Volume 29, Number 6—June 2023
Research Letter
Isolated Ocular Mpox without Skin Lesions, United States
Cite This Article
Citation for Media
Abstract
We report a case of a 53-year-old HIV-negative patient in San Francisco, California, USA, with no classic mpox prodromal symptoms or skin lesions who experienced fulminant, vision-threatening scleritis, keratitis, and uveitis. Deep sequence analysis identified monkeypox virus RNA in the aqueous humor. We confirmed the virus on the cornea and sclera by PCR.
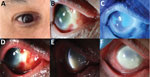
We report a case of ocular-only mpox infection in a 53-year-old man in San Francisco, California, USA. His medical history included chronic lymphocytic leukemia (CLL), inactive 2 years after treatment with obinutuzumab and venetoclax but with persistent lymphopenia. He reported male sexual partners but was HIV negative. Symptoms in his right eye began August 2022 as itching and nasal scleral redness (Figure, panel A). There was no fever, rash, or lymphadenopathy. Eye redness worsened; the patient sought care at an urgent care facility and was given erythromycin ointment. Continued vision loss led to an emergency department visit, resulting in a diagnosis of preseptal cellulitis, treated with was trimethoprim/sulfamethoxazole plus amoxicillin/clavulanic acid.
In early September 2022, the patient sought care at a county eye clinic for purulent conjunctivitis and corneal epithelial defects. Clinicians suspected gonococcal conjunctivitis and administered intramuscular ceftriaxone and topical moxifloxacin 0.5%. Bacterial and fungal ocular cultures and herpetic viral PCR returned negative results. Topical prednisolone acetate 1% and oral valacyclovir failed to control the eye inflammation. Three weeks after initial symptoms appeared, the patient’s ocular inflammation increased; keratic precipitates and a moderate corneal opacity developed. Uveitis and scleritis workups did not yield a specific diagnosis (Table).
In late September 2022, at a second opinion examination at a University of California clinic, the patient’s right eye acuity was 20/640. Examination showed a nasal patch of avascular scleral necrosis (Figure, panel B), and corneal epithelial sloughing (Figure, panel C) with microcystic edema. Repeat ocular surface cultures and PCR were negative. Given the negative results of extensive infectious etiology testing, we prescribed oral prednisone (40 mg/d) for presumed undifferentiated necrotizing anterior scleritis and keratitis.
One week later, corneal inflammation worsened (Figure, panel D). Clinical deterioration on systemic steroids continued to raise suspicion for ocular infection; we stopped steroid treatments. Again, cultures and PCR remained negative. Without prednisone, the patient’s limbal infiltrates worsened, with progressive corneal haze. White corneal endothelial plaques appeared. We performed anterior chamber paracentesis for cultures, viral PCR, and an RNA deep-sequencing (RNA-seq) protocol previously described (1). The patient’s scleritis, keratitis, and anterior uveitis worsened (Figure, panels E, F). Right eye vision decreased to hand motion only. We performed a diagnostic scleral and corneal biopsy and initiated voriconazole for presumed fungal infection. The biopsy results returned negative for infection, and voriconazole was stopped.
Given the progressive clinical disease and negative infectious workup, we processed residual aqueous fluid for RNA-seq, which revealed a high number of reads aligning to monkeypox virus (MPXV) (Appendix Figure 1). Orthogonal testing from corneal and scleral swab specimens also returned positive results for MPXV by PCR. A nasal swab specimen obtained on the same day tested negative for MPXV by PCR.
When presented with those findings (Table), the patient offered additional information regarding high-risk sexual activities, previously withheld because his definition of sexual activity included only penetrative intercourse. He described an encounter with a male partner with semen deposition into his right eye 2 weeks before the onset of his symptoms. He received the first dose of mpox vaccine for higher risk groups the week after this encounter. Several months later, during casual text messaging, the partner from this activity revealed a subsequent penile mpox diagnosis. (Appendix Figure 2).
Following treatment guidance from the Centers for Disease Control and Prevention, we initiated oral tecovirimat (600 mg 2×/d), topical trifluridine (6×/d), and weekly topical ophthalmic betadine washes (2). Four months into this course, worsening vision to light perception and repeat positive conjunctival mpox PCR led to inpatient admission for intravenous tecovirimat and cidofovir. Used in a compassionate-use capacity, tecovirimat and cidofovir have shown good activity against other orthopoxviruses, and in vitro and animal models have shown the antivirals to be effective against mpox (3). Unfortunately, after 3 weeks of inpatient treatment, the ocular disease did not improve. The patient’s eye remained PCR positive for MPXV with an opacified and vascularized cornea.
Ocular manifestations of mpox during the 2010–2013 outbreak were associated with a more severe systemic presentation (4). However, during the recent 2022 outbreak, ocular findings do not appear to have correlated with systemic disease severity (5–7). Isolated ocular mpox in the absence of systemic or skin findings is exceedingly rare.
In conclusion, we describe a case of isolated ocular mpox with no skin lesions or systemic prodromal symptoms in a relatively immunocompromised patient. Because pertinent history and clinical suspicion were lacking, metagenomic RNA sequencing was highly valuable in helping identify the pathogen. Semen was the likely vehicle for direct transmission (8), although hand-to-eye contact with unobserved lesions or exposure through nondisclosed encounters cannot be excluded. Intraocular mpox involving the sclera, cornea, and anterior chamber, along with persistently PCR-positive ocular surface mpox, has a poor visual prognosis. There is no established treatment for ocular mpox (9). Continued mpox disease progression over several months is unusual and could raise suspicion of immunosuppression or treatment resistance.
Dr. Nguyen is a fellow in cornea and external disease at the University of California San Francisco and F.I. Proctor Foundation in San Francisco, California. His research interests include regenerative ophthalmology and ocular infectious diseases.
Acknowledgment
Research reported in this manuscript was supported by the National Eye Institute of the National Institutes of Health under award no. R01EY03861 (TD), a Research to Prevent Blindness unrestricted grant, and the Pacific Huang Foundation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
References
- Doan T, Hinterwirth A, Worden L, Arzika AM, Maliki R, Abdou A, et al. Gut microbiome alteration in MORDOR I: a community-randomized trial of mass azithromycin distribution. Nat Med. 2019;25:1370–6. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Interim clinical considerations for management of ocular mpox virus infection. 2022 [cited 2023 Feb 20]. https://www.cdc.gov/poxvirus/monkeypox/clinicians/ocular-infection.html
- Frenois-Veyrat G, Gallardo F, Gorgé O, Marcheteau E, Ferraris O, Baidaliuk A, et al. Tecovirimat is effective against human monkeypox virus in vitro at nanomolar concentrations. Nat Microbiol. 2022;7:1951–5. DOIPubMedGoogle Scholar
- Hughes C, McCollum A, Pukuta E, Karhemere S, Nguete B, Lushima RS, et al. Ocular complications associated with acute monkeypox virus infection, DRC. Int J Infect Dis. 2014;21:276–7. DOIGoogle Scholar
- Iñigo Martínez J, Gil Montalbán E, Jiménez Bueno S, Martín Martínez F, Nieto Juliá A, Sánchez Díaz J, et al. Monkeypox outbreak predominantly affecting men who have sex with men, Madrid, Spain, 26 April to 16 June 2022. Euro Surveill. 2022;27:
2200471 . DOIPubMedGoogle Scholar - Vusirikala A, Charles H, Balasegaram S, Macdonald N, Kumar D, Barker-Burnside C, et al. Epidemiology of early monkeypox virus transmission in sexual networks of gay and bisexual men, England, 2022. Emerg Infect Dis. 2022;28:2082–6. DOIPubMedGoogle Scholar
- Thornhill JP, Barkati S, Walmsley S, Rockstroh J, Antinori A, Harrison LB, et al.; SHARE-net Clinical Group. Monkeypox virus infection in humans across 16 countries—April–June 2022. N Engl J Med. 2022;387:679–91. DOIPubMedGoogle Scholar
- Lapa D, Carletti F, Mazzotta V, Matusali G, Pinnetti C, Meschi S, et al.; INMI Monkeypox Study Group. Monkeypox virus isolation from a semen sample collected in the early phase of infection in a patient with prolonged seminal viral shedding. Lancet Infect Dis. 2022;22:1267–9. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Ocular monkeypox—United States, July–September 2022. 2022 [cited 2022 Dec 15]. https://www.cdc.gov/mmwr/volumes/71/wr/mm7142e1.htm
Figure
Table
Cite This ArticleOriginal Publication Date: May 02, 2023
Table of Contents – Volume 29, Number 6—June 2023
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Gerami D. Seitzman, University of California San Francisco—Ophthalmology, 490 Illinois St, San Francisco, CA 94158, USA
Top