Volume 32, Number 3—March 2026
Research Letter
lsaC and Tandem lsaE-lnuB Resistance Genes in Invasive Group A Streptococcus
Figure 1
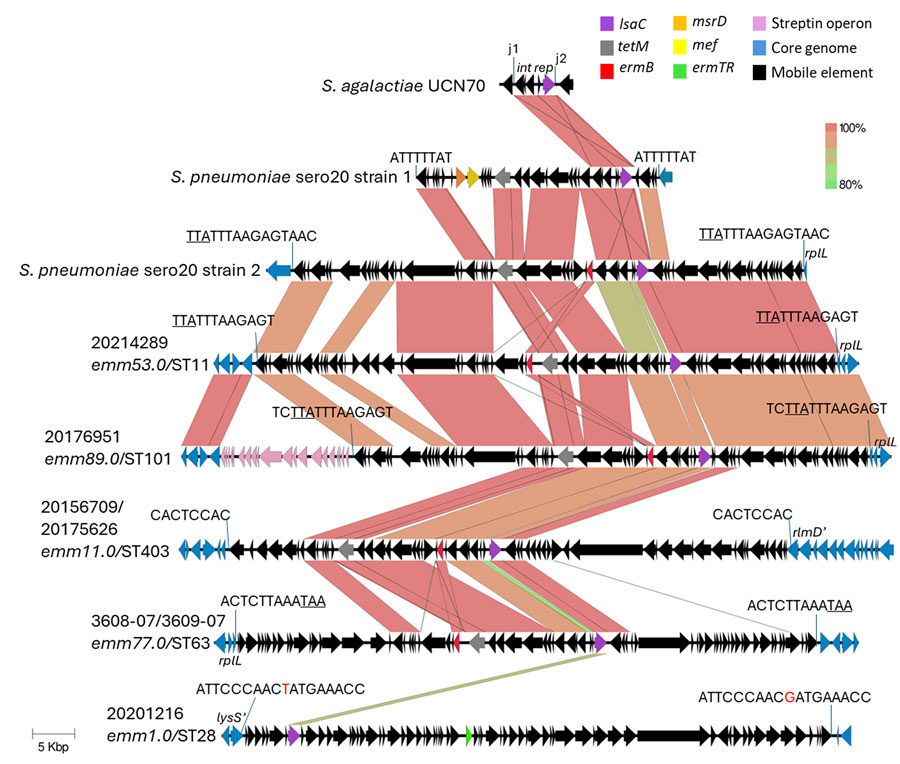
Figure 1. Alignments of 5 different group A Streptococcus lsaC-carrying accessory elements from study of repeated acquisitions of lsaC and tandem lsaE-lnuB resistance genes. Alignments include a partial element from S. agalactiae strain UCN70 (6) and 2 complete elements recently described in pneumococci (9). The j1 and j2 (junctions 1 and 2) sequences depict 24–25 bp sequences that demarcate a 5,258–5,816 bp mobilizable lsaC-carrying cassette that is highly conserved between all of the strains shown except for iGAS strain 20201216 (Appendix Figure 1, panel B). The 8–18 bp target sequence repeat flanking each complete element shown is perfect except in strain 20201216 (nonconserved base in red font). Underlined text indicates the stop codon of the rplL gene in 4 strains (including S. pneumoniae strain 2). The insertion within strain 20156709/20175626 targeted an 8-bp internal sequences within the rlmD gene, resulting in a truncated allele, rmlD’; the insertion within strain 20201216 targeted an 18-bp internal sequence within the lysS gene, resulting another truncated allele, lysS’. Scale bar indicates 5,000 base pairs. ST, sequence type.
References
- Khan ZZ. Group A streptococcal (GAS) infections treatment & management, 2024 [cited 2026 Feb 1]. https://emedicine.medscape.com/article/228936-treatment
- Li Y, Rivers J, Mathis S, Li Z, McGee L, Chochua S, et al. Continued increase of erythromycin nonsusceptibility and clindamycin nonsusceptibility among invasive group A streptococci driven by genomic clusters, United States, 2018–2019. Clin Infect Dis. 2023;76:e1266–9. DOIGoogle Scholar
- Schwarz S, Shen J, Kadlec K, Wang Y, Brenner Michael G, Feßler AT, et al. Lincosamides, streptogramins, phenicols, and pleuromutilins: mode of action and mechanisms of resistance. Cold Spring Harb Perspect Med. 2016;6:
a027037 . DOIGoogle Scholar - File TM Jr, Goldberg L, Das A, Sweeney C, Saviski J, Gelone SP, et al. Efficacy and safety of intravenous-to-oral lefamulin, a pleuromutilin antibiotic, for the treatment of community-acquired bacterial pneumonia: the phase III Lefamulin Evaluation Against Pneumonia (LEAP 1) trial. Clin Infect Dis. 2019;69:1856–67. DOIGoogle Scholar
- Paukner S, Sader HS, Ivezic-Schoenfeld Z, Jones RN. Antimicrobial activity of the pleuromutilin antibiotic BC-3781 against bacterial pathogens isolated in the SENTRY antimicrobial surveillance program in 2010. Antimicrob Agents Chemother. 2013;57:4489–95. DOIGoogle Scholar
- Malbruny B, Werno AM, Murdoch DR, Leclercq R, Cattoir V. Cross-resistance to lincosamides, streptogramins A, and pleuromutilins due to the lsa(C) gene in Streptococcus agalactiae UCN70. Antimicrob Agents Chemother. 2011;55:1470–4. DOIGoogle Scholar
- Douarre PE, Sauvage E, Poyart C, Glaser P. Host specificity in the diversity and transfer of lsa resistance genes in group B Streptococcus. J Antimicrob Chemother. 2015;70:3205–13.
- Berbel D, Càmara J, García E, Tubau F, Guérin F, Giard JC, et al. A novel genomic island harbouring lsa(E) and lnu(B) genes and a defective prophage in a Streptococcus pyogenes isolate resistant to lincosamide, streptogramin A and pleuromutilin antibiotics. Int J Antimicrob Agents. 2019;54:647–51. DOIGoogle Scholar
- Beall B, Lin W, Li Z, Tran T, Metcalf BJ, Anderson BJ, et al. Two independent acquisitions of multidrug resistance gene lsaC in Streptococcus pneumoniae serotype 20 multilocus sequence type 1257. Emerg Infect Dis. 2025;31:2098–108. DOIGoogle Scholar
- Chochua S, Rivers J, Mathis S, Li Z, Velusamy S, McGee L, et al. Emergent invasive group A Streptococcus dysgalactiae subsp. equisimilis, United States, 2015–2018. Emerg Infect Dis. 2019;25:1543–7. DOIGoogle Scholar