Volume 5, Number 5—October 1999
Synopsis
West Nile Fever–a Reemerging Mosquito-Borne Viral Disease in Europe
Cite This Article
Citation for Media
Abstract
West Nile virus causes sporadic cases and outbreaks of human and equine disease in Europe (western Mediterranean and southern Russia in 1962-64, Belarus and Ukraine in the 1970s and 1980s, Romania in 1996-97, Czechland in 1997, and Italy in 1998). Environmental factors, including human activities, that enhance population densities of vector mosquitoes (heavy rains followed by floods, irrigation, higher than usual temperature, or formation of ecologic niches that enable mass breeding of mosquitoes) could increase the incidence of West Nile fever.
The 1996-97 outbreak of West Nile fever in and near Bucharest, Romania, with more than 500 clinical cases and a case-fatality rate approaching 10% (1-3), was the largest outbreak of arboviral illness in Europe since the Ockelbo-Pogosta-Karelian fever epidemic caused by Sindbis virus in northern Europe in the 1980s. This latest outbreak reaffirmed that mosquito-borne viral diseases may occur on a mass scale, even in temperate climates.
West Nile virus is a member of the Japanese encephalitis antigenic complex of the genus Flavivirus, family Flaviviridae (4). All known members of this complex (Alfuy, Japanese encephalitis, Kokobera, Koutango, Kunjin, Murray Valley encephalitis, St. Louis encephalitis, Stratford, Usutu, and West Nile viruses) are transmissible by mosquitoes and many of them can cause febrile, sometimes fatal, illnesses in humans.
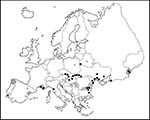
West Nile virus was first isolated from the blood of a febrile woman in the West Nile district of Uganda in 1937 (5) and was subsequently isolated from patients, birds, and mosquitoes in Egypt in the early 1950s (6-7). The virus was soon recognized as the most widespread of the flaviviruses, with geographic distribution including Africa and Eurasia. Outside Europe (Figure), the virus has been reported from Algeria, Asian Russia, Azerbaijan, Botswana, Central African Republic, Côte d'Ivoire, Cyprus, Democratic Republic of Congo (former Zaire), Egypt, Ethiopia, India, Israel, Kazakhstan, Madagascar, Morocco, Mozambique, Nigeria, Pakistan, Senegal, South Africa, Tajikistan, Turkmenia, Uganda, and Uzbekistan. Furthermore, West Nile virus antibodies have been detected in human sera from Armenia, Borneo, China, Georgia, Iraq, Kenya, Lebanon, Malaysia, the Philippines, Sri Lanka, Sudan, Syria, Thailand, Tunisia, and Turkey (8-10). Kunjin virus is closely related to West Nile virus (11,12), representing a counterpart or subtype for Australia and Southeast Asia; some West Nile virus seroreactions in Southeast Asia may, in fact, represent antibodies to Kunjin virus.
Arthropod Vectors
Mosquitoes, largely bird-feeding species, are the principal vectors of West Nile virus. The virus has been isolated from 43 mosquito species, predominantly of the genus Culex (Table 1). In Africa and the Middle East, the main vector is Cx. univittatus (although Cx. poicilipes, Cx. neavei, Cx. decens, Aedes albocephalus, or Mimomyia spp. play an important role in certain areas). In Europe, the principal vectors are Cx. pipiens, Cx. modestus, and Coquillettidia richiardii, and in Asia, Cx. quinquefasciatus, Cx. tritaeniorhynchus, and Cx. vishnui predominate. Successful experimental transmission of the virus has been described in Culiseta longiareolata, Cx. bitaeniorhynchus, and Ae. albopictus (8,13). Transovarial transmission of the virus has been demonstrated in Cx. tritaeniorhynchus, Ae. aegypti, and Ae. albopictus, though at low rates.
Virus isolations have occasionally been reported from other hematophagous arthropods (e.g., bird-feeding argasid [soft] or amblyommine [hard] ticks) (Table 1), and experimental transmission has been observed in Ornithodoros savignyi, O. moubata, O.maritimus, O. erraticus, Rhipicephalus sanguineus, R. rossicus, Dermacentor reticulatus, and Haemaphysalis leachii (8,13).
Vertebrate Hosts
Wild birds are the principal hosts of West Nile virus. The virus has been isolated from a number of wetland and terrestrial avian species in diverse areas (7-10,14-16). High, long-term viremia, sufficient to infect vector mosquitoes, has been observed in infected birds (7,17,18). The virus persists in the organs of inoculated ducks and pigeons for 20 to 100 days (18). Migratory birds are therefore instrumental in the introduction of the virus to temperate areas of Eurasia during spring migrations (12,14-16,19).
Rarely, West Nile virus has been isolated from mammals (Arvicanthis niloticus, Apodemus flavicollis, Clethrionomys glareolus, sentinel mice and hamsters, Lepus europaeus, Rousettus leschenaulti, camels, cattle, horses, dogs, Galago senegalensis, humans) in enzootic foci (8-10). Mammals are less important than birds in maintaining transmission cycles of the virus in ecosystems. Only horses and lemurs (20) have moderate viremia and seem to support West Nile virus circulation locally. Frogs (Rana ridibunda) also can harbor the virus, and their donor ability for Cx. pipiens has been confirmed (21).
Transmission Cycles
Although Palearctic natural foci of West Nile virus infections are mainly situated in wetland ecosystems (river deltas or flood plains) and are characterized by the bird-mosquito cycle, argasid and amblyommine ticks may serve as substitute vectors and form a bird-tick cycle in certain dry and warm habitats lacking mosquitoes. Even a frog-mosquito cycle (21) may function under certain circumstances.
In Europe, West Nile virus circulation is confined to two basic types of cycles and ecosystems: rural (sylvatic) cycle (wild, usually wetland birds and ornithophilic mosquitoes) and urban cycle (synanthropic or domestic birds and mosquitoes feeding on both birds and humans, mainly Cx. pipiens/molestus). The principal cycle is rural, but the urban cycle predominated in Bucharest during the 1996-97 outbreak (2,3). Circulation of West Nile fever in Europe is similar to that of St. Louis encephalitis in North America, where the rural cycle of exoanthropic birds—Cx. tarsalis alternates with the urban cycle of synanthropic birds—Cx. pipiens/quinquefasciatus.
Humans
West Nile fever in humans usually is a febrile, influenzalike illness, characterized by an abrupt onset (incubation period is 3 to 6 days) of moderate to high fever (3 to 5 days, infrequently biphasic, sometimes with chills), headache (often frontal), sore throat, backache, myalgia, arthralgia, fatigue, conjunctivitis, retrobulbar pain, maculopapular or roseolar rash (in approximately half the cases, spreading from the trunk to the extremities and head), lymphadenopathy, anorexia, nausea, abdominal pain, diarrhea, and respiratory symptoms (9). Occasionally (<15% of cases), acute aseptic meningitis or encephalitis (associated with neck stiffness, vomiting, confusion, disturbed consciousness, somnolence, tremor of extremities, abnormal reflexes, convulsions, pareses, and coma), anterior myelitis, hepatosplenomegaly, hepatitis, pancreatitis, and myocarditis occur. Laboratory findings involve a slightly increased sedimentation rate and a mild leukocytosis; cerebrospinal fluid in patients with central nervous system involvement is clear, with moderate pleocytosis and elevated protein. The virus can be recovered from the blood for up to 10 days in immunocompetent febrile patients, as late as 22 to 28 days after infection in immunocompromised patients; peak viremia occurs 4 to 8 days postinfection. Recovery is complete (less rapid in adults than in children, often accompanied by long-term myalgias and weakness), and permanent sequelae have not been reported. Most fatal cases have been recorded in patients older than 50 years. Many of the West Nile fever symptoms have been reproduced in volunteers with underlying neoplastic disease who had been inoculated with virus to achieve pyrexia and oncolysis (22).
Hundreds of West Nile fever cases have been described in Israel and South Africa. The largest African epidemic, with approximately 3,000 clinical cases, occurred in an arid region of the Cape Province after heavy rains in 1974 (23). An outbreak with approximately 50 patients, eight of whom died, was described in Algeria in 1994 (1). Other cases or outbreaks have been observed in Azerbaijan, Central African Republic, Democratic Republic of Congo (former Zaire), Egypt, Ethiopia, India, Madagascar, Nigeria, Pakistan, Senegal, Sudan, and in a few European countries.
Horses
Equine disease, called Near Eastern equine encephalitis in Egypt and lourdige in France, was observed and experimentally reproduced as fever and diffuse encephalomyelitis with a moderate to high fatality rate in Egypt (24), France (c. 50 cases in 1962-65) (25), Italy (14 cases in 1998, six died or were euthanised) (R. Lelli, G. Ferrari, pers. comm.), Portugal (26) and Morocco (42 of 94 affected horses died) (27). In the 1960s, the biphasic, encephalomyelitic form, which causes staggering gait and weakness to paralysis of the hind legs, was apparent among infected semiferal horses in Camargue (25).
Other Mammals
Inoculation of sheep with West Nile virus results in fever, abortion in pregnant ewes, and rare encephalitis, in contrast to the asymptomatic infection seen in pigs and dogs (9,28). Rabbits, adult albino rats, and guinea pigs are resistant to West Nile virus infection, but laboratory mice and Syrian hamsters are markedly susceptible; they often become ill with fatal encephalitis, even when inoculated peripherally (8). Adult rodents stressed or immunosuppressed by cold, isolation, cyclophosphamide, corticosterone, or bacterial endotoxin contract fatal encephalitis, even when an attenuated viral strain is given (29). Inoculation of rhesus and bonnet monkeys (but not cynomolgus monkeys or chimpanzees) causes fever, ataxia, and prostration with occasional encephalitis, tremor of extremities, pareses, or paralysis. Infection may be fatal or cause long-term virus persistence in survivors (5,6,30).
Birds
Birds usually do not show any symptoms when infected with West Nile virus. However, natural disease due to the virus has been observed in a pigeon in Egypt (7), and inoculation of certain avian species (e.g., pigeons, chickens, ducks, gulls, and corvids) causes occasional encephalitis and death or long-term virus persistence (7,10,17,18). Chick embryos may be killed by the virus (8).
In Europe, the presence of West Nile virus was indicated in 1958, when two Albanians had specific West Nile virus antibodies (31). The first European isolations of the virus were recorded in 1963 from patients and mosquitoes in the Rhône Delta (32) and from patients and Hyalomma marginatum ticks in the Volga Delta (33,34). West Nile virus was subsequently isolated in Portugal (35), Slovakia (36), Moldavia (37), Ukraine (38), Hungary (39), Romania (2), Czechland (40), and Italy (V. Deubel, G. Ferrari, pers. comm.).
The incidence of West Nile fever in Europe is largely unknown. In the 1960s, cases were observed in southern France (25), southern Russia (41), Spain (26), southwestern Romania (42), in the 1970s, 1980s, and 1990s in Belarus (43), western Ukraine (44), southeastern Romania (1,2), and Czechland (45). West Nile fever in Europe occurs during the period of maximum annual activity of mosquito vectors (July to September) (Table 2).
West Nile virus can cause sporadic human cases, clusters, or outbreaks of West Nile fever, even in temperate Europe. Environmental factors, including human activities that enhance vector population densities (irrigation, heavy rains followed by floods, higher than usual temperatures, and formation of ecologic niches enabling the mass breeding of mosquitoes) allow the reemergence of this mosquito-borne disease. For instance, global warming scenarios hypothesize warmer, more humid weather that may produce an increase in the distribution and abundance of mosquito vectors (71). Surveillance for West Nile fever (monitoring population densities and infection rates of principal vectors, serosurveys on vertebrates and exposed human groups, and routine diagnosis of human infections) should therefore be carried out in affected areas.
The mechanism of West Nile virus persistence in disease-endemic foci of temperate Europe presents a challenge for further research. General hypotheses of how an arbovirus could overwinter under adverse climatic conditions have already been postulated (72). The virus could persist in hibernating female Culex spp.; transovarially infected Culex spp. progeny; or chronically infected vertebrate hosts, perhaps birds or frogs. Alternatively, the virus may be reintroduced by chronically infected migratory birds from tropical or subtropical foci at irregular intervals. These issues have to be addressed, because present data substantiate all particular mechanisms and their combinations. For instance, the hibernating vector idea has been supported by a few field and experimental data on female Cx. univittatus (7,73). On the other hand, if the reintroduction scheme is correct, a greatly increased activity of West Nile virus in Africa should be followed by an epidemic occurrence of West Nile fever in Europe in the next few years.
Zdenek Hubálek and Jirí Halouzka are scientists at the Academy of Sciences of the Czech Republic. They are interested in the ecology of arthropodborne human pathogenic viruses and bacteria.
References
- Le Guenno B, Bougermouh A, Azzam T, Bouakaz R. West Nile: A deadly virus? Lancet. 1996;348:1315. DOIPubMedGoogle Scholar
- Tsai TF, Popovici F, Cernescu C, Campbell GL, Nedelcu NI. West Nile encephalitis epidemic in southeastern Romania. Lancet. 1998;352:767–71. DOIPubMedGoogle Scholar
- Savage HM, Ceianu C, Nicolescu G, Karabatsos N, Lanciotti R, Vladimirescu A, Entomologic and avian investigations of an epidemic of West Nile fever in Romania, 1996, with serologic and molecular characterization of a virus isolate from mosquitoes. Am J Trop Med Hyg. 1999. In press.
- Calisher CH, Karabatsos N, Dalrymple JM, Shope RE, Porterfield JS, Westaway EG, Antigenic relationships between flaviviruses as determined by cross-neutralization tests with polyclonal antisera. J Gen Virol. 1989;70:37–43. DOIPubMedGoogle Scholar
- Smithburn KC, Hughes TP, Burke AW, Paul JH. A neurotropic virus isolated from the blood of a native of Uganda. Am J Trop Med Hyg. 1940;20:471–92.
- Melnick JL, Paul JR, Riordan JT, Barnett VHH, Goldblum N, Zabin E. Isolation from human sera in Egypt of a virus apparently identical to West Nile virus. Proc Soc Exp Biol Med. 1951;77:661–5.PubMedGoogle Scholar
- Taylor RM, Work TH, Hurlbut HS, Rizk F. A study of the ecology of West Nile virus in Egypt. Am J Trop Med Hyg. 1956;5:579–620.PubMedGoogle Scholar
- Karabatsos N, ed. International catalogue of arboviruses, including certain other viruses of vertebrates. 3rd ed., and Supplements 1986-98. San Antonio: American Society of Tropical Medicine and Hygiene; 1985.
- Peiris JSM, Amerasinghe FP. West Nile fever. In: Beran GW, Steele JH, editors. Handbook of zoonoses. Section B: Viral. 2nd ed. Boca Raton (FL): CRC Press; 1994. p. 139-48.
- Hubálek Z, Halouzka J. Arthropod-borne viruses of vertebrates in Europe. Acta Scientiarum Naturalium Brno. 1996;30:1–95.
- Zanotto PM A, Gould EA, Gao GF, Harvey PH, Holmes EC. Population dynamics of flaviviruses revealed by molecular phylogenies. Proc Natl Acad Sci U S A. 1996;93:548–53. DOIPubMedGoogle Scholar
- Berthet FX, Zeller HG, Drouet MT, Rauzier J, Digoutte JP, Deubel V. Extensive nucleotide changes and deletions within the envelope glycoprotein gene of Euro-African West Nile viruses. J Gen Virol. 1997;78:2293–7.PubMedGoogle Scholar
- Hurlbut HS. West Nile virus infection in arthropods. Am J Trop Med Hyg. 1956;5:76–85.PubMedGoogle Scholar
- Nir Y, Goldwasser R, Lasowski Y, Avivi A. Isolation of arboviruses from wild birds in Israel. Am J Epidemiol. 1967;86:372–8.PubMedGoogle Scholar
- Watson GE, Shope RE, Kaiser MN. An ectoparasite and virus survey of migratory birds in the eastern Mediterranean. In: Cherepanov IA, editor. Transcontinental connections of migratory birds and their role in the distribution of arboviruses. Novosibirsk: Nauka; 1972 p.176-80.
- Ernek E, Kozuch O, Nosek J, Teplan J, Folk C. Arboviruses in birds captured in Slovakia. Journal of Hygiene, Epidemioloigy. Microbiology and Immunology (Prague). 1977;21:353–9.
- Work TH, Hurlbut HS, Taylor RM. Indigenous wild birds of the Nile Delta as potential West Nile virus circulating reservoirs. Am J Trop Med Hyg. 1955;4:872–8.PubMedGoogle Scholar
- Semenov BF, Chunikhin SP, Karmysheva VY, Yakovleva NI. Studies of chronic arbovirus infections in birds. 1. Experiments with West Nile, Sindbis, Bhanja and SFS viruses [in Russian]. Vestnik Akademii Medicinskikh NaukSSSR (Moskva). 1973;2:79–83.
- Hannoun C, Corniou B, Mouchet J. Role of migrating birds in arbovirus transfer between Africa and Europe. In: Cherepanov AI, editor. Transcontinental connections of migratory birds and their role in the distribution of arboviruses. Novosibirsk: Nauka; 1972; p. 167-72.
- Rodhain F, Petter JJ, Albignac R, Coulanges P, Hannoun C. Arboviruses and lemurs in Madagascar: experimental infection of Lemur fulvus with yellow fever and West Nile viruses. Am J Trop Med Hyg. 1985;34:816.PubMedGoogle Scholar
- Kostyukov MA, Alekseev AN, Bulychev VP, Gordeeva ZE. Experimental infection of Culex pipiens mosquitoes with West Nile virus by feeding on infected Rana ridibunda frogs and its subsequent transmission [in Russian]. Med Parazitol (Mosk). 1986;6:76–8.PubMedGoogle Scholar
- Southam CM, Moore AE. Induced virus infections in man by the Egypt isolates of West Nile virus. Am J Trop Med Hyg. 1954;3:19–50.PubMedGoogle Scholar
- McIntosh BM, Jupp PG, Dos Santos I, Meenehan GM. Epidemics of West Nile and Sindbis viruses in South Africa with Culex (Culex) univittatus Theobald as vector. S Afr J Sci. 1976;72:295–300.
- Schmidt JR, El Mansoury HK. Natural and experimental infection of Egyptian equines with West Nile virus. Am J Trop Med Hyg. 1963;57:415–27.
- Hannoun C, Panthier R, Corniou B. Epidemiology of West Nile infections in the South of France. In: Bárdoš V, editor. Arboviruses of the California complex and the Bunya-mwera group. Bratislava: Publ House SAS; 1969. p. 379-87.
- Filipe AR, De Andrade HR. Arboviruses in the Iberian Peninsula. Acta Virol. 1990;34:582–91.PubMedGoogle Scholar
- Abdelhaq AT. West Nile fever in horses in Morocco. Bull Off Int Epizoot. 1996;108:867–9.
- Barnard BJH, Voges SF. Flaviviruses in South Africa: pathogenicity for sheep. Onderstepoort J Vet Res. 1986;53:235–8.PubMedGoogle Scholar
- Ben-Nathan D, Lustig S, Feuerstein G. The influence of cold or isolation stress on neuroinvasiveness and virulence of an attenuated variant of West Nile virus. Arch Virol. 1989;109:1–10. DOIPubMedGoogle Scholar
- Pogodina VV, Frolova MP, Malenko GV, Fokina GI, Koreshkova GV, Kiseleva LL, Study on West Nile virus persistence in monkeys. Arch Virol. 1983;75:71–86. DOIPubMedGoogle Scholar
- Bárdoš V, Adamcová J, Dedei S, Gjini N, Rosický B, Šimková A. Neutralizing antibodies against some neurotropic viruses determined in human sera in Albania. Journal of Hygiene, Epidemiology. Microbiology and Immunology (Prague). 1959;3:277–82.
- Hannoun C, Panthier R, Mouchet J, Eouzan JP. Isolement en France du virus West Nile à partir de malades et du vecteur Culex molestus Ficalbi. Compte Rendu de l'Académie des Sciences. 1964;D259:4170–2.
- Chumakov MP, Belyaeva AP, Butenko AM. Isolation and study of an original virus from Hyalomma plumbeum plumbeum ticks and from the blood of a febrile patient in the Astrakhan region (in Russian). Materialy XI Nauchnoi Sessii Instituta Poliomielita i Virusnykh Encefalitov (Moskva) 1964:5-7.
- Butenko AM, Chumakov MP, Stolbov DN. Serological and virological examinations in a natural focus of West Nile fever in the Astrakhan region [in Russian]. Voprosy Medicinskoi Virusologii. 1967;1:208–11.
- Filipe AR. Isolation in Portugal of West Nile virus from Anopheles maculipennis mosquitoes. Acta Virol. 1972;16:361.PubMedGoogle Scholar
- Labuda M, Kozuch O, Grešíková M. Isolation of West Nile virus from Aedes cantans mosquitoes in west Slovakia. Acta Virol. 1974;18:429–33.
- Chumakov MP, Bashkirtsev VN, Golger EI, Dzagurova TK, Zavodova TI, Konovalov YN, Isolation and identification of Crimean hemorrhagic fever and West Nile fever viruses from ticks collected in Moldavia [in Russian]. Medicinskaya Virusologia. 1974;22:45–9.
- Vinograd IA, Obukhova VR. Isolation of arboviruses from birds in western Ukraine [in Russian]. Sbornik Trudov Instituta Virusologii (Moskva). 1975;3:84–7.
- Molnár E, Gulyas MS, Kubinyi L, Nosek J, Kozuch O, Ernek E, Studies on the occurrence of tick-borne encephalitis in Hungary. Acta Vet Acad Sci Hung. 1976;26:419–38.PubMedGoogle Scholar
- Hubálek Z, Halouzka J, Juricová Z, Šebesta O. First isolation of mosquito-borne West Nile virus in the Czech Republic. Acta Virol. 1998;42:119–20.PubMedGoogle Scholar
- Butenko AM, Chumakov MP, Bashkirtsev VN, Tkachenko EA, Rubin SG, Stolbov DN. New investigations of West Nile virus infections in the USSRAstrakhan region [in Russian]. Materialy XV Nauchnoi Sessii Instituta Poliomielita i Virusnykh Encefalitov (Moskva). 1968;3:175–6.
- Topciu V, Rosiu N, Arcan P. Contribution to the study of arboviruses in Banat. Rev Roum Inframicrobiol. 1971;8:101–6.PubMedGoogle Scholar
- Voinov IN, Rytik PG, Grigoriev AI. Arbovirus infections in Belarus (in Russian). In: Drozdov SG, editor. Virusy i virusnyje infektsii. Moskva: Inst Poliomiel Virus Enc; 1981. p. 86-7.
- Buletsa BA, Turak JA, Korol MJ, Ignatovich II, Vitvitskyi AA. Neurological manifestations of West Nile fever in the Transcarpathian region, Ukrainian SSR [in Russian]. Zhurnal Nevrologii Psikhiatrii. 1989;89:29–30.
- Hubálek Z, Halouzka J, Juricová Z. West Nile fever in Czechland. Emerg Infect Dis. 1999;5:594–5. DOIPubMedGoogle Scholar
- Rollin PE, Rollin D, Martin P, Baylet R, Rodhain F, Hannoun C. Résultats d'enquetes séroépidemiologiques récentes sur les arboviroses en Camargue: populations humaines, équines, bovines et aviaires. Med Mal Infect. 1982;12:77–80. DOIGoogle Scholar
- Verani P, Balducci M, Lopes MC. Arboviruses in Italy. In: Kurstak E, editor. Arctic and tropical arboviruses. New York: Academic Press; 1979. p. 101-21.
- Vesenjak-Hirjan J, Punda-Poli V, Dobec M. Geographical distribution of arboviruses in Yugoslavia. Journal of Hygiene, Epidemiology. Microbiology and Immunology (Prague). 1991;35:129–40.
- Eltari E, Gina A, Bitri T, Sharofi F. Some data on arboviruses, especially tick-borne encephalitis, in Albania. Giornale de Malattie Infettive e Parassitaire. 1993;45:404–11.
- Koptopoulos G, Papadopoulos O. A serological survey for tick-borne encephalitis and West Nile viruses in Greece. Zentralbl Bakteriol. 1980;Suppl 9:185–8.
- Antoniadis A, Alexiou-Daniel S, Malissiovas N, Doutsos J, Polyzoni T, LeDuc JW, Seroepidemiological survey for antibodies to arboviruses in Greece. Arch Virol. 1990;Suppl 1:277–85.
- Rusakiev M. Studies on the distribution of arboviruses transmitted by mosquitoes in Bulgaria. In: Bárdoš V, editor. Arboviruses of the California complex and the Bunyamwera group. Bratislava: Publ House SAS; 1969. p. 389-92.
- Katsarov G, Vasilenko S, Vargin V, Butenko S, Tkachenko E. Serological studies on the distribution of some arboviruses in Bulgaria. Problems of Infectious and Parasitic Diseases. 1980;8:32–5.
- Draganescu N, Gheorghiu V. On the presence of group B arbovirus infections in Romania. Investigations on the incidence of West Nile antibodies in humans and certain domestic animals. Rev Roum Inframicrobiol. 1968;5:255–8.
- Antipa C, Girjabu E, Iftimovici R, Draganescu N. Serological investigations concerning the presence of antibodies to arboviruses in wild birds. Revue Roumaine de Médicine - Virologie 1984;35:5-9.
- Molnár E, Grešíková M, Kubászová T, Kubínyi L, Szabó JB. Arboviruses in Hungary. Journal of Hygiene, Epidemiology. Microbiology and Immunology (Prague). 1973;17:1–10.
- Kozuch O, Nosek J, Labuda M. Arboviruses occurring in western Slovakia and their hosts. In: Labuda M, Calisher CH, editors. New aspects in ecology of arboviruses. Bratislava: Inst Virol SAS; 1980. p. 323-33.
- Kozuch O, Nosek J, Grešíková M, Ernek E. Surveillance on mosquito-borne natural focus in Záhorská lowland. In: Sixl W, Troger H, editors. Naturherde von Infektionskrankheiten in Zentraleuropa. Graz: Hyg Inst Univ; 1976. p. 115-8.
- Sekeyová M, Grešíková M, Kozuch O. Haemagglutination-inhibition antibodies to some arboviruses in sera of pigeons trapped in Bratislava. In: Sixl W, Troger H, editors. Naturherde von Infektionskrankheiten in Zentraleuropa. Graz: Hyg Inst Univ; 1976. p. 187-9.
- Grešíková M, Sekeyová M. Haemagglutination-inhibiting antibodies against arboviruses in the population of Slovakia. Journal of Hygiene, Epidemiology. Microbiology and Immunology (Prague). 1967;11:278–85.
- Aspöck H, Kunz C, Picher O, Böck F. Virologische und serologische Untersuchungen über die Rolle von Vögeln als Wirte von Arboviren in Österreich. Zentralbl Bakteriol. 1973;A224:156–67.
- Grešíková M, Thiel W, Batiková M, Stünzner D, Sekeyová M, Sixl W. Haemagglutination-inhibiting antibodies against arboviruses in human sera from different regions in Steiermark (Austria). Zentralbl Bakteriol. 1973;224:298–302.
- Grešíková M, Sekeyová M, Vošta J, Hanák P. Haemagglutination-inhibiting antibodies to some arboviruses in human and animal sera from Ceské Budejovice. In: Sixl W, editor. Naturherde von Infektionskrankheiten in Zentraleuropa. Graz-Seggau: Hyg Inst Univ; 1979. p. 25-9.
- Juricová Z. Antibodies to arboviruses in game animals in Moravia, Czechland [in Czech]. Veterinární Medicina (Praha). 1992;37:633–6.
- Juricová Z, Hubálek Z, Halouzka J, Machácek P. Virological examination of cormorants for arboviruses [in Czech]. Veterinární Medicina (Praha). 1993;38:375–9.
- Hubálek Z. Jur icová Z, Halouzka J, Pellantová J, Hudec K. Arboviruses associated with birds in southern Moravia, Czechoslovakia. Acta Scientiarum Naturalium Brno. 1989;23:1–50.
- Juricová Z, Halouzka J. Serological examination of domestic ducks in southern Moravia for antibodies against arboviruses of the groups A, B, California and Bunyamwera [in Czech]. Biologia (Bratisl). 1993;48:481–4.
- Juricová Z, Pinowski J, Literák I, Hahm KH, Romanowski J. Antibodies to Alphavirus, Flavivirus, and Bunyavirus arboviruses in house sparrows (Passer domesticus) and tree sparrows (P. montanus) in Poland. Avian Dis. 1998;42:182–5. DOIPubMedGoogle Scholar
- Vinograd IA, Beletskaya GV, Chumachenko SS, Omelchenko GA, Lozinski IN, Yartys OS, Ecological aspects of arbovirus studies in the Ukrainian SSR (in Russian). In: Lvov DK, Gaidamovich SY, editors. Ecology of viruses and diagnostics of arbovirus infections. Moscow: Acad Med Sci USSR; 1989. p. 21-7.
- Chumakov MP, Spasskiy AA, Uspenskaya IG, Tikhon EI, Zaitsev NA, Konovalov YN, Virological, serological, zoological, and ecological investigations of natural foci of arbovirus infections in Moldavian SSR (in Russian). Abstr Conf "Viruses and virus infections of humans." Moskva 1981, p. 101.
- Reeves WC, Hardy JL, Reisen WK, Milby MM. Potential effect of global warming on mosquito-borne arboviruses. J Med Entomol. 1994;310:323–32.
- Cornel AJ, Jupp PG, Blackburn NK. Effect of environmental temperature on the vector competence of Culex univittatus (Diptera, Culicidae) for West Nile virus. J Med Entomol. 1993;30:449–56.PubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 5, Number 5—October 1999
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Z. Hubálek, Institute of Vertebrate Biology, Academy of Sciences, Klášterní 2, CZ-69142 Valtice, Czech Republic; fax: 420-627-352-387
Top