Volume 28, Number 4—April 2022
Dispatch
Rigidoporus corticola Colonization and Invasive Fungal Disease in Immunocompromised Patients, United States
Cite This Article
Citation for Media
Abstract
We report 2 cases of Rigidoporus corticola (Oxyporus corticola) infection in humans in the United States. Clinical manifestations consisted of angioinvasive fungal sinusitis in 1 patient and pulmonary intracavitary fungus ball in the other patient. These cases illustrate previously undescribed clinicopathologic manifestations of infection by this filamentous basidiomycete in humans.
Rigidoporus corticola is a white-rot decay fungus of plants and is widely distributed in North America and Europe (1). R. corticola is a filamentous basidiomycete in the Agaricomycotina subphylum, also known as Oxyporus corticola because no morphologic differences or type species are in the same phylogenetic clade. Rigidoporus is the preferred name because it was described prior to Oxyporus (2,3).
Filamentous basidiomycetes occur in the environment as gilled mushrooms and shelf or bracket fungi (4). Filamentous basidiomycetes (other than R. corticola) have been documented in immunocompetent and immunocompromised persons (5,6), but R. corticola has not been established as an agent of human disease. We describe the clinical, microbiologic, and histopathologic features of 2 cases of human R. corticola infection in the United States (Table).
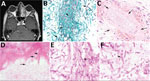
The first patient was a 43-year-old man with chronic granulomatous disease and recurrent acute myeloid leukemia in whom hemifacial pain developed after he received induction chemotherapy. Magnetic resonance imaging showed mucosal thickening and opacification of the patient’s left sinuses (Figure 1, panel A). Nasal endoscopy revealed a black eschar on the middle turbinate, which our clinical colleagues interpreted as highly characteristic of mucormycosis, and we performed surgical debridement. Histologic sections showed numerous thin hyaline septate hyphae (≈5 µm in diameter) with predominantly 90°-angle branching but no conidiogenous cells or conidia (Figure 1, panel B). We noted vascular invasion and infiltrative growth into sinonasal ossicles (Figure 1, panels C, D). Careful examination revealed bulge and hook-like hyphal outgrowths compatible with clamp connections (Figure 1, panels E, F), which are characteristic of filamentous basidiomycetes. Concurrent fungal culture on Sabouraud dextrose agar grew white cottony colonies. Lactophenol cotton blue preparation from subcultures onto potato flake agar revealed thin sterile hyphae. We sent a culture to the Fungus Testing Laboratory (FTL) at the University of Texas Health Science Center (San Antonio, Texas, USA). FTL also noted growth on benomyl agar suggesting a possible basidiomycete. FTL amplified genomic DNA from the isolate and sequenced for the internal transcribed spacer (ITS) and large subregion (D1/D2) of the 28S rRNA gene (BMBC-R/NL4). Compared with sequences from the NCBI database (https://www.ncbi.nlm.nih.gov), most blastn matches were R. corticola, 98.53%–100% similarity for ITS and 98.55%–100% for D1/D2; subsequent phylogenetic analysis confirmed R. corticola (Appendix). Antifungal susceptibility testing demonstrated similar MIC values for voriconazole (0.5 μg/mL) and isavuconazole (1 μg/mL) but a high MIC (8 μg/mL) for posaconazole. The patient was initially treated empirically with liposomal amphotericin B for 20 days, then was transitioned to voriconazole after fungal identification and susceptibility testing. However, the patient experienced recurrent episodes of neutropenic fever and died 10 months later of invasive fungal disease. We presumed the infection to be aspergillosis on the basis of increased serum galactomannan on sequential samples, but this assumption was not confirmed by culture or PCR.
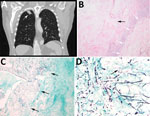
The second patient was a 63-year-old man with a history of lung adenocarcinoma and a new 2.4 cm right upper lobe mass resected for possible recurrence (Figure 2, panel A). Histologic sections showed a cavitary lesion with necrotic center and peripheral fibrous capsule (Figure 2, panel B). Sections stained with Gomori methenamine silver showed numerous septate hyphae in the cavity but no invasion into blood vessels or surrounding tissue (Figures 2, panels C, D). Neither conidiogenous cells nor conidia were evident; we did not identify clamp connections in samples after careful examination. The Centers for Disease Control and Prevention (Atlanta, Georgia, USA) identified O. corticola (R. corticola) by PCR from formalin-fixed, paraffin-embedded (FFPE) tissue sections of the wedge resection. We did not attempt culture, and the patient did not receive antifungal therapy. He died 11 months later of recurrent lung adenocarcinoma.
Filamentous basidiomycetes are common environmental fungi increasingly recognized as agents of human disease, causing saprobic colonization of the respiratory tract, allergic disease, and invasive fungal infections (6,7). A review of 218 pathogenic cases reported worldwide found the most common species are Schizophylum commune (52%), Hormographiella aspergillata (5.9%), and Ceriporia lacerata (5%) (7). Anecdotal reports describe galactomannan and β-d glucan production by filamentous basidiomycetes such as H. aspergillata, suggesting that testing for these polysaccharides might help diagnose invasive disease.
Two cases of R. corticola infection have been reported in canines. One was a disseminated R. corticola infection reported in 2009 in a German shepherd dog, which was ultimately euthanized 20 months after clinical presentation because of progressive disease and central nervous system involvement despite therapy with itraconazole and terbinafine (1). Another case, in 2012, was reported as generalized lymphadenopathy in a beagle, which was confirmed by sequencing of a culture isolate to be caused by R. corticola (8). This dog also was euthanized because of progressive disease despite empiric treatment with itraconazole 14 weeks after clinical presentation (8). Histopathology from both canine cases showed septate branching hyphae with parallel to slightly tapering walls.
We document R. corticola as an agent of human disease in 2 immunocompromised patients; the fungus colonized the pulmonary cavity of one patient and caused invasive sinusitis in the other. The spectrum of disease we describe resembles that observed for other filamentous basidiomycete fungi. Our findings align with the literature; these organisms are difficult to identify by in vitro culture because of lack of sporulation and are not yet reliably identifiable by mass spectrometry (9). Standard sequencing of 18S, 28S, or ITS rRNA and other fungal genomic regions can assist in species-level identification, but accurate identification is limited by available sequencing databases (10–12). However, molecular identification of fungi by itself is insufficient to establish pathogenicity because it might not distinguish between an agent of disease and a contaminant, particularly in cases involving common environmental fungi and uncommon species not established as agents of human disease, such as R. corticola. The 2 clinical cases we describe were detected by histopathology, confirming a pathogenic role in invasive fungal disease in 1 case and colonization of an existing cavity in the other, according to current guidelines (13); fungal sequencing of a culture isolate and FFPE tissue subsequently confirmed identification. These data support observations that filamentous basidiomycetes are being recognized more frequently in clinical specimens and that identification by fungal cultures and biochemical methods remains challenging. Consequently, filamentous basidiomycetes and their burden of disease likely are underrecognized.
Although we performed susceptibility testing against 1 isolate according to the Clinical and Laboratory Standards Institute M38 reference standard (10), no breakpoints for antifungal agents against this species have been established. Of note, the MIC for posaconazole was elevated compared with other basidiomycetes reported to cause human infections, such as S. commune. Although data are limited, of 5 R. corticola isolates in the FTL database with MIC results for posaconazole, 4 had elevated posaconazole MICs (range 2.0 to >16 μg/mL), but voriconazole MICs were lower (range 0.25–1.0 μg/mL). Lack of evidence-based treatment guidelines might have contributed to illness in these patients, both of whom died from complications of their underlying diseases <1 year after seeking care for R. corticola infection.
Although filamentous fungi cannot be speciated by histology, detection of the characteristic clamp connections should prompt consideration of filamentous basidiomycetes in the differential diagnosis. Admittedly, this finding might be rare because most clinical isolates are reportedly monokaryons (14), but 1 study documented clamp connections in 78% of clinical isolates of S. commune and C. lacerata (15).
In summary, our report adds R. corticola to the list of increasingly recognized filamentous basidiomycete fungi associated with human disease. Clinicians should consider R. corticola when assessing immunocompromised patients for invasive fungal infections or respiratory tract colonization.
Dr. Laga is an infectious disease pathology consultant at Brigham and Women’s Hospital in Boston, Massachusetts, USA. His primary research interests include medical mycology and tropical and neglected infectious diseases.
References
- Brockus CW, Myers RK, Crandell JM, Sutton DA, Wickes BL, Nakasone KK. Disseminated Oxyporus corticola infection in a German shepherd dog. Med Mycol. 2009;47:862–8. DOIPubMedGoogle Scholar
- Wu F, Chen JJ, Ji XH, Vlasák J, Dai YC. Phylogeny and diversity of the morphologically similar polypore genera Rigidoporus, Physisporinus, Oxyporus, and Leucophellinus. Mycologia. 2017;109:749–65. DOIPubMedGoogle Scholar
- Pouzar Z. Studies in the taxonomy of polypores 2. Folia Geobot Phytotaxon. 1966;1:356–75. DOIGoogle Scholar
- Lacaz CS, Heins-Vaccari EM, De Melo NT, Hernandez-Arriagada GL. Basidiomycosis: a review of the literature. Rev Inst Med Trop São Paulo. 1996;38:379–90. DOIPubMedGoogle Scholar
- Greer DL. Basidiomycetes as agents of human infections: a review. Mycopathologia. 1978;65:133–9. DOIPubMedGoogle Scholar
- Brandt ME. Filamentous basidiomycetes in the clinical laboratory. Curr Fungal Infect Rep. 2013;7:219–23. DOIPubMedGoogle Scholar
- Chowdhary A, Kathuria S, Agarwal K, Meis JF. Recognizing filamentous basidiomycetes as agents of human disease: A review. Med Mycol. 2014;52:782–97. DOIPubMedGoogle Scholar
- Miller SA, Roth-Johnson L, Kania SA, Bemis DA. Isolation and sequence-based identification of Oxyporus corticola from a dog with generalized lymphadenopathy. J Vet Diagn Invest. 2012;24:178–81. DOIPubMedGoogle Scholar
- McMullen AR, Wallace MA, Pincus DH, Wilkey K, Burnham CA. Evaluation of the Vitek MS matrix-assisted laser desorption ionization-time of flight mass spectrometry system for identification of clinically relevant filamentous fungi. J Clin Microbiol. 2016;54:2068–73. DOIPubMedGoogle Scholar
- Clinical and Laboratory Standards Institute. Interpretive criteria for identification of bacteria and fungi by targeted DNA sequencing, 2nd edition (MM18). Wayne (PA): The Institute; 2018.
- Lau A, Chen S, Sorrell T, Carter D, Malik R, Martin P, et al. Development and clinical application of a panfungal PCR assay to detect and identify fungal DNA in tissue specimens. J Clin Microbiol. 2007;45:380–5. DOIPubMedGoogle Scholar
- Romanelli AM, Sutton DA, Thompson EH, Rinaldi MG, Wickes BL. Sequence-based identification of filamentous basidiomycetous fungi from clinical specimens: a cautionary note. J Clin Microbiol. 2010;48:741–52. DOIPubMedGoogle Scholar
- Lockhart SR, Bialek R, Kibbler CC, Cuenca-Estrella M, Jensen HE, Kontoyiannis DP. Molecular techniques for genus and species determination of fungi from fresh and paraffin-embedded formalin-fixed tissue in the revised EORTC/MSGERC definitions of invasive fungal infection. Clin Infect Dis. 2021;72(Suppl 2):S109–13. DOIPubMedGoogle Scholar
- Sigler L, de la Maza LM, Tan G, Egger KN, Sherburne RK. Diagnostic difficulties caused by a nonclamped Schizophyllum commune isolate in a case of fungus ball of the lung. J Clin Microbiol. 1995;33:1979–83. DOIPubMedGoogle Scholar
- Singh PK, Kathuria S, Agarwal K, Gaur SN, Meis JF, Chowdhary A. Clinical significance and molecular characterization of nonsporulating molds isolated from the respiratory tracts of bronchopulmonary mycosis patients with special reference to basidiomycetes. J Clin Microbiol. 2013;51:3331–7. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: March 09, 2022
Table of Contents – Volume 28, Number 4—April 2022
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Alvaro C. Laga, Brigham and Women’s Hospital, Department of Pathology Amory-3, 75 Francis St, Boston, MA 02115, USA
Top