Volume 31, Number 9—September 2025
Research Letter
Subarachnoid Neurocysticercosis Caused by Larval-Stage Taenia crassiceps Tapeworm, Slovenia
Cite This Article
Citation for Media
Abstract
We present a case of subarachnoid neurocysticercosis caused by Taenia crassiceps in an elderly woman in Slovenia with no underlying disease or immunosuppressive treatment. The parasite was identified by 12S rDNA PCR and sequencing. Despite prolonged therapy with albendazole and praziquantel, the disease recurred after treatment was discontinued.
Human neurocysticercosis is a severe infection of the central nervous system, generally caused by larvae of the tapeworm Taenia solium and, rarely, by other Taenia species, such as T. crassiceps. So far, 2 cases of T. crassiceps neurocysticercosis have been reported in humans (1,2).
The adult T. crassiceps is an intestinal parasite of carnivores, mainly foxes; small mammals, such as rodents, serve as natural intermediate hosts for cyst-like larvae that proliferate by budding in their body cavities or subcutaneous tissues, leading to massive infections. Humans can become accidental intermediate hosts by ingesting parasite eggs excreted in the definitive hosts’ feces or by contamination of open wounds with eggs, as suspected in subcutaneous infections (3). In addition to neural and subcutaneous infections, infestation of eyes, muscle tissue, and tendons has been reported in humans (1,3). We describe a case of T. crassiceps infection in an elderly patient with meningitis and progressive deterioration of neurologic symptoms diagnosed by a combination of serologic and molecular methods.
Neurologic symptoms developed in a 74-year-old woman from northeastern Slovenia with no underlying diseases in December 2022. Symptoms worsened and led to gait ataxia; tetraparesis, which was markedly left-sided; urinary incontinence; and cognitive decline within a year.
Lumbar puncture (LP) performed in May 2023 confirmed aseptic meningitis. Cerebrospinal fluid (CSF) showed elevated protein levels (0.72 g/L; reference range 0.15–0.45 g/L). Glucose (2.8 mmol/L; reference range 2.5–3.9 mmol/L) and glucose ratio between CSF and serum (0.44; reference >0.31) were unremarkable. Pleocytosis was present with a total leukocyte count of 108 × 106 (reference <5 × 106) cells/L. Analysis of CSF sediment revealed 1% neutrophils, 75% lymphocytes and 3% plasma cells, 9% monocytes, and 12% eosinophils. Intrathecal synthesis of IgG (163.9 mg/L), IgM (5.0 mg/L), and IgA (5.7 mg/L) was confirmed. Results of blood tests, including a differential blood count, were unremarkable.
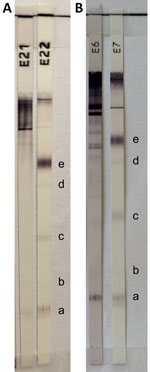
Magnetic resonance imaging (MRI) of the brain revealed an enlarged ventricular system that was more pronounced on the right side without changes in the brain parenchyma. Follow-up LPs confirmed the persistence of pleocytosis in the CNS. Extensive microbiological analyses of CSF and blood samples for infectious agents (Appendix) and tests for autoimmune and paraneoplastic encephalitis were repeatedly negative, with the exception of serologic testing of blood and CSF samples for T. solium IgG, which was equivocal (Figure 1). Because this result was suspicious for neurocysticercosis, we tested CSF using cestode-specific PCR amplifying the mitochondrial 12S rRNA gene (4); the result was positive. After sequencing and BLAST analysis (https://blast.ncbi.nlm.nih.gov/Blast.cgi) of the 242-bp amplicon obtained (Gen Bank accession no. PQ764695), the sequence showed 100% homology with T. crassiceps.
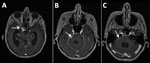
MRI of the brain with contrast performed in March 2024 showed changes consistent with a subarachnoid form of neurocysticercosis (Figure 2). Results of investigations to identify additional foci of cysticercosis, including MRI of the spinal cord, were unremarkable.
We initiated dual therapy for neurocysticercosis with albendazole (800 mg/d) and praziquantel (2,400 mg/d), along with dexamethasone (6 mg/d) to prevent inflammation, in April 2024. Dexamethasone was administered for 4 weeks and praziquantel with albendazole for 85 days. After 85 days of therapy, subsequent CSF PCRs were negative. Praziquantel was discontinued, and treatment with albendazole was continued for a further 80 days. An LP performed 10 days after discontinuation of treatment showed no signs of meningitis. One month later, eosinophilic meningitis was again confirmed with a total leukocyte count of 20 × 106 cells/L in CSF, of which 27% were eosinophils in the sediment, and PCR was again positive. The patient was restarted on dexamethasone, albendazole, and praziquantel for 2 weeks in November 2024, after which she continued treatment with albendazole. At least 1 year of albendazole treatment was planned and, in case of recurrence, lifelong therapy.
The patient’s cognitive status improved substantially after treatment, but spastic tetraparesis did not. She was no longer able to care for herself and moved into a nursing home.
Human T. crassiceps infections are rare; 16 cases have been reported during 1973–2023, mostly in immunocompromised but also in immunocompetent persons (1–3,5–9). Although our patient had no underlying conditions, her age might have led to immunosenescence, increasing her susceptibility to infection.
Laboratory diagnosis of T. crassiceps cysticercosis is challenging, especially when no clinical material is available for parasitological and pathological evaluation, as in our patient who had subarachnoid neurocysticercosis. The cystic appearance of the larvae was not visible on MRI, which differs from 2 other described cases of the parenchymal form of the disease (1,2). Equivocal or weak positive results of serologic tests for other helminthiases might indicate a possible infection (1,8,9). In fact, the initial suspicion of cysticercosis in this patient arose from equivocal blood and CSF T. solium serologic testing.
The source of this patient’s infection is unknown, but she owned a dog, as did several other reported case-patients (1,5,7,8). Increased recreational activity in wildlife areas raises risk for T. crassiceps infection in domestic carnivores, making regular canid deworming essential to prevent infections in humans.
Dr. Šoba is head of the Laboratory of Parasitology at the Institute of Microbiology and Immunology at the Faculty of Medicine, University of Ljubljana, Slovenia. Her primary research interests are diagnostics, epidemiology, and clinical significance of human parasites and their molecular typing.
Acknowledgments
We thank the patient and her family for consenting to publication of this case report.
This study was partly supported by funding from the Slovenian Research and Innovation Agency (grant no. P3-0083).
References
- Ntoukas V, Tappe D, Pfütze D, Simon M, Holzmann T. Cerebellar cysticercosis caused by larval Taenia crassiceps tapeworm in immunocompetent woman, Germany. Emerg Infect Dis. 2013;19:2008–11. DOIPubMedGoogle Scholar
- Floß N, Dolff S, Junker A, Blau T, Rauschenbach L, Sure U, et al. Cerebral Taenia crassiceps larvae infection in a 71-year-old immunocompetent male. Infection. 2023;51:277–81. DOIPubMedGoogle Scholar
- Deplazes P, Eichenberger RM, Grimm F. Wildlife-transmitted Taenia and Versteria cysticercosis and coenurosis in humans and other primates. Int J Parasitol Parasites Wildl. 2019;9:342–58. DOIPubMedGoogle Scholar
- Roelfsema JH, Nozari N, Pinelli E, Kortbeek LM. Novel PCRs for differential diagnosis of cestodes. Exp Parasitol. 2016;161:20–6. DOIPubMedGoogle Scholar
- Goesseringer N, Lindenblatt N, Mihic-Probst D, Grimm F, Giovanoli P. Taenia crassiceps upper limb fasciitis in a patient with untreated acquired immunodeficiency syndrome and chronic hepatitis C infection—the role of surgical debridement. J Plast Reconstr Aesthet Surg. 2011;64:e174–6. DOIPubMedGoogle Scholar
- Heldwein K, Biedermann HG, Hamperl WD, Bretzel G, Löscher T, Laregina D, et al. Subcutaneous Taenia crassiceps infection in a patient with non-Hodgkin’s lymphoma. Am J Trop Med Hyg. 2006;75:108–11. DOIPubMedGoogle Scholar
- Roesel C, Welter S, Stamatis G, Theegarten D, Tappe D. Management of a chest-wall soft-tissue tumor caused by an infection with the larval tapeworm pathogen Taenia crassiceps. Am J Trop Med Hyg. 2014;91:541–3. DOIPubMedGoogle Scholar
- Schmid S, Grimm F, Huber M, Beck B, Custer P, Bode B. Taenia crassiceps infection—an unusual presentation of a tapeworm diagnosed by FNA cytology and PCR. Cytopathology. 2014;25:340–1. DOIPubMedGoogle Scholar
- Tappe D, Berkholz J, Mahlke U, Lobeck H, Nagel T, Haeupler A, et al. Molecular identification of zoonotic tissue-invasive tapeworm larvae other than Taenia solium in suspected human cysticercosis cases. J Clin Microbiol. 2016;54:172–4. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: August 20, 2025
Table of Contents – Volume 31, Number 9—September 2025
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Barbara Šoba, Institute of Microbiology and Immunology, Faculty of Medicine, University of Ljubljana, Zaloška 4, 1000 Ljubljana, Slovenia
Top