Confirming ERVEBO Vaccination to Support Ebola Virus Surveillance
Elif Karaaslan, Amy Whitesell, Jason Malenfant, William C. Carson, Michael Townsend, Kasongo Kayembe Jolie, Enogo Koivogui, Siba Michel Grovogui, Boubacar Diallo, Nouonan Gbamou, Salomon Corvil, Sanaba Boumbaly, Lise Martel, Julie R. Sinclair, Alimou Camara, Trevor Shoemaker, Mary J. Choi, Joel M. Montgomery, Christina F. Spiropoulou, and Éric Bergeron

Author affiliation: Centers for Disease Control and Prevention, Atlanta, Georgia, USA (E. Karaaslan, A. Whitesell, J. Malenfant, W.C. Carson, M. Townsend, L. Martel, J.R. Sinclair, T. Shoemaker, M.J. Choi, J.M. Montgomery, C.T. Spiropoulou, É. Bergeron); African Field Epidemiology Network, Conakry, Guinea (K.K. Jolie, S. Corvil); Ministry of Health, Conakry (E. Koivogui, S.M. Grovogui); US Centers for Disease Control and Prevention, Conakry (B. Diallo); Agence Nationale de Sécurité Sanitaire, Conakry (N. Gbamou); International Center for Research of Tropical Infections in Guinea, N’Zerekore, Guinea (S. Boumbaly); Ministry of Higher Education, Scientific Research, and Innovation, Conakry (A. Camara); University of Georgia, Athens, Georgia, USA (É. Bergeron).
Main Article
Figure 2
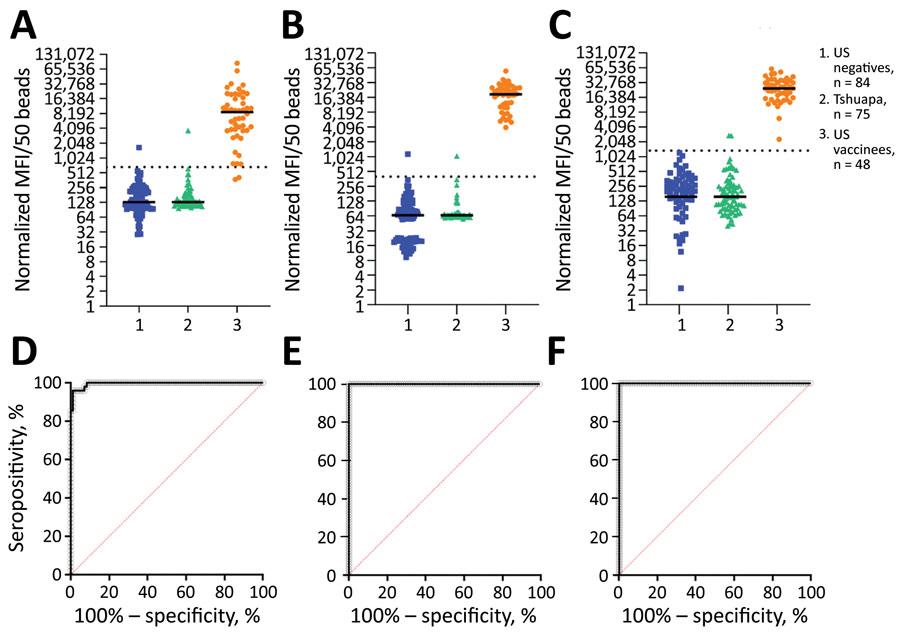
Figure 2. Detection of ERVEBO vaccinees enhanced with EBOV sGP and VSV-P-N for study of development of multiplex assay to confirm Ebola vaccination. A–C) The log2-normalized MFI values for EBOV GP1,2 (A), EBOV sGP (B), and VSV-P-N (C) assays for US negative controls (n = 84), Tshuapa samples (n = 75), and US vaccinees (n = 48) are shown in scatter plots. Horizontal solid lines indicate medians; horizontal dotted lines indicate the assay-specific cutoff values. D–F) Receiver operating characteristic (ROC) curves (black lines) and sensitivity and specificity were measured at the defined cutoffs for EBOV GP1,2 (D), EBOV sGP (E), and VSV-P-N (F). ROC area values: D, 0.9950 (95% CI 0.9870–1.000); E, F, 1.000. Sensitivity: D, 95.8% (95% CI 86.02%–99.26%); E, 100% (95% CI 92.59%–100%); F, 100% (95% CI 92.59%–100%). Specificity: D, 98.6% (95% CI 92.83%–99.93%); E, 97.6% (95% CI 91.73%–99.58%); F, 98.8% (95% CI 93.56%–99.94%). EBOV, Ebola virus; GP, glycoprotein; MFI, mean fluorescence intensity; sGP, secreted glycoprotein; VSV-P-N, vesicular stomatitis virus nucleoprotein N-terminally fused with P peptide.
Main Article
Page created: March 04, 2026
Page updated: April 15, 2026
Page reviewed: April 15, 2026
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.
