Disclaimer: Early release articles are not considered as final versions. Any changes will be reflected in the online version in the month the article is officially released.
Genomic Surveillance of Lassa Virus through In-Country Sequencing, Guinea
Jacob Camara
1, Giuditta Annibaldis
1
, Joon Klaps
1, Kékoura Ifono
1, Fara Raymond Koundouno, Youssouf Sidibé, Sarah Ryter, Moussa Conde, Saa Lucien Millimono, Mette Hinrichs, Julia Hinzmann, Nils Peter Petersen, Mia Le, Annick Renevey, Ehizojie Ehiremen Emua, Philippe Lemey, Simon Dellicour, Stephan Günther, N’Faly Magassouba
2, Sophie Duraffour
2, Liana Eleni Kafetzopoulou
2, and Sanaba Boumbaly
2
Author affiliation: Center for Virology Research—Laboratory for Viral Hemorrhagic Fevers in Guinea, Conakry, Guinea (J. Camara, M. Conde, N. Magassouba, S. Boumbaly); Bernhard Nocht Institute for Tropical Medicine, Hamburg, Germany (G. Annibaldis, S. Ryter, M. Hinrichs, J. Hinzmann, N.P. Petersen, M. Le, A. Renevey, S. Günther, S. Duraffour); German Center for Infection Research, partner site Hamburg–Lübeck–Borstel–Riems, Hamburg (G. Annibaldis, S. Ryter, M. Hinrichs, J. Hinzmann, N.P. Petersen, M. Le, A. Renevey, S. Günther, S. Duraffour); Rega Institute, KU Leuven, Leuven, Belgium (J. Klaps, P. Lemey, S. Dellicour, L.E. Kafetzopoulou); Laboratory for Viral Hemorrhagic Fevers of Guéckédou, Prefectural Health Department of Guéckédou, Guéckédou, Guinea (K. Ifono, F.R. Koundouno, S.L. Millimono); Laboratory for Viral Hemorrhagic Fevers at Regional Hospital of N’Zérékoré, Regional Hospital of N’Zérékoré, N’Zérékoré, Guinea (Y. Sidibé); University of Hamburg, Hamburg (M. Le); Irrua Specialist Teaching Hospital, Irrua, Nigeria (E.E. Emua); Spatial Epidemiology Lab, Université Libre de Bruxelles, Brussels, Belgium (S. Dellicour); Interuniversity Institute of Bioinformatics in Brussels, Université Libre de Bruxelles, Vrije Universiteit Brussel, Brussels (S. Dellicour); Leiden University Medical Center, Leiden, the Netherlands (L.E. Kafetzopoulou)
Main Article
Figure 2
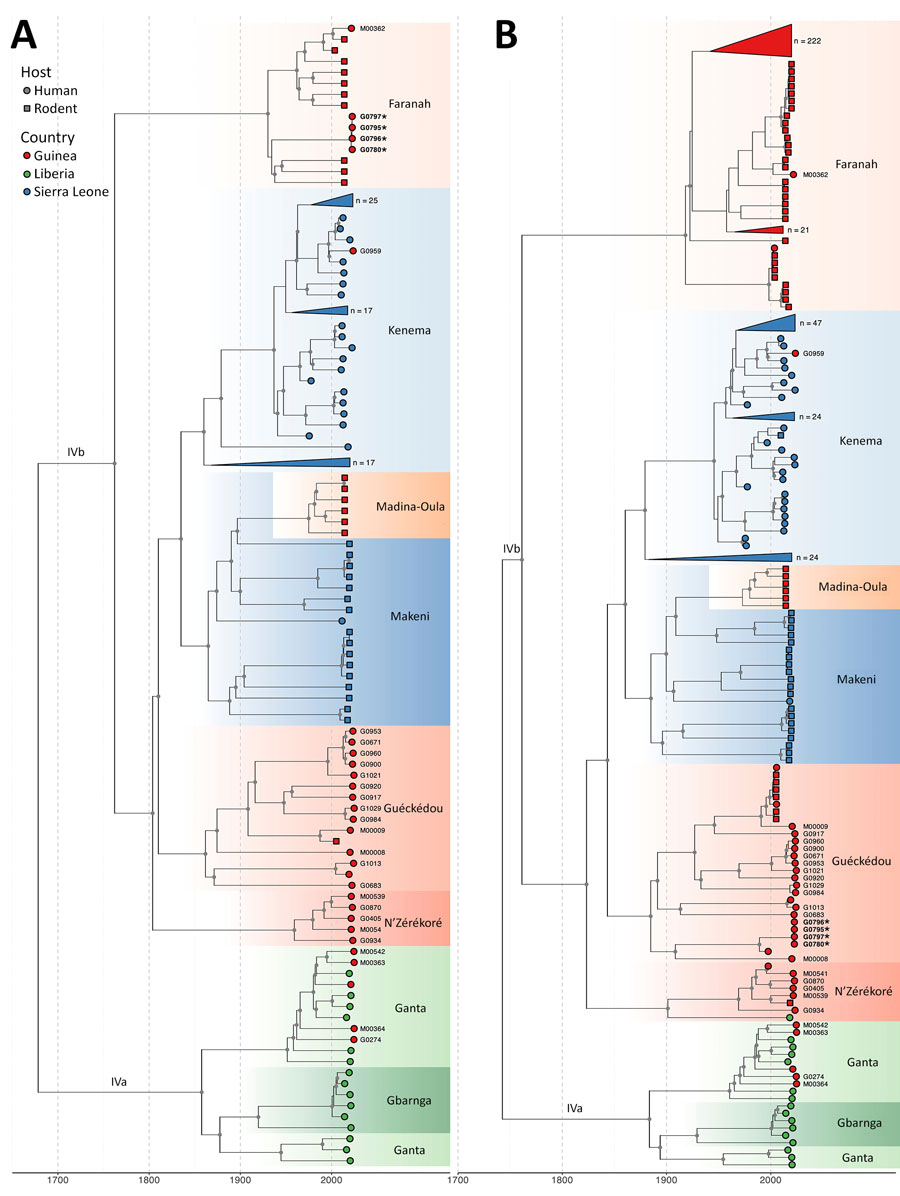
Figure 2. Temporal evolution of the large and small segments of Lassa virus from study of genomic surveillance of Lassa virus through in-country sequencing, Guinea. Time-scaled maximum clade credibility trees are shown for the large (A) and small (B) segments. Tips are colored by country of origin. Sublineages are annotated and colored by their predominant geographic location (e.g., Kenema, Faranah, Ganta). Clades that fall outside the sequence diversity of interest (i.e., clades that have no association with the sequences reported in this article) are collapsed and annotated with the total number of sequences they include. Gray dots indicate internal nodes with a clade credibility of >80%. Sequences reported in this manuscript have their sample identification codes indicated next to their respective tip. We detected 2 cocirculating sublineages of Lassa virus in N’Zérékoré: a locally established IVb lineage (M00539, M00541, G0405, G0870, G0934), and the IVa lineage linked to the region of Ganta in Liberia (M00363, M00542, M00364, G0274). The sample from Guéckédou (G0959) grouped within the Kenema, Sierra Leone, Lassa virus cluster, reflecting its travel-linked origin. Strains that form the nosocomial transmission chain with reassorted genomes have been highlighted in bold and are indicated with a star.
Main Article
Page created: March 23, 2026
Page updated: April 28, 2026
Page reviewed: April 28, 2026
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.
