Volume 3, Number 4—December 1997
THEME ISSUE
Foodborne
Controlling Emerging Foodborne Microbial Hazards
Quantitative Microbiology: A Basis for Food Safety
Factors Affecting Microbial Behavior in Foods
Predictive Microbiology
Problems with Predictive Microbiology and Research Needs
Current Status of Predictive Microbiology
Defining the Growth/No Growth Interface
Microbial Responses to Stress and Microbial Physiology
Application of Predictive Models
Conclusions
Cite This Article
Cite This Article
Citation for Media
Abstract
Because microorganisms are easily dispersed, display physiologic diversity, and tolerate extreme conditions, they are ubiquitous and may contaminate and grow in many food products. The behavior of microbial populations in foods (growth, survival, or death) is determined by the properties of the food (e.g., water activity and pH) and the storage conditions (e.g., temperature, relative humidity, and atmosphere). The effect of these properties can be predicted by mathematical models derived from quantitative studies on microbial populations. Temperature abuse is a major factor contributing to foodborne disease; monitoring temperature history during food processing, distribution, and storage is a simple, effective means to reduce the incidence of food poisoning. Interpretation of temperature profiles by computer programs based on predictive models allows informed decisions on the shelf life and safety of foods. In- or on-package temperature indicators require further development to accurately predict microbial behavior. We suggest a basis for a "universal" temperature indicator. This article emphasizes the need to combine kinetic and probability approaches to modeling and suggests a method to define the bacterial growth/no growth interface. Advances in controlling foodborne pathogens depend on understanding the pathogens' physiologic responses to growth constraints, including constraints conferring increased survival capacity.
Ensuring the microbial safety and shelf life of foods depends on minimizing the initial level of microbial contamination, preventing or limiting the rate of microbial growth, or destroying microbial populations. With many foods, these strategies have been practiced successfully for thousands of years. However, in the last decade, the incidence of foodborne disease has increased in the industrialized world (1), despite the introduction of the Hazard Analysis and Critical Control Points (HACCP) concept and the promulgation of regulations in food safety. The increased incidence of foodborne disease is caused by changes in agricultural and food processing practices, increasing international trade in foods, and social changes (which include changed eating habits and increased population mobility) (2).
This article develops the propositions that available quantitative information, properly applied, is a basis for improved food safety; the information available, largely an empiric description of microbial behavior in foods, highlights a lack of understanding of the physiology of foodborne pathogens; and knowledge of the physiology may lead to more precise control of foodborne bacteria and novel protocols to ensure the microbiologic safety of foods.
Bacteria have inhabited the earth for approximately three and a half billion years and have colonized almost every conceivable habitat (3). In fact, the development of microbial populations is probably precluded only when liquid water is absent or conditions are so extreme that rapid denaturation of proteins outpaces their rate of replacement. The major characteristics that underpin the success of prokaryotes are small size and ease of dispersal, physiologic diversity, and tolerance of extreme conditions (4). The temperature range over which microbial populations develop is -12°C (the temperature at which intracellular water freezes) to +112°C (the temperature at which liquid water is maintained only under elevated pressure). The pH range is pH 1 to pH 12, and the salinity range is zero to saturated (4).
Langeveld et al. (5), who studied microbial development in biofilms in a tubular heat exchanger used to pasteurize milk, report the exploitation of different ecologic niches by bacteria. Through the ascending temperature range of the tube (~20°C to ~90°C), the dominant microbiota changed from gram-negative bacteria such as Acinetobacter, to coliforms to Streptococcus thermophilus to thermophilic bacilli. At the highest temperature, the wall of the exchanger was colonized by Thermus thermophilus. Thus, it appears that contaminants deposited along the length of the tube were selected by the in situ temperature, with the fastest-growing organism dominating.
Most studies in food microbiology are concerned with the rapid growth of populations, but in many ecosystems, the survival characteristics of the population also need to be considered. The longevity of bacterial spores and their resistance to harsh conditions are well documented. However, the ability of vegetative cells to resist stressful conditions is increasingly recognized as an important ecologic trait (6). Attention also needs to be given to relatively slow-growing populations in various situations, e.g., when the shelf life of a product is extended by control of rapidly growing spoilage organisms. The behavior of foodborne microorganisms, be it the growth or death of microbial populations, is based on the time of exposure to environmental factors affecting population development; for example, equivalent kills of bacteria in milk are achieved by low temperature—long time pasteurization (60°C/30 min) and high temperature—short time pasteurization (72°C/15 sec). When populations are in the biokinetic range, the rate at which they develop is determined by factors such as temperature, water availability, and pH applied in food preservation procedures. The extent of microbial growth is a function of the time the population is exposed to combinations of intrinsic food properties (e.g., salt concentration and acidity) and extrinsic storage conditions (e.g., temperature, relative humidity, and gaseous atmosphere).
Different factors assume dominance in different foods and preservation strategies. In many foods, the full preservation potential of a single property is restricted because of considerations related to the esthetic, organoleptic, and nutritional properties of the product. However, several properties or conditions may be combined to provide a desired level of stability (7). In situations where the preservation strategy is designed to slow the rate of population growth, the effect will always be increased by storage temperature. Temperature control in processing, distribution, and storage (the cold chain) is crucial to ensure the adequate shelf life and safety of many common foods, including meat, fish, poultry, and milk. Newer technologies, including modified atmosphere packaging and sophisticated products such as sous-vide meals, do not obviate the need for strict temperature control. Indeed, the requirement for vigilance increases with increased shelf life and the possibility of growth of psychrotrophic pathogens over an extended period.
Predictive microbiology involves knowledge of microbial growth responses to environmental factors summarized as equations or mathematical models. The raw data and models may be stored in a database from which the information can be retrieved and used to interpret the effect of processing and distribution practices on microbial proliferation. Coupled with information on environmental history during processing and storage, predictive microbiology provides precision in making decisions on the microbiologic safety and quality of foods. The term "quantitative microbial ecology" has been suggested as an alternative to "predictive microbiology" (8).
The development, validation, and application of predictive microbiology has been extensively reviewed in the last decade (9,10). Modeling studies have concentrated on descriptions of the effect of constraints on microbial growth (rather than survival or death), often using a kinetic model approach (rather than probability modeling) and most often describing the effect of temperature as the sole or one of a number of controlling factors. For example, the temperature dependence model for growth of Clostridium botulinum demonstrated a good fit to data, but the authors noted that "care must be taken at extremes of growth, as no growth may be registered in a situation where growth is indeed possible but has a low probability" (11).
The emphasis in modeling efforts on temperature (often in combination with other factors) may be justified, given its crucial role in the safe distribution and storage of foods. Surveys carried out over several decades in the United Kingdom, United States, Canada, and Australia point to the predominant role of temperature abuse in outbreaks of foodborne disease (12-14).
Several commonly perceived problems with predictive modeling (8) are reviewed below. While considerable progress has been made in defining philosophic approaches and experimental protocols for growth model development and many models have been developed and published, more validation studies are required, particularly involving independent and industry-based trials. More emphasis should be placed on modeling the death kinetics of foodborne pathogens with low infective doses.
Measurement of environmental factors (e.g., temperature) can be achieved with precision, but in some situations, (e.g., in chilling of meat carcasses), it is more difficult. Location of the sensor can be an important consideration (15,16). In meat chilling, where control of microbial development is a function of the combined effects of falling temperature and water activity, development of a technique to measure water activity in situ at the carcass surface would provide valuable information. Furthermore, development of techniques to measure constraints such as water activity, pH, or redox potential on a microscale might provide useful information for a complex food such as salami. This would allow definition of the role of the microenvironment in determining microbial behavior. The concept of a microenvironment is well developed in soil microbiology (17) but has been neglected in food microbiology.
The inherent variability of response times (generation time and lag phase duration) as an issue in predictive microbiology was first raised by Ratkowsky et al. (18), who related the variance of responses on a transformed rate scale such as V(Ök) or V(lnk) to the variances of responses on a time (q) scale. The variance was shown to be proportional to the square or cube of the response time. It was later confirmed (19-21) that nonnormal gamma and inverse Gaussian distributions described the distribution of response times in predictive microbiology, which indicate that variance is proportional to the square or cube of the response time, respectively.
The practical implication of these findings for the application of kinetic models is that inherent biologic variability increases markedly with increasing response times, and thus the confidence limits associated with predictions also increase markedly. However, if the probability distribution of the response time is known, one can determine the probability that an organism will grow more quickly than a predicted response time (21). Thus, kinetic models are appropriate to describe consistent microbial growth responses, but under extreme conditions a probability approach may be required.
Models are normally developed under static conditions (growth rates and lag times are measured at a series of set temperatures, water activity values, and pH levels), and the results are combined to describe the effects of each factor or a combination of factors on population development. Subsequently, models must be validated in foods under conditions that mimic situations encountered in normal practice, e.g., decreasing temperature and water activity during active chilling of meat carcasses or fluctuating temperatures during the distribution and storage of many food commodities.
Shaw (22) and later several other authors (23-26) reported on the effect of fluctuating temperatures. Depending on the magnitude of the temperature deviation, the organism may change its rate of growth to a rate characteristic of the new temperature, or it may stop growing if a lag phase is introduced. In both situations, Salmonella Typhimurium has responded nearly as predicted by the model (24,25). Baranyi et al. (26) presented similar results for the spoilage bacterium Brocothrix thermosphacta. When cycled between 25°C and 5°C, the model predicted behavior well in both the growth rate and lag phase duration. However, a temperature shift from 25°C to 3°C caused deviations from model predictions due to decline in viable cell numbers or extended lag phases. During the final extended phase of growth at 2.8°C, the rate resumed at that predicted. Baranyi et al. (26) also examined the perceived problem of modeling lag phase duration. The difficulty in predicting lag phase duration in foods is not due to the lack of a suitable model: the difficulty comes from the lack of knowledge of the physiologic status of the organisms contaminating the food. The organisms may include cells that are actively growing, exhibiting a physiologic lag phase, damaged and under repair, exhibiting physiologic (endospores) or exogenous dormancy (VNC cells), damaged but unable to reproduce because of ineffective repair mechanisms, and dead. At least part of the confusion surrounding the measurement of lag phase duration arises from experiments in which inocula of different physiologic statuses were used (27,28).
Methods to define the physiologic status of foodborne contaminants under various conditions need to be developed. This will require observations on individual cells or small populations of cells either directly by microscopy or an indicator of single-cell metabolic activity (26). Luminescent Salmonella strains have been used as real-time reporters of growth and recovery from sublethal injury (29). Alternatively, a parameter to describe the suitability of cells to grow in a new environment may be incorporated in the model (26).
Some problems with predictive microbiology have been discussed, and, for each, needed research has been suggested. Opinions vary on the efficacy of models to predict outcomes under real life conditions. At one end of the scale, accuracy such as that described for the growth of Pseudomonas in minced beef (Figure 1) can be obtained in trials conducted independently of the laboratory in which the model was developed. At the other end, models developed in laboratory broth systems have been reported to be inappropriate to describe growth on food (30). Dalgaard (31) reported similar discrepancies and suggested an iterative approach to model development using food, rather than laboratory media, as the growth substrate for model development. Such reports emphasize the need for rigorous validation of models under practical conditions. Deviations from predictions do not necessarily imply that the model is defective but more likely that knowledge of some food ecosystems is incomplete and factors other than those used in model development have an effect on microbial behavior.
The common theme of the problems in predictive microbiology discussed above is that of uncertainty—uncertainty in terms of the starting conditions (e.g., initial microbial numbers and types) and the microbial response in a given or changing environment. Uncertainty translates to variability if the distribution of response times is understood and the variance can be described. As we have indicated above, the variability associated with very long response times limits the utility of kinetic models and requires a probability approach. Thus, while in the last decade predictive modelers were justified in their selection of temperature as a primary factor to model in kinetic approaches, the next decade may see a return to probability modeling as pioneered by Genigeorgis (32) and Roberts (33). This shift will derive impetus from the emergence of dangerous pathogens with very low infective doses, and continued kinetic modeling will concentrate on survival and death rather than growth of populations.
The first kinetic death model to find widespread use in the food industry was for thermal destruction (34). One can consider a model describing a 12-log cycle reduction in C. botulinum spores in a short time with considerable certainty. However, as we move toward less severe processes with longer response times and the added complications of "shoulders" and "tails" to define the growth/no growth interface, biologic variability will again dictate a probability approach to describe the survival and slow decline of microbial populations.
Because growth of pathogenic bacteria in foods always increases the risk for foodborne disease, defining the conditions at which no growth is possible is of considerable practical significance for food manufacturers and regulators. Bacterial growth/no growth interface models quantify the combined effect of various hurdles on the probability of growth and define combinations at which the growth rate is zero. Increasing the level of one or more hurdles at the interface by only a small amount will significantly increase the probability of "fail safe" events and decrease the probability that a few cells in the population will resolve the lag phase and begin to grow (a "fail dangerous" event) (7). The growth/no growth interface also has great physiologic significance because at that point biosynthetic processes are insufficient to support population growth, and survival mechanisms are in place.
A procedure to derive the interface was proposed by Ratkowsky and Ross (35); it employs a logistic regression model to define the probability of growth as a function of one or more controling environmental factors. From this model, the boundary between growth and no growth, at some chosen level of probability, can be determined. The form of the expression containing the growth limiting factors is suggested by a kinetic model, while the response at a given combination of factors is either presence or absence (i.e., growth/no growth) or probabilistic (i.e., the fraction of positive responses in n trials). This approach represents an integration of probability and kinetic approaches to predictive modeling.
Bacteria have physiologic mechanisms enabling them to survive in environments that preclude their growth. While some tolerance to environmental insults is adaptive, a wide range of protective mechanisms is induced when cells enter a stationary phase or become starved. These phenomena are under the control of the rpoS gene, which codes for a stationary-phase—specific sigma factor, expression of which triggers the development of a semidormant state in which bacteria can better resist multiple physical challenges (36). This factor and the gene products whose expression it controls are of vital significance to food microbiology; they form the basis for a global stress response in which one stress can confer protection to a wide range of other stresses. Under the influence of this factor, bacterial cells respond very quickly to unfavorable changes in their environment. Often these responses are phenotypic and remain in place only during stress (37).
Low pH Tolerance
Brown et al. (37) demonstrated "acid habituation" (38), a phenotypic response to an environmental insult, for five strains of Escherichia coli. These strains showed a wide range of intrinsic acid tolerance, which for each strain was enhanced by exposure to nonlethal acidity (pH 5) before exposure to a lethal acid challenge (pH 3). Neutralization of the growth medium partially reversed tolerance to acid stress, underlining that acid habituation is a phenotypic response. Furthermore, acid tolerance was correlated with changes in the fatty acid composition of the cell membrane. During acid habituation, monounsaturated fatty acids (16:1w7c and 18:1w7c) in the phospholipids of E. coli were either converted to their cyclopropane derivatives (cy17:0 and cy19:0) or replaced by saturated fatty acids. The degree of acid tolerance of the five strains of E. coli was highly correlated with the membrane cyclopropane fatty acid content, which may enhance the survival of cells exposed to low pH.
Low Water Activity Tolerance
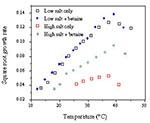
Bacterial cells, when confronted by lowered water activity, regulate the internal environment by rapidly accumulating compatible solutes such as glycine betaine or carnitine (39). The solutes, which may be scavenged by the cell, exert their influence at very low concentrations; the effect is demonstrated both in limits and rate growth. These compounds appear also to provide cryotolerance as well as osmotolerance (40; Figure 2).
Energy Diversion
Microbial responses to stressful conditions may constitute a drain on the energy resources of the cell, e.g., in relation to the accumulation of compatible solutes (41). Knochel and Gould (42) argued "that restriction of the availability of energy will interfere with a cell's reaction to osmotic stress." The energy diversion hypothesis was supported by McMeekin et al. (9) on the basis of observations on the growth of Staphylococcus xylosus at nine different levels of water activity. Though Tmin, the theoretic minimum temperature for growth, remained constant, the actual minimum temperature at which growth was observed increased with decreasing water activity, suggesting energy expenditure to cope with aw stress.
Krist and Ross (unpub. data), however, challenged this explanation because of findings from growth rate and yield experiments on E. coli growing in a glucose-limited minimal minerals medium. With both decreasing temperature and water activity, the growth rate declined gradually, but the yield was not greatly affected until close to the point where growth ceased. As the substrate was converted to the same amount of biomass, this suggests that the stresses imposed by suboptimal temperature or water activity are not a major drain on the cell's energy reserves. Compatible solutes likely ameliorate the effect of both factors by maintaining enzymes in an active configuration (39). With pH, the growth rate of many organisms is unaffected across a wide range of pH values. To maintain intracellular pH, the cell uses considerable energy to export protons (43), and thus it is anticipated that yield will be affected.
Increasing knowledge of the physiologic response of bacterial cells to individual constraints and combinations of constraints will provide greater precision in defining growth-limiting conditions and possibly allow development of novel protocols to ensure the microbial safety of foods. As an example, the remarkable effect of compatible solutes on the growth rate and growth range conditions is an obvious advantage for bacteria growing under stressful conditions (Figure 2). Compatible solutes, such as betaine and carnitine, are widely distributed in foods of plant and animal origin and are easily available to bacteria and rapidly taken up by specific transport mechanisms (39,40). It is unlikely that growth might be controlled by "creating a hostile environment devoid of osmolytes," as suggested by Smith (44). However, it might be possible to use the specific uptake mechanism to deliver a compatible solute analogue with lethal effects on the cell. Alternatively, if the cell is moved from an environment in which growth is possible to one where growth ceases, compatible solutes may also allow enzymic reactions to continue within the cell, depleting energy reserves and inducing a greatly extended lag phase or death. This hypothesis is supported by the observations of many authors that survival is better at low rather than ambient temperatures. For example, Clavero and Beuchat (45) state, "Regardless of the pH and aw, survival of E. coli O157:H7 was better at 5°C than at 20°C or 30°C." Furthermore, preliminary experiments in this laboratory suggest that E. coli declines more rapidly in the presence of betaine than in its absence (Krist, unpub. data).
The incorporation of predictive models into devices such as temperature loggers has been described for E. coli (46) and Pseudomonas (47), as has the development of expert systems from predictive modeling databases (48,49).
The current food poisoning crisis indicates that existing quantitative information on microbial growth, survival, and death, if properly applied, would have an immediate impact on the incidence of foodborne disease in the industrialized world. Even without the synthesis of data into mathematical models, simply logging the temperature history of food processing, distribution, and storage operations would provide much useful information. Loggers provide a hard copy of a temperature profile in real time and thus evidence of temperature abuse and the source of the abuse.
For loggers with appropriate software (46,47), the temperature profile may be interpreted in terms of microbial growth. However, the interpretation must be based on an informed analysis of the temperature history by a trained operator. The operator may, for example, be required to enter default values for initial bacterial numbers or provide an estimate of lag phase duration under specified conditions. Estimates of both imply general knowledge of food microbiology and specific knowledge of the process and products under consideration. The equivalence of an estimate of microbial growth derived from temperature profile to that obtained from conventional microbiologic criteria may also be necessary (15).
An alternative to the use of temperature loggers is the development of in- or on-package temperature tags as recommended in the U.S. Food Safety Initiative draft document Food Safety from Farm to Table (50). With temperature tags, informed interpretation is not required because abuse is indicated directly by the tag response. Therefore, the tag must indicate the significance of the environmental history for microbial behavior. The time/temperature tags available are based on physical or chemical changes that follow Arrhenius kinetics (9). While these may give a reasonable approximation of microbial growth in the normal range, the deviation of microbial responses becomes increasingly large as conditions move from normal to stressful. The intriguing possibility of a universal time/temperature indicator was flagged (51) on the basis of observations made of temperature effects on foodborne pathogens in this laboratory and by Snyder (52). The universal indicator is based on a relationship that describes the maximum specific growth rate of a continuum of organisms from psychrophiles to thermophiles in terms of Arrhenius kinetics with an apparent activation energy of ~80 kJ/mole. This value can be related to the activation energy/growth rate at any other temperature by a relative rate function derived from Belehradek (square root) kinetics.
We have argued that a thorough understanding of microbial ecology and physiology offers the best opportunity to control microbial populations in food and reverse the upward trend in the incidence of foodborne disease. Many food preservation strategies have their origin in empirical observations practiced for thousands of years. The systematic collection and collation of data on microbial behavior in foods spawned the discipline of food microbiology, within which predictive microbiology (quantitative microbial ecology) has accelerated our understanding of the microbial ecology of foodborne bacteria. Studies in microbial physiology will further enhance our knowledge and offer new possibilities for food preservation.
The most disturbing aspect of the current crisis is that simple application of existing knowledge would lead to a marked reduction in the incidence of foodborne disease. Education of food handlers and consumers in basic hygiene and the consequences of temperature abuse is urgently needed as is a greater depth of understanding for those in technical positions in the food industry or those with regulatory responsibilities. Furthermore, an appreciation of the need for shared responsibility for food safety within all sectors of the continuum from farm to table, including the consumer, has to be developed. The U.S. Food Safety Initiative draft document emphasizes this point, as does the structure of the Australian Meat Research Corporation's Microbial Food Safety Key Program (53).
Acknowledgment
The work described in this manuscript was funded largely by the Australian Meat Research Corporation (MRC) through a Core Funding Project and the Microbial Food Safety Key Program. The authors are indebted to Dr. Stefan Fabiansson for his support and encouragement of research into the ecology and physiology of foodborne pathogens.
References
- Maurice J. The rise and rise of food poisoning. New Sci. 1994;144:28–33.
- McMeekin TA, Olley J. Predictive microbiology and the rise and fall of food poisoning. ATS Focus. 1995;88:14–20.
- Pace NR. Perspective on the natural microbial world: molecular microbial ecology. ASM News. 1996;63:463–70.
- Schelgel HG, Jannasch HW. Prokaryotes and their habitats. In: Balows A, Truper HG, Dworken M, Harder W, Scheifer K-H, editors. The prokaryotes. New York: Springer Verlag; 1992. p. 75-125.
- Langeveld PLM, van der Klijn W, van der Waals CB. Bacterie groei in indampers voor milk en wei. Overdruk van Voedingsmiddelentechnologie. 1990;23:13–7.
- Archer DL. Preservation microbiology and safety: evidence that stress enhances virulence and triggers adaptive mutations. Trends Food Sci Technol. 1996;7:91–5. DOIGoogle Scholar
- Ross T, McMeekin TA. Predictive microbiology and HACCP. In: Pearson AM, Dutson TR, editors. HACCP in meat, poultry and fish processing. London: Blackie Academic and Professional; 1995. p. 330-53.
- McMeekin TA, Olley J, Ross T, Ratkowsky DA. Predictive microbiology: theory and application. Taunton, UK: Research Studies Press; 1993.
- Whiting RC, Buchanan RLB. Predictive modeling. In: Doyle MP, Beuchat LR, Montville TJ, editors. Food microbiology fundamentals and frontiers. Washington (DC): American Society for Microbiology Press 1997. p. 728-39.
- Graham A, Lund BM. The effect of temperature on the growth of non proteolytic type B Clostridium botulinum. Lett Appl Microbiol. 1993;16:158–60. DOIGoogle Scholar
- Davey GR. Food poisoning in New South Wales: 1977-84. Food Technology in Australia. 1985;37:453–7.
- Enneking U. Hazard analysis of critical control points (HACCP) as part of the Lufthansa in-flight service quality assurance. International Food Safety News. 1993;2:52–3.
- Adams M, Moss MO. Food microbiology. Cambridge: Royal Society of Chemistry; 1995.
- Gill CO, Harrison JCL, Phillips DM. Use of a temperature function integration technique to assess the hygienic efficiency of a beef carcass cooling process. Food Microbiol. 1991;8:83–94. DOIGoogle Scholar
- Gill CO, Jones SDM, Tong AKW. Application of a temperature function integration technique to assess the hygienic efficiency for spray chilling beef carcasses. J Food Prot. 1991;54:731–6.
- Madigan MT, Martinko JM, Parker J. Microbial ecology. In: Brock, editor. Biology of microorganisms. 8th ed. New Jersey: Prentice-Hall; 1996.
- Ratkowsky D, Ross T, McMeekin TA, Olley J. Comparison of Arrhenius-type and Belehradek-type models for the prediction of bacterial growth in foods. J Appl Bacteriol. 1996;71:452–9.
- Ratkowsky DA. Predicting response times in predictive microbiology. Occasional paper. Tasmania, Australia: Research and Development Unit, Biometrics Section, Dept of Primary Industry, Fisheries and Energy; 1991.
- Alber SA, Schaffner DW. Evaluation of data transformations used with the square root and Schoolfield models for predicting bacterial growth rate. Appl Environ Microbiol. 1992;58:3337–42.PubMedGoogle Scholar
- Ratkowsky DA, Ross T, Macario N, Dommett TW, Kamperman L. Choosing probability distributions for modelling generation time variability. J Appl Bacteriol. 1996;80:131–7.
- Shaw MK. Effect of abrupt temperature shift on the growth of mesophilic and psychrophilic yeasts. J Bacteriol. 1967;93:1332–6.PubMedGoogle Scholar
- Labuza TP, Fu B. Growth kinetics for shelf-life prediction: theory and practice. J Ind Microbiol. 1993;12:309–23. DOIGoogle Scholar
- Mitchell GA, Brocklehurst TF, Parker R, Smith AC. The effect of transient temperatures on the growth of Salmonella typhimurium LT2. I: Cycling within the growth region. J Appl Bacteriol. 1994;77:113–9.PubMedGoogle Scholar
- Mitchell GA, Brocklehurst TF, Parker R, Smith AC. The effect of transient temperature on the growth of Salmonella typhimurium LT2. II: Excursions outside the growth region. J Appl Bacteriol. 1995;79:128–34.PubMedGoogle Scholar
- Baranyi J, Robinson TP, Kaloti A, Mackey BM. Predicting growth of Brocothrix thermosphacta at changing temperature. Int J Food Microbiol. 1995;27:61–75. DOIPubMedGoogle Scholar
- Walker SJ, Archer P, Banks JG. Growth of Listeria monocytogenes at refrigeration temperatures. J Appl Bacteriol. 1990;68:157–62.PubMedGoogle Scholar
- Dufrenne J, Delfgou E, Ritmeester W, Notermans S. The effect of previous growth conditions on the lag phase of some foodborne pathogenic microorganisms. Int J Food Microbiol. 1997;34:89–94. DOIPubMedGoogle Scholar
- Chen J, Griffiths MW. Luminescent Salmonella strains as real time reporters of growth and recovery from sublethal injury in food. Int J Food Microbiol. 1996;31:27–44. DOIPubMedGoogle Scholar
- Gill CO, Greer GG, Dilts BD. The aerobic growth of Aeromonas hydrophila and Listeria monocytogenes in broths and on pork. Int J Food Microbiol. 1997;35:67–74. DOIPubMedGoogle Scholar
- Dalgaard P. Predictive microbiological modelling and seafood quality. In: Seafood from producer to consumer, integrated approach to quality. Proceedings of the International Seafood Conference, 1995 Noordwijkerhout. The Netherlands: Elsevier, Amsterdam. In press 1997.
- Genigeorgis CA. Factors affecting the probability of growth of pathogenic microorganisms in foods. J Am Vet Med Assoc. 1981;179:1410–7.PubMedGoogle Scholar
- Roberts TA, Gibson AM, Robinson A. Prediction of toxin production by Clostridium botulinum in pasteurised pork slurry. J Food Technol. 1981;16:337–55.
- Stumbo CR, Prokit KS, Ramarkrishnan TV, Evans DA, Franaas FJ. CRC handbook of lethality guides for low-acid canned foods. 1. Boca Raton (FL): CRC Press; 1983.
- Ratkowsky DA, Ross T. Modelling the bacterial growth/no growth interface. Lett Appl Microbiol. 1995;20:29–33. DOIGoogle Scholar
- Lee IS, Lin J, Hall HK, Bearson B, Foster JW. The stationary-phase sigma factor (RpoS) is required for a sustained acid tolerance response in virulent Salmonella typhimurium. Mol Microbiol. 1995;17:155–67. DOIPubMedGoogle Scholar
- Brown J, Ross T, Nichols PD, McMeekin TA. Acid habituation of Escherichia coli and the potential role of cyclopropane and fatty acids in low pH tolerance. Int J Food Microbiol. 1997. In press.
- Goodson M, Rowbury RJ. Resistance of acid-habituated Escherichia coli to organic acid and its medical and applied significance. Lett Appl Microbiol. 1989;8:211–4. DOIGoogle Scholar
- Galinski EA. Compatible solutes of halophilic eubacteria: molecular principles, water solute interaction, stress protection. Experientia. 1993;49:487–96. DOIGoogle Scholar
- Ko R, Smith LT, Smith GC. Glycine betaine confers enhanced osmotolerance and cryotolerance on Listeria monocytogenes. J Bacteriol. 1994;176:426–31.PubMedGoogle Scholar
- Csonka LN. Physiological and genetic responses of bacteria to osmotic stress. Microbiol Rev. 1989;53:121–47.PubMedGoogle Scholar
- Knochel S, Gould G. Preservation microbiology and safety: quo vadis? Trends Food Sci Technol. 1995;6:127–31. DOIGoogle Scholar
- Davidson PM. Chemical preservatives and natural antimicrobial compounds. In: Doyle MP, Beuchat LR, Montville TJ, editors. Food microbiology fundamentals and frontiers. Washington (DC): American Society for Microbiology Press, 1997.
- Smith LT. Role of osmolytes in adaptation of osmotically stressed and chill stressed Listeria monocytogenes grown in liquid media and on processed meat surfaces. Appl Environ Microbiol. 1996;62:3088–93.PubMedGoogle Scholar
- Clavero MRS, Beuchat LR. Survival of Escherichia coli O157:H7 in broth and processed salami as influenced by pH, water activity and temperature and suitability of media for its recovery. Appl Environ Microbiol. 1996;61:2735–40.
- Gill CO, Phillips DM. Hygienically appropriate time/temperature parameters for raw meat processing. Proceedings of the 36th International Congress of Meat Science and Technology; 27 Aug_2 Sep 1990; Havana, Cuba; 1990. p. 458-70.
- McMeekin TA, Ross T. Modeling applications. J Food Prot. 1996;Suppl:1–88.
- Adair C, Briggs PA. The concept and application of expert systems in the field of microbiological safety. J Ind Microbiol. 1993;12:263–7. DOIGoogle Scholar
- Jones JE. A real-time database/models base/expert system in predictive microbiology. J Ind Microbiol. 1993;12:268–72. DOIGoogle Scholar
- Glickman D, Salala DE, Browner CM. Food safety from farm to table: a new strategy for the 21st Century. Washington (DC): U.S. Department of Agriculture;1997.
- McMeekin TA, Ross T. Shelf life prediction: status and future possibilities. Int J Food Microbiol. 1996;33:65–83. DOIPubMedGoogle Scholar
- Snyder OP. Use of time and temperature specifications for holding and storing food in retail operations. Dairy Food Environ Sanitat. 1996;116:374–88.
- Fabiansson S, Wrigley J, Sumner J, McMeekin TA, Orr C. Meat Research Corporation Business Plan, Microbial Food Safety Key Program, 1996. Sydney, Australia: Meat Research Corporation; 1996.
Figures
Cite This ArticleTable of Contents – Volume 3, Number 4—December 1997
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Professor T.A. McMeekin, Department of Agricultural Science, University of Tasmania, GPO Box 252-54, Hobart, TAS 7001, Australia; fax: 61-3-62-26-2642
Top