Volume 28, Number 3—March 2022
Research
Plasmodium falciparum pfhrp2 and pfhrp3 Gene Deletions from Persons with Symptomatic Malaria Infection in Ethiopia, Kenya, Madagascar, and Rwanda
Cite This Article
Citation for Media
Abstract
Histidine-rich protein 2 (HRP2)–based rapid diagnostic tests detect Plasmodium falciparum malaria and are used throughout sub-Saharan Africa. However, deletions in the pfhrp2 and related pfhrp3 (pfhrp2/3) genes threaten use of these tests. Therapeutic efficacy studies (TESs) enroll persons with symptomatic P. falciparum infection. We screened TES samples collected during 2016–2018 in Ethiopia, Kenya, Rwanda, and Madagascar for HRP2/3, pan-Plasmodium lactate dehydrogenase, and pan-Plasmodium aldolase antigen levels and selected samples with low levels of HRP2/3 for pfhrp2/3 genotyping. We observed deletion of pfhrp3 in samples from all countries except Kenya. Single-gene deletions in pfhrp2 were observed in 1.4% (95% CI 0.2%–4.8%) of Ethiopia samples and in 0.6% (95% CI 0.2%–1.6%) of Madagascar samples, and dual pfhrp2/3 deletions were noted in 2.0% (95% CI 0.4%–5.9%) of Ethiopia samples. Although this study was not powered for precise prevalence estimates, evaluating TES samples revealed a low prevalence of pfhrp2/3 deletions in most sites.
The World Health Organization (WHO) estimates there were 228 million cases of malaria in 2019, which resulted in 409,000 deaths; >90% of these deaths occurred in sub-Saharan Africa (1). Although all 4 human malaria Plasmodium species are present in Africa, Plasmodium falciparum accounts for most symptomatic infections (1). After the WHO recommended confirming Plasmodium infection before initiating treatment (2), malaria rapid diagnostic tests (RDTs) have been widely deployed because of their ease of use and high diagnostic sensitivity for symptomatic infection (3–5). The histidine-rich protein 2 (HRP2) antigen is produced exclusively by P. falciparum parasites, and RDTs detecting this antigen provide a practical tool for diagnosis in both healthcare and community settings (1,3,6) and have revolutionized the diagnosis of malaria throughout Africa.
HRP2-based RDTs are an accurate diagnostic tool because HRP2 is abundantly expressed during the erythrocytic stage of P. falciparum infection (6). The pfhrp3 gene is paralogous to pfhrp2 and has a high level of similarity in both gene sequence and the expressed histidine-rich protein 3 (HRP3) antigen, although the HRP3 antigen is substantially shorter in length (7,8). However, because of common epitopes on both antigens, they jointly contribute to an overall HRP2-based RDT positive result or laboratory assay signal (6,9). In many areas of the world, P. falciparum variants have been identified with loss-of-function mutations or complete deletions of the pfhrp2 and pfhrp3 (pfhrp2/3) genes, which lead to false-negative RDT results (6,10). Multiple countries in sub-Saharan Africa have reported the presence of P. falciparum with deletions in these genes (9,11–16), although only Eritrea and Djibouti have reported a prevalence of >5% among isolates from symptomatic infections (17,18).
WHO recommends routine therapeutic efficacy studies (TESs) approximately every 2 years in malaria-endemic countries to assess antimalarial drug efficacy, and US President’s Malaria Initiative funding ensures these studies routinely occur in many countries throughout sub-Saharan Africa (19). According to established WHO protocol (20), symptomatic patients with uncomplicated P. falciparum malaria are enrolled in healthcare facilities after infection is verified by light microscopy examination of a blood smear. In addition, on the day of enrollment and subsequent follow-up days, a blood sample from a finger prick is dried on filter paper to form a dried blood spot (DBS) to monitor chemotherapeutic efficacy and test for putative drug resistance genetic markers (19). TESs are often implemented at multiple sites in a country because efficacy might vary depending on local endemicity, P. falciparum haplotypes, and antimalarial use.
We investigated deletions in pfhrp2/3 genes by using samples from TESs in Ethiopia (2017), Kenya (2016–2017), Madagascar (2018), and Rwanda (2018). DBS samples from day of enrollment were subjected to multiplex antigen detection and subsequent PCR assays if pfhrp2/3 genotyping was warranted on the basis of the antigen profile.
Therapeutic Efficacy Studies
This study focuses on TESs in 4 countries: Ethiopia (enrollment during September–December 2017) (21), Kenya (enrollment during June 2016–March 2017) (22), Madagascar (enrollment during May–September 2018) (23), and Rwanda (enrollment during May–December 2018) (24). Specific site information and enrollment criteria are provided for each TES by the indicated reference. Of note, enrollment criteria in the Madagascar TES included a positive HRP2-based RDT result. CDC human subjects review for laboratory analyses for all TES samples were determined independently for each study: Ethiopia as engaged research (#6892.0), Rwanda as program evaluation (#2018-060), Madagascar as nonengaged research (#2018-435), and Kenya as engaged research (#6696.0).
Bead-Based Multiplex Assay for Malaria Antigen Detection
All DBS samples were processed and analyzed within 1 year of creation. We performed elution of whole blood from DBS samples and the bead-based multiplex assay for malaria antigen detection as described previously (25) (Appendix). Differences among parasite densities or antigen levels were assessed by Student t test for unequal variances using the log-transformed data.
Selection of Samples for Further Genetic Assays
Using the strategy reported previously (26,27), we selected samples for further genetic assays on the basis of the relationship between the 2 pan-Plasmodium antigens (aldolase and lactate dehydrogenase [LDH]) and the HRP2/3 signal. Samples were selected if they completely lacked an assay signal for HRP2/3 or if the assay signal for HRP2/3 was atypically lower compared with the level of pAldolase or pLDH antigens.
We extracted total genomic DNA from 6-mm punches of selected DBS samples by using the QIAGEN DNA extraction kit (QIAGEN, https://www.qiagen.com) following the manufacturer’s instructions for blood dried on filter paper. The DNA was eluted in 150 μL of elution buffer, aliquoted, and stored at −20°C until further use.
Photo-Induced Electron Transfer PCR and Genotyping for pfmsp1, pfmsp2, pfhrp2, and pfhrp3
After DNA extraction, we performed photo-induced electron transfer PCR as described previously (28) to ensure presence of P. falciparum DNA. We used nested PCR to genotype pfmsp1, pfmsp2, and pfhrp3 as described previously (29). For pfhrp2 genotyping, we performed PCR on these samples under conditions described previously (30). Results for pfhrp2/3 genotyping were only reported if both pfmsp1 and pfmsp2 (both single-copy genes in the P. falciparum genome) were successfully amplified for a DNA sample (31).
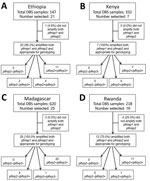
The Kenya 2016–2017 TES had the fewest number of sites at 1, followed by the Ethiopia 2017 TES at 2, the Rwanda 2018 TES at 3, and the Madagascar 2018 TES at 5 (Figure 1). The number of participants providing DBS samples from each site at enrollment varied for each of the 4 countries and ranged from a low of 15 participants at the Arba Minch, Ethiopia, site to a high of 332 participants at the Siaya, Kenya, site (Table 1). Reflecting the different enrollment criteria for each TES, the median and range of participant ages were unique to each TES; median age was 18.0 years in Ethiopia, 2.7 years in Kenya, 7.0 years in Madagascar, and 3.3 years in Rwanda. Enrollment by sex was mostly equal for the Kenya, Madagascar, and Rwanda TESs; 48.8% (Kenya), 47.1% (Madagascar), and 50.9% (Rwanda) of participants providing DBS samples in these studies were women. Enrollment of women in the Ethiopia TES was notably lower at 32.9%.
The antigen screening methodology provided phenotypic rationale for categorizing the infecting P. falciparum as a high producer of HRP2/3 antigens or a HRP2/3 low-producer requiring subsequent characterization though genetic assays (25,26). Correlation of antigen assay signal with parasite density (as determined by microscopy during enrollment for each TES) (Appendix Figure 1) showed that the 2 pan-Plasmodium antigens displayed a moderate correlation with microscopy-estimated P. falciparum parasite density, whereas the HRP2 antigen showed higher variability, as seen previously (25). We compared the pAldolase and pLDH assay signal to the HRP2 assay signal for all samples from each of the 4 countries and chose select samples for DNA extraction (Table 2; Appendix Figures 2–5). Ethiopia had the highest percentage (n = 21, 14.3% of all DBS samples) of samples selected for DNA extraction and PCR genotyping, followed by Rwanda (n = 16, 7.3%), Madagascar (n = 25, 4.0%), and Kenya (n = 7, 2.1%).
After initial sample selection for genotyping, we evaluated DNA quantity and quality appropriate for genotyping by amplification of both pfmsp1 and pfmsp2 genes. Final pfhrp2/3 genotyping results were evaluated for amplification of (+) or failure to amplify (–) these 2 different gene targets (Figure 2). Only samples from which both the single-copy pfmsp1 and pfmsp2 genes were successfully amplified had pfhrp2/3 genotype reported (31); from all selected samples, only 1 sample from Ethiopia and 4 samples from Rwanda were unsuccessfully amplified for these control genes. Most selected samples (76.6%) showed a wild-type genotype of pfhrp2+/pfhrp3+. Single gene deletions were observed in samples from 3 countries: single gene pfhrp2 deletions from Ethiopia and Madagascar, and single gene pfhrp3 deletions from Ethiopia, Madagascar, and Rwanda (Table 3). The pfhrp2–/pfhrp3– double deletion genotype was observed only in Ethiopia; 3 of the 20 samples selected, all from the Pawe site, showed this genotype. Because the pfhrp3 nested PCR includes a reaction for an exon 1-to-2 spanning primer and a separate reaction for an exon 2 primer, a nested PCR reaction could have amplified one of these targets and not the other if the gene was not fully deleted from the genome. For 9 samples classified as negative for the pfhrp3 gene, both of these nested PCR targets failed to amplify in all, with the exception of a single sample from Ethiopia (exon 1–2 target did not amplify and exon 2 target did; the sample was positive for the pfhrp2 gene).
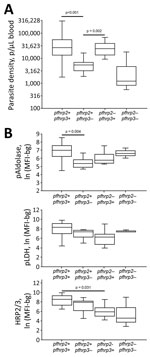
In an exploratory analysis of the 64 samples that were successfully genotyped, different pfhrp2/3 genotype combinations showed significant differences in microscopy-estimated parasite densities (Figure 3, panel A). In comparison to wild-type parasites, significantly lower parasite densities were observed in infections with P. falciparum lacking the pfhrp3 gene alone. Parasites lacking the pfhrp2 gene alone showed significantly higher mean parasite densities when compared with the pfhrp3 single-deleted infections. To link the phenotypic data of antigen expression with the pfhrp2/3 genotypic data, we plotted the antigen detection assay signal by genotype for the 64 total samples that underwent successful genotyping. Assay signals for pan-Plasmodium aldolase, pan-Plasmodium LDH, and HRP2/3 by the 4 potential combinations of pfhrp2/3 genotypes are given (Figure 3, panel B). With loss of either of the pfhrp2 or pfhrp3 genes and loss of both, no overall trend was observed for changes in pAldolase or pLDH signal, although the numbers of each of these genotypes were small. However, we observed a lower HRP2/3 assay signal with either the loss of the pfhrp3 gene or the pfhrp2 gene; the lowest mean HRP2/3 assay signal occurred when both genes were absent.
Deletion of the pfhrp2 and pfhrp3 genes poses a threat to the accuracy of HRP2-based RDT diagnosis of P. falciparum malaria, and parasites with deletions in one or both these genes have now been found in numerous countries (6,10). By far, most malaria cases in Africa are caused by P. falciparum, and the presence of these deletion genotypes in many countries throughout the continent poses an additional challenge to malaria control because of false-negative diagnostic results (1). Most countries in Africa have adopted the HRP2-based RDT as a pragmatic and sensitive diagnostic tool and the only P. falciparum-specific diagnostic test available in many settings. Loss of this tool would be a substantial setback to accurate monitoring of malaria case incidence within a country and to achieving the goal of universal confirmation of malaria infection before administrating antimalarials (3).
In this study, we sought to identify the presence of deletions in either the pfhrp2 or pfhrp3 genes from samples collected during routine TESs that enroll participants with microscopically confirmed P. falciparum infection. The primary objective of a TES is to assess in vivo efficacy of antimalarials, and some studies have also investigated the presence of P. falciparum drug resistance genetic markers (19). Because DBS samples are collected for many of these TESs, residual patient samples represent a convenience sampling of known P. falciparum infections with estimated parasite densities, and enrollment at healthcare facilities conforms to the WHO pfhrp2 deletion guidance to sample symptomatic patients (32). Quantitative detection of malaria antigens in these DBS samples not only enables the confirmation of the presence or absence of HRP2, HRP3, or both in the patient’s blood sample, it also enables the simultaneous detection of other Plasmodium antigens for comparison. For these 4 TESs, a total of 1,317 DBS samples were available, and performing genetic characterization for all these samples would have required a large time and financial commitment. However, by initially employing a low-cost, high-throughput antigen screening step, fewer samples can be carefully selected for more definitive investigation into production of these RDT targets (25–27). This strategy of phenotypic screening and genetic confirmation is not unique for the TES sampling design and has also been used for healthcare facility (25,27) and community (26) surveys. Further exploration of this strategy with large datasets is needed throughout global P. falciparum populations to determine the overall accuracy of this methodology and its ability to generalize antigen levels with deletions of pfhrp2 and pfhrp3.
Many TESs seek to enroll participants at multiple sites throughout a country to gain a more geographically representative sampling of P. falciparum for in vivo efficacy estimates. Of the data presented in this study, 3 of the 4 countries had multiple enrollment sites; only Kenya enrolled persons from just 1 site. Ultimately, high global variation has been observed in pfhrp2 gene sequences (7,33,34), and deletions in the pfhrp2 and pfhrp3 genes can arise de novo in a P. falciparum population (18,35). Therefore, presence (or absence) of these gene deletions could not be accurately ascertained for an entire country by sampling a limited number of sites. Recent WHO guidance recommended enrolling from >10 health facilities per province to estimate whether pfhrp2 deletions exceed 5% of all P. falciparum infections for a country (32). Because TESs do not enroll at many study sites, the data presented in this study do not provide country-representative or even precise local estimates of gene deletion prevalence, but they generate a data signal to point toward the presence of deletions at a site of a previous TES. Troublesome data signals generated from TES samples could be followed up with a more thorough study, such as the WHO-recommended approach of enrolling from a minimum of 10 health facilities per province to estimate whether pfhrp2 deletions exceed 5% of all P. falciparum infections for a country. A benefit of this sampling design is that TESs are routinely performed in countries that receive support from the US President’s Malaria Initiativeevery 2–3 years (19); consistently collecting quantitative antigen data from these sample sets will provide a longitudinal approach to better identify emerging deletion genotypes in a country.
Samples with an absence of both the pfhrp2 and pfhrp3 are of greatest concern because these 2 genes express the only antigen targets recognized by an HRP2-based RDT. For the 69 samples selected for genotyping on the basis of antigen profile, only 3 double-deletions were noted, all arising from the Pawe study site in the Benishangul-Gumuz region in northeastern Ethiopia. As an external evaluation activity, persons enrolling in the Ethiopia TES were also tested by HRP2-based RDTs, and of the 3 persons with double-deleted P. falciparum infections, 2 tested negative by the HRP2 band on the RDT. These same 2 persons had a complete absence of HRP2/3 antigens by the bead-based assay, whereas the third double-deleted infection had an HRP2/3 antigen concentration in blood of 27.5 ng/mL. An additional 2 samples from Pawe were also found to be deleted for the pfhrp2 gene alone (both of these persons were HRP2-RDT positive), meaning of the 132 total P. falciparum isolates available from Pawe, 3.8% (95% CI 1.2%–8.6%) showed a deletion of the pfhrp2 gene. Multiple recent reports have uncovered the presence of pfhrp2/3 deletions in central (36) and northern (37,38) Ethiopia at levels above the 5% WHO recommendation to reevaluate national RDT selection (32). The data presented in this study do not attempt to provide a prevalence estimate of single- or double-deletion P. falciparum genotypes in Ethiopia, but they add to the growing evidence of the pervasiveness of these parasites lacking pfhrp2 in the country by demonstrating their presence in 2017.
Deletions in pfhrp2 were also observed in samples from Madagascar, a country with numerous haplotypes circulating according to previous studies (39,40). Even with the identification of pfhrp2 deletions in Madagascar, these cases represent a very small proportion (4 of 620, 0.6%) of all P. falciparum–infected children providing DBS samples for this study. Three of the 4 pfhrp2-deleted samples came from the Ankazomborona study site in the northern part of the country, which could provide rationale for further investigation of deletions in this part of Madagascar. Of note, the Madagascar TES had enrollment criteria of positivity to both microscopy and HRP2-based RDT. Because infections in persons with double-deleted parasites would likely have been excluded from enrollment, these data should be taken in that context.
Separate investigations have (9) and have not (7) detected pfhrp2 and pfhrp3 deletions in Kenya. From the 332 DBS samples available from the 2016–2017 Kenya TES, most P. falciparum infections produced high amounts of HRP2/3 antigens, and no phenotypic or genotypic evidence was seen for gene deletions of these targets. A single report from Rwanda also identified nonamplification of the pfhrp2 gene from microscopically positive P. falciparum infections, although the primers were only targeting the exon 2 of the gene (13), which is not crucial for antigen expression. In this study, no deletions of the pfhrp2 gene were identified in Rwanda, and only 1 P. falciparum isolate was found with a pfhrp3 deletion.
Existence of HRP2/3 antigens in a blood sample does not necessarily indicate that the currently infecting P. falciparum parasite possesses functioning pfhrp2/3 genes. The bead assay limit of detection is ≈10 pg/mL, and HRP2 antigen can remain in blood for months after successful treatment of a P. falciparum infection (6,41). A person could therefore be actively infected with a deleted strain but have HRP2 antigen in their blood from a previous infection (although the levels would be expected to be atypically low in this scenario). Because of the phenotypic selection criteria outlined in this study, infections with high levels of HRP2/3 would not be selected for genotyping but might still harbor parasites with deletions of the pfhrp2/3 genes and would not be captured, although this possibility is likely low. If deleted parasites were more likely to induce asymptomatic or less symptomatic infections, this enrollment criteria in healthcare facilities would lead to underestimating actual deletion prevalence in a population, although data have not demonstrated this effect. Simultaneous infection with multiple P. falciparum haplotypes was also not investigated in this study, so the presence of deleted parasites could be masked by the presence of wild-type parasites in the same host (42). These 2 scenarios would be more probable in a higher-transmission setting, where the likelihood for residual HRP2/3, as well as higher multiplicity of infection and more frequent infections, would be more common. In addition, the genetic assays used in this study attempted to simply amplify a region of DNA and do not provide information regarding potential loss-of-function point mutations or other genetic scenarios which would cause these 2 antigens not to be expressed. Because antigen degradation might occur in DBS samples over time, quantitative antigen detection should occur as soon as possible.
In conclusion, with appropriate patient consent, screening samples that were previously collected for routine TESs for pfhrp2 and pfhrp3 deletions represent a useful convenience sampling of persons with symptomatic and microscopically confirmed P. falciparum infection. These phenotypic and genotypic data provide information for a country to evaluate whether these genotypes exist and promote a basis for more targeted future surveys to obtain precise point estimates of prevalence.
Dr. Rogier is a microbiologist in the Malaria Branch, Division of Parasitic Diseases and Malaria, Center for Global Health, Centers for Disease Control and Prevention. His current research interests focus on multiplex IgG and antigen detection assays for malaria surveillance purposes and integrated disease serosurveys.
Acknowledgments
We thank Veronika Laird for her help with creating the maps in Figure 1.
This study was funded by the US President’s Malaria Initiative. J.H., J.M., S.S.S., and E.S.H. receive salary support from the US President’s Malaria Initiative.
The findings and conclusions in this report are those of the author(s) and do not necessarily represent the official position of the Centers for Disease Control and Prevention or US President’s Malaria Initiative. The manuscript was published with the permission of the Director, KEMRI.
References
- World Health Organization. World malaria report 2020. Geneva: The Organization; 2020.
- World Health Organization. Guidelines for the treatment of malaria: 2nd edition. Geneva: The Organization; 2010.
- World Health Organization. Universal access to malaria diagnostic testing: an operational manual. Geneva: The Organization; 2011.
- Harvey SA, Bell D. How to use a rapid diagnostic test (Generic Pf): a guide for training at the village and clinic level. Geneva: World Health Organization; 2008.
- Plucinski M, Dimbu R, Candrinho B, Colborn J, Badiane A, Ndiaye D, et al. Malaria surveys using rapid diagnostic tests and validation of results using post hoc quantification of Plasmodium falciparum histidine-rich protein 2. Malar J. 2017;16:451. DOIPubMedGoogle Scholar
- Poti KE, Sullivan DJ, Dondorp AM, Woodrow CJ. HRP2: transforming malaria diagnosis, but with caveats. Trends Parasitol. 2020;36:112–26. DOIPubMedGoogle Scholar
- Nderu D, Kimani F, Thiong’o K, Karanja E, Akinyi M, Too E, et al. Plasmodium falciparum histidine-rich protein (PfHRP2 and 3) diversity in Western and Coastal Kenya. Sci Rep. 2019;9:1709. DOIPubMedGoogle Scholar
- Fontecha G, Pinto A, Escobar D, Matamoros G, Ortiz B. Genetic variability of Plasmodium falciparum histidine-rich proteins 2 and 3 in Central America. Malar J. 2019;18:31. DOIPubMedGoogle Scholar
- Beshir KB, Sepúlveda N, Bharmal J, Robinson A, Mwanguzi J, Busula AO, et al. Plasmodium falciparum parasites with histidine-rich protein 2 (pfhrp2) and pfhrp3 gene deletions in two endemic regions of Kenya. Sci Rep. 2017;7:14718. DOIPubMedGoogle Scholar
- Thomson R, Parr JB, Cheng Q, Chenet S, Perkins M, Cunningham J. Prevalence of Plasmodium falciparum lacking histidine-rich proteins 2 and 3: a systematic review. Bull World Health Organ. 2020;98:558–568F. DOIPubMedGoogle Scholar
- Koita OA, Doumbo OK, Ouattara A, Tall LK, Konaré A, Diakité M, et al. False-negative rapid diagnostic tests for malaria and deletion of the histidine-rich repeat region of the hrp2 gene. Am J Trop Med Hyg. 2012;86:194–8. DOIPubMedGoogle Scholar
- Wurtz N, Fall B, Bui K, Pascual A, Fall M, Camara C, et al. Pfhrp2 and pfhrp3 polymorphisms in Plasmodium falciparum isolates from Dakar, Senegal: impact on rapid malaria diagnostic tests. Malar J. 2013;12:34. DOIPubMedGoogle Scholar
- Kozycki CT, Umulisa N, Rulisa S, Mwikarago EI, Musabyimana JP, Habimana JP, et al. False-negative malaria rapid diagnostic tests in Rwanda: impact of Plasmodium falciparum isolates lacking hrp2 and declining malaria transmission. Malar J. 2017;16:123. DOIPubMedGoogle Scholar
- Funwei R, Nderu D, Nguetse CN, Thomas BN, Falade CO, Velavan TP, et al. Molecular surveillance of pfhrp2 and pfhrp3 genes deletion in Plasmodium falciparum isolates and the implications for rapid diagnostic tests in Nigeria. Acta Trop. 2019;196:121–5. DOIPubMedGoogle Scholar
- Parr JB, Verity R, Doctor SM, Janko M, Carey-Ewend K, Turman BJ, et al. Pfhrp2-deleted Plasmodium falciparum parasites in the Democratic Republic of the Congo: a national cross-sectional survey. J Infect Dis. 2017;216:36–44.PubMedGoogle Scholar
- Thomson R, Beshir KB, Cunningham J, Baiden F, Bharmal J, Bruxvoort KJ, et al. pfhrp2 and pfhrp3 gene deletions that affect malaria rapid diagnostic tests for Plasmodium falciparum: analysis of archived blood samples from 3 African countries. J Infect Dis. 2019;220:1444–52. DOIPubMedGoogle Scholar
- Iriart X, Menard S, Chauvin P, Mohamed HS, Charpentier E, Mohamed MA, et al. Misdiagnosis of imported falciparum malaria from African areas due to an increased prevalence of pfhrp2/pfhrp3 gene deletion: the Djibouti case. Emerg Microbes Infect. 2020;9:1984–7. DOIPubMedGoogle Scholar
- Berhane A, Anderson K, Mihreteab S, Gresty K, Rogier E, Mohamed S, et al. Major threat to malaria control programs by Plasmodium falciparum lacking histidine-rich protein 2, Eritrea. Emerg Infect Dis. 2018;24:462–70. DOIPubMedGoogle Scholar
- Halsey ES, Venkatesan M, Plucinski MM, Talundzic E, Lucchi NW, Zhou Z, et al. Capacity Development through the US President’s Malaria Initiative-Supported Antimalarial Resistance Monitoring in Africa Network. Emerg Infect Dis. 2017;23:23. DOIPubMedGoogle Scholar
- World Health Organization. Methods for surveillance of antimalarial drug efficacy. Geneva: The Organization; 2009.
- Leonard CM, Mohammed H, Tadesse M, McCaffery JN, Nace D, Halsey ES, et al. Missed Plasmodium falciparum and Plasmodium vivax mixed infections in Ethiopia threaten malaria elimination. Am J Top Med Hyg. 2021 Nov 30 [Epub ahead of print].
- Chebore W, Zhou Z, Westercamp N, Otieno K, Shi YP, Sergent SB, et al. Assessment of molecular markers of anti-malarial drug resistance among children participating in a therapeutic efficacy study in western Kenya. Malar J. 2020;19:291. DOIPubMedGoogle Scholar
- Dentinger CM, Rakotomanga TA, Rakotondrandriana A, Rakotoarisoa A, Rason MA, Moriarty LF, et al. Efficacy of artesunate-amodiaquine and artemether-lumefantrine for uncomplicated Plasmodium falciparum malaria in Madagascar, 2018. Malar J. 2021;20:432. DOIPubMedGoogle Scholar
- Uwimana A, Umulisa N, Venkatesan M, Svigel SS, Zhou Z, Munyaneza T, et al. Association of Plasmodium falciparum kelch13 R561H genotypes with delayed parasite clearance in Rwanda: an open-label, single-arm, multicentre, therapeutic efficacy study. Lancet Infect Dis. 2021;21:1120–8. DOIPubMedGoogle Scholar
- Plucinski MM, Herman C, Jones S, Dimbu R, Fortes F, Ljolje D, et al. Screening for Pfhrp2/3-deleted Plasmodium falciparum, non-falciparum, and low-density malaria infections by a multiplex antigen assay. J Infect Dis. 2019;219:437–47. DOIPubMedGoogle Scholar
- Bakari C, Jones S, Subramaniam G, Mandara CI, Chiduo MG, Rumisha S, et al. Community-based surveys for Plasmodium falciparum pfhrp2 and pfhrp3 gene deletions in selected regions of mainland Tanzania. Malar J. 2020;19:391. DOIPubMedGoogle Scholar
- Herman C, Huber CS, Jones S, Steinhardt L, Plucinski MM, Lemoine JF, et al. Multiplex malaria antigen detection by bead-based assay and molecular confirmation by PCR shows no evidence of Pfhrp2 and Pfhrp3 deletion in Haiti. Malar J. 2019;18:380. DOIPubMedGoogle Scholar
- Lucchi NW, Narayanan J, Karell MA, Xayavong M, Kariuki S, DaSilva AJ, et al. Molecular diagnosis of malaria by photo-induced electron transfer fluorogenic primers: PET-PCR. PLoS One. 2013;8:
e56677 . DOIPubMedGoogle Scholar - Abdallah JF, Okoth SA, Fontecha GA, Torres RE, Banegas EI, Matute ML, et al. Prevalence of pfhrp2 and pfhrp3 gene deletions in Puerto Lempira, Honduras. Malar J. 2015;14:19. DOIPubMedGoogle Scholar
- Jones S, Subramaniam G, Plucinski MM, Patel D, Padilla J, Aidoo M, et al. One-step PCR: A novel protocol for determination of pfhrp2 deletion status in Plasmodium falciparum. PLoS One. 2020;15:
e0236369 . DOIPubMedGoogle Scholar - Cheng Q, Gatton ML, Barnwell J, Chiodini P, McCarthy J, Bell D, et al. Plasmodium falciparum parasites lacking histidine-rich protein 2 and 3: a review and recommendations for accurate reporting. Malar J. 2014;13:283. DOIPubMedGoogle Scholar
- World Health Organization. Response plan to pfhrp2 gene deletions. Geneva: The Organization; 2019.
- Lee N, Baker J, Andrews KT, Gatton ML, Bell D, Cheng Q, et al. Effect of sequence variation in Plasmodium falciparum histidine- rich protein 2 on binding of specific monoclonal antibodies: Implications for rapid diagnostic tests for malaria. J Clin Microbiol. 2006;44:2773–8. DOIPubMedGoogle Scholar
- Lee N, Gatton ML, Pelecanos A, Bubb M, Gonzalez I, Bell D, et al. Identification of optimal epitopes for Plasmodium falciparum rapid diagnostic tests that target histidine-rich proteins 2 and 3. J Clin Microbiol. 2012;50:1397–405. DOIPubMedGoogle Scholar
- Gamboa D, Ho MF, Bendezu J, Torres K, Chiodini PL, Barnwell JW, et al. A large proportion of P. falciparum isolates in the Amazon region of Peru lack pfhrp2 and pfhrp3: implications for malaria rapid diagnostic tests. PLoS One. 2010;5:
e8091 . DOIPubMedGoogle Scholar - Golassa L, Messele A, Amambua-Ngwa A, Swedberg G. High prevalence and extended deletions in Plasmodium falciparum hrp2/3 genomic loci in Ethiopia. PLoS One. 2020;15:
e0241807 . DOIPubMedGoogle Scholar - Alemayehu GS, Blackburn K, Lopez K, Cambel Dieng C, Lo E, Janies D, et al. Detection of high prevalence of Plasmodium falciparum histidine-rich protein 2/3 gene deletions in Assosa zone, Ethiopia: implication for malaria diagnosis. Malar J. 2021;20:109. DOIPubMedGoogle Scholar
- Girma S, Cheaveau J, Mohon AN, Marasinghe D, Legese R, Balasingam N, et al. Prevalence and epidemiological characteristics of asymptomatic malaria based on ultrasensitive diagnostics: a cross-sectional study. Clin Infect Dis. 2019;69:1003–10. DOIPubMedGoogle Scholar
- Willie N, Mehlotra RK, Howes RE, Rakotomanga TA, Ramboarina S, Ratsimbasoa AC, et al. Insights into the performance of SD Bioline Malaria Ag P.f/Pan rapid diagnostic test and Plasmodium falciparum histidine-rich protein 2 gene variation in Madagascar. Am J Trop Med Hyg. 2018;98:1683–91. DOIPubMedGoogle Scholar
- Baker J, Ho MF, Pelecanos A, Gatton M, Chen N, Abdullah S, et al. Global sequence variation in the histidine-rich proteins 2 and 3 of Plasmodium falciparum: implications for the performance of malaria rapid diagnostic tests. Malar J. 2010;9:129. DOIPubMedGoogle Scholar
- Plucinski MM, Dimbu PR, Fortes F, Abdulla S, Ahmed S, Gutman J, et al. Posttreatment HRP2 clearance in patients with uncomplicated Plasmodium falciparum malaria. J Infect Dis. 2018;217:685–92. DOIPubMedGoogle Scholar
- Koita OA, Doumbo OK, Ouattara A, Tall LK, Konaré A, Diakité M, et al. False-negative rapid diagnostic tests for malaria and deletion of the histidine-rich repeat region of the hrp2 gene. Am J Trop Med Hyg. 2012;86:194–8. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleOriginal Publication Date: February 16, 2022
Table of Contents – Volume 28, Number 3—March 2022
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Eric Rogier, Centers for Disease Control and Prevention, 1600 Clifton Rd NE, Mailstop D-67, Atlanta, GA, 30029-4027, USA
Top