMedGoogle Scholar
- Aguiar DM, Ziliani TF, Zhang X, Melo AL, Braga IA, Witter R, et al. A novel Ehrlichia genotype strain distinguished by the TRP36 gene naturally infects cattle in Brazil and causes clinical manifestations associated with ehrlichiosis. Ticks Tick Borne Dis. 2014;5:537–44. DOIPubMedGoogle Scholar
- Eberhardt AT, Fernandez C, Fargnoli L, Beldomenico PM, Monje LD. A putative novel strain of Ehrlichia infecting Amblyomma tigrinum associated with Pampas fox (Lycalopex gymnocercus) in Esteros del Iberá ecoregion, Argentina. Ticks Tick Borne Dis. 2020;11:
101318 . DOIPubMedGoogle Scholar - Cicuttin GL, De Salvo MN, Nava S. Two novel Ehrlichia strains detected in Amblyomma tigrinum ticks associated to dogs in peri-urban areas of Argentina. Comp Immunol Microbiol Infect Dis. 2017;53:40–4. DOIPubMedGoogle Scholar
- Lopes MG, Muñoz-Leal S, de Lima JTR, Fournier GFDSR, Acosta IDCL, Martins TF, et al. Ticks, rickettsial and erlichial infection in small mammals from Atlantic forest remnants in northeastern Brazil. Int J Parasitol Parasites Wildl. 2018;7:380–5. DOIPubMedGoogle Scholar
- Nava S, Venzal JM, Gonzalez-Acuña DA, Martins TF, Guglielmone AA, editors. Ticks of the Southern Cone of America: diagnosis, distribution and hosts with taxonomy, ecology and sanitary importance. London: Elsevier; 2017. p. 135–42.
- Monje LD, Costa FB, Colombo VC, Labruna MB, Antoniazzi LR, Gamietea I, et al. Dynamics of exposure to Rickettsia parkeri in cattle in the Paraná River delta, Argentina. J Med Entomol. 2016;53:660–5. DOIPubMedGoogle Scholar
- Monje LD, Fernandez C, Percara A. Detection of Ehrlichia sp. strain San Luis and Candidatus Rickettsia andeanae in Amblyomma parvum ticks. Ticks Tick Borne Dis. 2019;10:111–4. DOIPubMedGoogle Scholar
- Allsopp BA. Natural history of Ehrlichia ruminantium. Vet Parasitol. 2010;167:123–35. DOIPubMedGoogle Scholar
Figure
Cite This ArticleOriginal Publication Date: April 01, 2020
Table of Contents – Volume 26, Number 5—May 2020
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Lucas D. Monje, Laboratorio de Ecología de Enfermedades, Instituto de Ciencias Veterinarias del Litoral, R.P. Kreder 2805, Esperanza, Santa Fe 3080, Argentina
Top