Volume 28, Number 11—November 2022
Dispatch
Imported Haycocknema perplexum Infection, United States1
Cite This Article
Citation for Media
Abstract
We report an imported case of myositis caused by a rare parasite, Haycocknema perplexum, in Australia in a 37-year-old man who had progressive facial, axial, and limb weakness, dysphagia, dysphonia, increased levels of creatine kinase and hepatic aminotransferases, and peripheral eosinophilia for 8 years. He was given extended, high-dose albendazole.
Haycocknema perplexum is an enigmatic nematode that is a rare cause of human parasitic myositis (1‒4). Twelve cases have been reported since its initial description in 1998, all in humans (1‒10). The mode of transmission is unclear, but 9 patients reported contact with native wildlife in Tasmania or tropical regions of Queensland, Australia. We report an imported case of myositis caused by infection with this rare parasite.
A 37-year-old man from New Zealand who had previous long-term residence in Australia came to the Mayo Clinic (Rochester, MN, USA) because of an 8-year history of progressive weakness, muscle atrophy, and 32-kg weight loss. Onset was gradual, first involving the pectoralis and biceps brachii, then neck, facial, and distal limb muscles. Additional symptoms included dysphagia, dysphonia, and dyspnea on exertion. Laboratory testing showed peripheral eosinophilia (5%, reference value <3%), and an increased level of creatine kinase (maximum ≈2,000 U/L, reference range 39–308 U/L). Toxoplasmosis had originally been suspected based on finding a possible Toxoplasma gondii cyst on muscle biopsy 1 year after symptom onset, but his weakness progressed despite trimethoprim/sulfamethoxazole therapy, and T. gondii serologic test results were negative. Prednisone therapy worsened his symptoms.
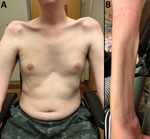
He became wheelchair-bound 7 years after onset of symptoms. He had lived in coastal northern Queensland (Mackay region), Australia from ages 8–20 years, where he had extensive bush exposure but denied bush meat consumption. Neurologic findings included profound asymmetric weakness predominantly affecting proximal upper and lower limbs, neck flexors, and sternocleidomastoids (Figure 1). He also had asymmetric scapular winging, severe weakness of the frontalis, and mild weakness of orbicularis oris.
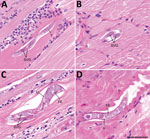
A formalin-fixed, paraffin-embedded muscle tissue from a previous muscle biopsy specimen was obtained, and additional sections showed nonencapsulated male and gravid female nematodes within muscle fibers consistent with H. perplexum (Figure 2). The presence of adult worms enabled trichinellosis to be definitively excluded because only the larval stage of Trichinella sp. is detected in muscle. Attempts at molecular amplification of the cytochrome c oxidase subunit 1 and 18S rRNA genes as described (2,8) from archival formalin-fixed, paraffin-embedded tissue were unsuccessful.
The patient was prescribed a 3-month course of albendazole (400 mg 2×/d). Nineteen months after completing albendazole, the patient reported no further deterioration. However, his muscle power did not improve. Creatinine kinase levels decreased to within the reference range.
Haycocknema perplexum is an enigmatic and presumably zoonotic nematode. Clinical histories of affected patients indicate that contact with wilderness or marsupial wildlife in Australia (n = 6) and consumption of bush meat (n = 4) might be associated with infection, but this possibility has not been confirmed (Table). The phylogenetic position of H. perplexum is unresolved, but it appears to be intermediary between Oxyuridomorpha (e.g., Enterobius vermicularis) and Ascaridomorpha (e.g., Ascaris lumbricoides) (2). All cases of haycocknematosis to date have originated in Australia, specifically in the tropical north of Queensland and Tasmania (Table). Nonhuman animal hosts are unknown. The route of human infection is also unknown but is presumed to be linked to consumption of, or contact with, mammalian wildlife. Because females are ovoviviparous (eggs hatch in utero within the female worm), infection caused by the ingestion of embryonated eggs is unlikely. With an apparent single-host (monoxenous) life cycle, an arthropod vector is also unlikely. Ongoing release and maturation of larvae results in persistent infections.
Of the 13 known case-patients (Table), 12 had weakness and muscle wasting, 7 had dysphagia, and 2 had dysarthria or dysphonia. One case was discovered incidentally during evaluation of low back pain; the patient was otherwise asymptomatic. All case-patients had increased levels of creatinine kinase (270–6,218 U/L). Peripheral eosinophilia was observed in 12 (92%) of 13 patients. Myalgias, unintentional weight loss, increases in erythrocyte sedimentation rates, and mild-to-moderate increases in levels of liver aminotransferases were also common. Needle electromyography findings were available for 8 patients (patients 2, 4, 7, 8, 10, 11, 12, and 13). Except for patients 10 and 12, who had ambiguous or limited findings, the remaining patients had myopathic motor unit potentials. Results of nerve conduction studies were within reference ranges when described. The time from symptom onset to diagnosis ranged from 1.5 to 8 years, with the case-patient in this study having the longest known timeframe.
Seven patients had received corticosteroids at some point in their illness for a presumptive diagnosis of polymyositis, during which time most experienced progressive deterioration, including our patient. All patients were given extended, high-dose albendazole therapy, and 7 patients had a partial to near complete recovery. One patient (7) died from complications resulting from corticosteroid administration, mechanical ventilation, and a prolonged stay in the intensive care unit.
Diagnosis of haycocknematosis is based primarily on histopathologic features. The morphologic characteristics of H. perplexum nematodes in histopathologic preparations include a thin cuticle, meromyarian/platymyarian musculature, amphidelphic uteri (females), lateral bacillary bands (especially conspicuous in immature females), and conspicuous subventral glands (10). There are no cephalic inflations or lateral alae. Adult males, adult females, and larvae might be observed in muscle specimens, but never in ex utero eggs. Adults often have an undulating, serpentine morphology, which is parallel with the muscle fibers. Male worms have a maximum width of 15 μm (range 14–15 μm), and female worms have a maximum width of 36 μm (range 15–36 μm) (10). Larvae are similar in size to adult males, have a maximum width of 15 μm (range 12–15 μm) (10), and complete their lifecycle in the host.
Although other parasites in biopsy specimens include Trichinella spp., Strongyloides stercoralis, and Halicephalobus gingivalis, these parasites can be differentiated by morphologic, clinical, and epidemiologic features. H. perplexum and other nematodes might also be potentially confused for tissue cysts of Toxoplasma gondii and Sarcocystis spp. when seen in cross-section (as in our case-patient), but this finding can usually be resolved by examining deeper sections from the tissue block to identify additional parasite forms.
A PCR was developed that enabled diagnosis of the 10th case of H. perplexum nematode infection from a muscle biopsy in the absence of visible nematodes (2,8). This PCR was unsuccessful when performed for our case-patient. However, this result is not unexpected, given the age of the block (7 years at time of testing) and the relatively large sizes of the PCR amplicons (400 bp for cytochrome c oxidase subunit 1 and 830 bp for 18S rRNA).
It is unknown why to date human cases appear to have been acquired only in Tasmania or the northern regions of Queensland. Molecular sequence data demonstrate that the strains from Queensland and Tasmania belong to the same species (2). Infections might occur in other areas of the East Coast of Australia, or wider mainland Australia, but these infections have not been detected because of lack of awareness and difficulty in diagnosis.
The optimal antimicrobial drug management for treatment of haycocknematosis is unknown. Our patient was given albendazole based on experiences from previously reported cases. In 1 case, viable nematodes were still observed after 4 weeks of treatment, but not after 9 weeks (7). Additional studies are needed to determine the most efficacious antiparasitic treatment for haycocknematosis.
Patients who have H. perplexum parasitic myositis might be a diagnostic challenge to clinicians and pathologists, particularly when seen outside disease-endemic regions. The disease is progressive, potentially life-threatening, and might persist for >8 years with a delayed diagnosis, as shown by our case-patient. A high degree of suspicion is required to diagnose this treatable mimic of muscular dystrophy and inflammatory myopathy, and to avoid harm through corticosteroid treatment. Additional studies are needed to clarify the exposure risks, parasite life cycle, disease prevention, and treatment.
Dr. Pritt is a professor of laboratory medicine and pathology at the Mayo Clinic, Rochester, MN. Her primary research interests are clinical parasitology, vector-borne diseases, and the pathology of infectious diseases.
References
- Dennett X, Siejka SJ, Andrews JR, Beveridge I, Spratt DM. Polymyositis caused by a new genus of nematode. Med J Aust. 1998;168:226–7. DOIPubMedGoogle Scholar
- Koehler AV, Spratt DM, Norton R, Warren S, McEwan B, Urkude R, et al. More parasitic myositis cases in humans in Australia, and the definition of genetic markers for the causative agents as a basis for molecular diagnosis. Infect Genet Evol. 2016;44:69–75. DOIPubMedGoogle Scholar
- Vos LJ, Robertson T, Binotto E. Haycocknema perplexum: an emerging cause of parasitic myositis in Australia. Commun Dis Intell Q Rep. 2016;40:E496–9.PubMedGoogle Scholar
- Haycocknema perplexum: life threatening disease of humans, 2018 [cited 2022 Jan 2]. https://www.wildlifehealthaustralia.com.au/Portals/0/Documents/FactSheets/Public%20health/Haycocknema%20perplexum.pdf
- Andrews JR, Ainsworth R, Abernethy D. Trichinella pseudospiralis in humans: description of a case and its treatment. Trans R Soc Trop Med Hyg. 1994;88:200–3. DOIPubMedGoogle Scholar
- Andrews JR, Ainsworth R, Pozio E. Nematodes in human muscle. Parasitol Today. 1997;13:488–9. DOIPubMedGoogle Scholar
- Basuroy R, Pennisi R, Robertson T, Norton R, Stokes J, Reimers J, et al. Parasitic myositis in tropical Australia. Med J Aust. 2008;188:254–6. DOIPubMedGoogle Scholar
- Koehler AV, Leung P, McEwan B, Gasser RB. Using PCR-based sequencing to diagnose Haycocknema perplexum infection in human myositis case, Australia. Emerg Infect Dis. 2018;24:2368–70. DOIPubMedGoogle Scholar
- McKelvie P, Reardon K, Bond K, Spratt DM, Gangell A, Zochling J, et al. A further patient with parasitic myositis due to Haycocknema perplexum, a rare entity. J Clin Neurosci. 2013;20:1019–22. DOIPubMedGoogle Scholar
- Spratt DM, Beveridge I, Andrews JR, Dennett X. Haycocknema perplexum n. g., n. sp. (Nematoda: Robertdollfusidae): an intramyofibre parasite in man. Syst Parasitol. 1999;43:123–31. DOIPubMedGoogle Scholar
- Ward K, Krishnan A, R Iyengar K, Robertson T, White R, Urkude R. Haycocknema perplexum myositis: the first description of subclinical disease and a proposed distinctive triad to evoke clinical suspicion. BMJ Neurol Open. 2022;4:
e000290 . DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: September 28, 2022
1This study was presented as a late breaker abstract at the 68th Annual Meeting of the American Society of Tropical Medicine and Hygiene, November 20‒24, 2019, National Harbor, Maryland, USA.
Table of Contents – Volume 28, Number 11—November 2022
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Bobbi S. Pritt, Division of Clinical Microbiology, Department of Laboratory Medicine and Pathology, Mayo Clinic, 200 1st St SW, Hilton Bldg 470-B, Rochester, MN 55905, USA
Top