Volume 32, Number 4—April 2026
Dispatch
Guillain-Barré Syndrome and Visual Impairment Associated with Emerging Oropouche Virus Lineage, Brazil, 2024
Cite This Article
Citation for Media
Abstract
We report a case of Guillain-Barré syndrome with visual impairment after confirmed Oropouche virus infection during the 2024 outbreak in Ceará, Brazil. Whole-genome sequencing revealed infection by a novel reassortant viral lineage (OROVBR_2025_2024), raising concern about the neurovirulence of this emerging orthobunyavirus strain.
Oropouche virus (OROV), an orthobunyavirus transmitted by mosquitoes and Culicoides paraensis midges, is emerging as a major arboviral pathogen in Latin America. Although typically associated with mild febrile illness, the current outbreak, which is linked to the novel reassortant lineage OROVBR-2015–2024 (1), has been associated with severe cases, including fatalities, vertical transmission with fetal deaths (2–4), and Guillain-Barré syndrome (GBS) (5). We report a case of GBS with possible bilateral optic neuritis after OROV infection during the 2024 outbreak in Ceará state, Brazil.
On August 12, 2024, a previously healthy 48-year-old woman from Capistrano in Ceará state developed acute febrile illness, characterized by high fever (39°C), chills, severe headache, and myalgia. OROV cases had been previously documented in this locality (2,6). On August 14, OROV infection was confirmed through quantitative reverse transcription PCR (qRT-PCR) performed by the Ceará Central Public Health Laboratory. Dengue, Zika, chikungunya, and Mayaro virus infections were excluded through both molecular and serologic testing (Table) (7,8). Fever and systemic symptoms persisted for 2 weeks and were followed by the onset of neurologic manifestations, including paresthesia in her lower limbs, which progressively developed into ascending paresis affecting both upper and lower limbs. In addition, bilateral facial weakness and profound visual loss developed. Initial treatment with corticosteroids (prednisone 40 mg/day for 7 days) was prescribed on September 15 but improvement was minimal, and the patient was referred to a tertiary hospital.
Upon admission on September 17, 2024, the patient was experiencing flaccid tetraparesis, bilateral facial paresis, and areflexia. Blood tests results were unremarkable. Analysis of cerebrospinal fluid revealed albuminocytologic dissociation, elevated protein levels, and unremarkable cell counts (Table), supporting a diagnosis of peripheral demyelinating neuropathy. Oligoclonal bands and IgG index testing of cerebrospinal fluid were unavailable.
Electroneuromyography showed absent sensory conduction in the left superficial fibular nerve, mildly prolonged motor latencies, and reduced motor potentials in fibular and tibial nerves. F-waves were delayed or absent, whereas the H-reflex was bilaterally absent. Electroneuromyography revealed reduced recruitment with high motor unit activity during voluntary movement, compatible with peripheral nervous system demyelination (Appendix Tables 1–5).
Magnetic resonance imaging (MRI) of the brain showed bilateral facial nerve swelling; cervical, thoracic, and lumbar spine MRI revealed edema of lumbar nerve roots (Figure 1). Orbital MRI results were unremarkable.
Bilateral vision loss developed, accompanied by painful eye movements, compromising functional capacity as reported by the patient and family. Although the neuroophthalmologic evaluation occurred only 3 weeks after onset and during steroid treatment, the examination revealed corrected visual acuity of 20/50 in both eyes, impaired pupillary light reflexes, red dyschromatopsia, nasal optic disc edema on fundoscopy, and peripapillary retinal nerve fiber layer edema on optical coherence tomography (Figure 1) without macular involvement. Visual field testing was not performed because of technical issues, and results of MRI performed at this stage were unremarkable. The evidence of subacute vision loss associated with orbital pain worsening with eye movement, reduced contrast and color vision, and pupillary deficit along with the mild optic disc swelling seen in optical coherence tomography favors the diagnosis of a possible optic neuritis (9). The temporal association of those findings with a preceding infectious episode (2–4 weeks) supports the possibility of bilateral postinfectious optic neuritis captured later in the disease evolution.
Empiric antiviral drugs and antibiotics were initiated but discontinued after excluding infectious etiologies such as Listeria and herpes. Plasmapheresis was administered in 5 sessions and was generally well tolerated, although 1 session was followed by transient dysautonomia.
The patient showed gradual but partial neurologic improvement and was discharged on October 1 with moderate gains in motor function and visual acuity. At the 90-day follow-up, the patient was able to walk with assistance, and her visual acuity remained at 20/50 in both eyes. Mild proprioceptive and sensory deficits persisted, particularly in the left foot. Deep tendon reflexes were normal except for slightly diminished Achilles reflexes.
A home visit on February 5, 2025 (6 months after symptom onset), revealed that the patient continued to experience persistent visual impairment, distal hypoesthesia, and paresis in the right lower limb, as well as difficulty standing for >15 minutes. She remained unable to resume agricultural activities or manage household routines independently and required assistance from family members for daily living. Despite partial neurologic improvement, those ongoing limitations led to functional dependence and the need for social security support. During this home visit, additional autoimmune screening tests, including tests for antinuclear antibody and aquaporin 4 and myelin oligodendrocyte glycoprotein antibodies, were performed. Results of all tests were negative (Table).
We performed whole-genome sequencing on patient OROV samples using an amplicon-based protocol (7). We recovered and concatenated near-complete coding sequences of all 3 genome segments (large, medium, and small) for phylogenetic reconstruction using maximum-likelihood inference on IQ-TREE multicore version 2.1.1 (10), employing a nonredundant dataset containing previously published sequences (1,2). Sequence data are available on GISAID (https://www.gisaid.org; accession no. EPI_ISL_20332357).
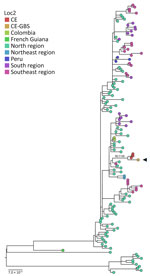
The genome generated belongs to the new OROVBR-2023–2024 lineage (1,11). Phylogenetic analysis (Figure 2) revealed a highly supported monophyletic clade (UltraFast Bootstrap 99.5, SH-aLRT 100) containing sequences from other severe cases, including a fatal vertical transmission case previously described in Ceará (2). This clade is part of a broader lineage linked to cases in Santa Catarina, Paraná, Amazonas, and Pernambuco states in Brazil, as well as to cases in Leticia, Colombia. Those sequences belong to the previously described AM-I sublineage of OROVBR-2015–2024 (1,2,11).
We report a case of GBS with visual impairment possibly caused by optic neuritis associated with the novel OROVBR-2015–2024 lineage, supported by clinical, laboratory, neuroimaging, electrophysiological, and genomic findings. Of note, the patient had been previously healthy and had no underlying conditions.
This case represents the second unusual severe event reported in this small region of Ceará, despite only a few hundred confirmed OROV cases, one of which involved a fetal death attributed to vertical transmission (2,6). This case highlights the neuropathogenic potential of OROV to trigger severe conditions. Although the patient received corticosteroids, intravenous immunoglobulin, and plasmapheresis, she continues to have severe neurologic impairment; whether that persistence is a result of delayed therapy or reflects a severe manifestation of OROV-associated GBS remains unclear. The phylogenetic relationship between this case and previously described cases of fetal and adult deaths (2,4), as well as GBS clusters reported in Cuba (5), reinforces the hypothesis that this viral lineage might possess increased pathogenicity (12).
Dr. Garcia Filho is a physician and epidemiologist at the Ceará State Health Department and a professor of medicine at the University of Fortaleza, Brazil. His primary research interests include epidemiologic surveillance and outbreak investigation.
Acknowledgments
We thank the epidemiologic surveillance professionals of the state, the municipality of Capistrano, and the Decentralized Health Area, whose efforts were instrumental in the identification, investigation, and follow-up of the case. We also thank Laboratorio Emilio Ribas, Fortaleza, Ceará, for voluntarily performing laboratory tests not available in the public health system.
The patient involved in this study was fully informed about the nature and purpose of this report. She reviewed the content, provided her consent for publication, and signed an Informed Consent Form in accordance with ethical guidelines.
The study was approved by the Research Ethics Committee of Centro Universitário Christus in accordance with the principles outlined in the Declaration of Helsinki. Ethical approval ensures that the investigation adhered to the highest standards of research ethics and the protection of patient rights.
This research received funding from the Brazilian Health Ministry Department of Science and Technology (file no. 421724/2017-0) and from the Brazilian Health Ministry Health Surveillance Secretariat (file no. 707272/19-002). L.P.d.G.C. receives a productivity grant from the Conselho Nacional de Desenvolvimento Científico e Tecnológico / Ministério da Ciência, Tecnologia e Inovações (CNPq/MCTI; file no. 310579/2022-8).
All authors contributed significantly to the conception, design, and writing of this manuscript and take full responsibility for its content. Each author has reviewed and approved the final version of the manuscript, ensuring its accuracy and compliance with scientific and ethical standards.
This article was preprinted at https://preprints.scielo.org/index.php/scielo/preprint/view/10912/19832.
References
- Naveca FG, Almeida TAP, Souza V, Nascimento V, Silva D, Nascimento F, et al. Human outbreaks of a novel reassortant Oropouche virus in the Brazilian Amazon region. Nat Med. 2024;30:3509–21. DOIPubMedGoogle Scholar
- Garcia Filho C, Lima Neto AS, Maia AMPC, Da Silva LOR, Cavalcante RDC, Monteiro HDS, et al. A case of vertical transmission of Oropouche virus in Brazil. N Engl J Med. 2024;391:2055–7. DOIPubMedGoogle Scholar
- Ribas Freitas AR, Schwartz DA, Lima Neto AS, Rodrigues R, Cavalcanti LPG, Alarcón-Elbal PM. Oropouche virus (OROV): expanding threats, shifting patterns, and the urgent need for collaborative research in Latin America. Viruses. 2025;17:353. DOIPubMedGoogle Scholar
- Bandeira AC, Pereira FM, Leal A, Santos SPO, Barbosa AC, Souza MSPL, et al. Fatal Oropouche virus infections in nonendemic region, Brazil, 2024. Emerg Infect Dis. 2024;30:2370–4. DOIPubMedGoogle Scholar
- de Armas Fernández JR, Peña García CE, Acosta Herrera B, Betancourt Plaza I, Gutiérrez de la Cruz Y, Resik Aguirre S, et al. Report of an unusual association of Oropouche fever with Guillain-Barré syndrome in Cuba, 2024. Eur J Clin Microbiol Infect Dis. 2024;43:2233–7. DOIPubMedGoogle Scholar
- de Lima STS, Hua X, Claro IM, Filho CG, Simões Mello LM, de Jesus R, et al. Molecular epidemiology of Oropouche virus, Ceará State, Brazil, 2024. Emerg Infect Dis. 2025;31:838–42. DOIPubMedGoogle Scholar
- Naveca FG, Nascimento VAD, Souza VC, Nunes BTD, Rodrigues DSG, Vasconcelos PFDC. Multiplexed reverse transcription real-time polymerase chain reaction for simultaneous detection of Mayaro, Oropouche, and Oropouche-like viruses. Mem Inst Oswaldo Cruz. 2017;112:510–3. DOIPubMedGoogle Scholar
- Mansuy JM, Lhomme S, Cazabat M, Pasquier C, Martin-Blondel G, Izopet J. Detection of Zika, dengue and chikungunya viruses using single-reaction multiplex real-time RT-PCR. Diagn Microbiol Infect Dis. 2018;92:284–7. DOIPubMedGoogle Scholar
- Petzold A, Fraser CL, Abegg M, Alroughani R, Alshowaeir D, Alvarenga R, et al. Diagnosis and classification of optic neuritis. Lancet Neurol. 2022;21:1120–34. DOIPubMedGoogle Scholar
- Minh BQ, Schmidt HA, Chernomor O, Schrempf D, Woodhams MD, von Haeseler A, et al. IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol. 2020;37:1530–4. DOIPubMedGoogle Scholar
- Gräf T, Delatorre E, Do Nascimento Ferreira C, Rossi A, Santos HGG, Pizzato BR, et al. Expansion of Oropouche virus in non-endemic Brazilian regions: analysis of genomic characterisation and ecological drivers. Lancet Infect Dis. 2025;25:379–89. DOIPubMedGoogle Scholar
- Scachetti GC, Forato J, Claro IM, Hua X, Salgado BB, Vieira A, et al. Re-emergence of Oropouche virus between 2023 and 2024 in Brazil: an observational epidemiological study. Lancet Infect Dis. 2025;25:166–75. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: April 10, 2026
Table of Contents – Volume 32, Number 4—April 2026
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Carlos Garcia Filho, Rua Oto de Alencar, nº 193, Segundo Andar (CEVEP), Centro, Fortaleza, Ceará, Brazil, CEP 60110-270
Top