Volume 7, Number 1—February 2001
Research
Emerging Chagas Disease: Trophic Network and Cycle of Transmission of Trypanosoma cruzi from Palm Trees in the Amazon
Cite This Article
Citation for Media
Abstract
A trophic network involving molds, invertebrates, and vertebrates, ancestrally adapted to the palm tree (Attalaea phalerata) microhabitat, maintains enzootic Trypanosoma cruzi infections in the Amazonian county Paço do Lumiar, state of Maranhão, Brazil. We assessed seropositivity for T. cruzi infections in the human population of the county, searched in palm trees for the triatomines that harbor these infections, and gathered demographic, environmental, and socioeconomic data. Rhodnius pictipes and R. neglectus in palm-tree frond clefts or in houses were infected with T. cruzi (57% and 41%, respectively). Human blood was found in 6.8% of R. pictipes in houses, and 9 of 10 wild Didelphis marsupialis had virulent T. cruzi infections. Increasing human population density, rain forest deforestation, and human predation of local fauna are risk factors for human T. cruzi infections.
The tropical moist broadleaf forests of Latin America are an important region for conservation of biodiversity (1). In the Amazon Basin (area 8,214,284 km2), blocks of original habitat are still intact, while some ecoregions 1 are almost completely converted or degraded, allowing major components of biodiversity to steadily erode (1,2). Populations of several endangered wildlife species have declined, and human habitat and land use are considered a threat to most native species and communities.
Palm trees propagate in ecoregions of the Amazon Basin ecosystem amid other vegetation or in enormous palm forests. Of approximately 2,800 palm species worldwide, 387 (13.8%) are native to the basin (3,4). Palm trees have been used to study the evolution of biological diversity and are excellent markers of ecologic fitness in the Amazon Basin (2,4). In addition, these trees may play an important role in the forest ecosystem. Palm trees produce 15 tons of dry organic material per hectare per year (threefold more than other species of trees) and recuperate more rapidly after fire than other forest species. A single native palm tree may serve as shelter and food for diverse fauna (wild mammals, snakes, scorpions, spiders, amphibians, and many species of insects). Palm trees are also an important economic resource for residents of the Amazon region, who collect and sell palm roots, stipe, leaves, fruits, seeds, heart of palm, and inflorescences (4,5). The babassu palm Attalaea phalerata reaches an average density of 200 trees per hectare in the state of Maranhão, but lower densities were reported in the states of Piaui, Goiás-Tocantins, and Mato Grosso (5), where babassu trees number an estimated 11 x 108.
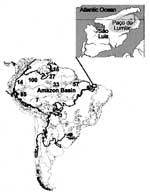
Not much attention has been given to human health conditions in this ecosystem (6), probably because pathologic conditions in tropical broadleaf forests are difficult to quantify in these isolated, often impoverished communities. Nineteen sylvatic species of triatomines were identified in the Amazon Basin (7-10), six in association with palm tree microhabitats (Table 1). Eleven of these species were infected with T. cruzi or T. cruzi-like flagellates (17,18). None of these triatomine species, with the possible exception of T. rubrofasciata, have adapted to human habitats in the Amazon Basin (19). Since 1924, when T. cruzi infection in wild squirrel monkeys (Crisotrix sciureus) was described (20), sporadic human T. cruzi infections have been reported in the basin (21). However, this enzootic protozoan infection received attention only after 1969, when acute cases of human Chagas disease were described in Belém, State of Pará, Brazil (22,23). Further evidence shows that T. cruzi infections are endemic in the Amazon Basin (12-15,21-28) (Figures 1 and 2).
Spellerberg and Hardes (29) describe the major threats to rain forest conservation as shifting agriculture, cattle ranching, logging, and industrialization (mining, hydroelectric dams), to which we add land colonization. We hypothesized that certain effects of human economic activity (population growth, land use, deforestation, lack of environmental education programs, and human predation on mammals and birds) in a defined ecoregion of this major ecosystem may therefore pose a risk for outbreaks of acute Chagas disease. We evaluated these risk factors in the Tocantins River moist forest ecoregion (1), which is considered vulnerable to human intervention. We conducted a pilot study to assess seropositivity for T. cruzi infections in the human population of Paço do Lumiar County, state of Maranhão, Brazil, and to search for triatomines that harbor these infections in palm trees. We also gathered and analyzed demographic, environmental, and socioeconomic data.
The field study was carried out in Paço do Lumiar county (population 55,000), 20 km from São Luiz, capital of the state of Maranhão. The county is part of the Tocantins moist forest ecoregion (1). We worked in 15 villages, separated by partly deforested argilaceous pathways with scattered houses, where mud-walled, thatch-roofed houses are usually located beneath or beside large palm trees. The surroundings consist of shady, partly deforested areas, where dogs, cats, chickens, pigs, cows, and horses live; no clear delineation separates peridomestic areas from the dense rain forest habitat of wild animals. The county's economy depends on subsistence agriculture and fishing. Raising domestic animals, producing manioc root flour and grains, and harvesting greens and fruits necessitate clearing areas of forest.
We conducted a serologic survey to assess the prevalence of T. cruzi infection in 25,451 county residents >1 and <75 years of age (<72% were 30 years of age) in these 15 villages with <1,200 houses. Fingerprick blood samples from study participants were collected onto Whatman (Clifton, NJ) 1-mm filter paper for seropositivity assessment. After air-drying at room temperature, each set of 10 blood samples was sealed in clean plastic wrap and kept dry in an ice box during the day of collection. The blood samples were then stored frozen until analysis. At the laboratory, filter paper blood samples were punched out and eluted in 100 µL of phosphate-buffered saline, pH 7.4, as described (30). The test was standardized for obtaining 5 µL of blood in 1 cm2 of the filter paper, and serum proteins were eluted in 100 µL of PBS, pH 7.4, yielded a 1:20 final dilution for screening seropositivity. For quality control, 10% of the samples were analyzed by a second examiner.
Trapping Triatomines
The strategy for trapping sylvatic triatomine bugs derived from published work (12,17), as well as observations by local residents that triatomines attracted by light fly from palm trees to houses at night. A night visit to one house resulted in capturing two triatomines on the wall near a light bulb; both these specimens had protozoan flagellates in the intestinal contents.
Researchers and field workers spoke to residents at clubs or social organizations. Dried triatomines were displayed, and community leaders requested that triatomines in houses be captured and stored (in a 5 x 3 cm translucent plastic container with holes in the cap). This strategy proved efficient for collecting triatomines in the rainy season, when the bugs invaded the houses.
We systematically dissected 23 palm trees (A. phalerata) in backyards in five villages of the county. Each tree was cut into segments: stipe, crown shaft, fronds, petiole, and leaves. Each segment was carefully searched for insects, mammals, bird nests, and animal vestiges.
During microhabitat dissection, we captured 67 nymphs and 95 adults of three species of triatomines. The precipitin test was used to type blood in the intestinal contents of 44 adult male and female triatomines. The test consisted of two-dimensional immunodiffusion of blood in the insects' intestinal fluid against taxon-specific antisera (31,32).
Trapping and Identifying Wild Animals
The rich bird fauna in the research area included Aratinga jandaia, Buzeo magnirostris, Colombina passerine, Coragyps atratus, Crotophaga ani, Guira guira, Otus choliba, Pitangus sulphuratus, Turdus fumigatus, and Tyranus muscivora. We did not capture birds because they are refractory to T. cruzi infections. However, we captured sylvatic mammals (Didelphis marsupialis) near houses in the study area. These ancient marsupials eat palm-tree fruits and rest and nest in the clefts between the stipe and their fronds. They leave these hiding places at night to search for fruits, chicken eggs, baby chicks, and food scraps. Using nylon net or wooden box-traps baited with mango and banana, we captured 12 adult D. marsupialis but were unable to trap Caluromys sp. seen in the palm trees around the study area.
Biologic Characterization of Kinetoplastid Flagellates
Parasitic protozoa in the feces of insect vectors of Chagas disease infections or in blood agar broth were demonstrated directly by light microscopy. Flagellate protozoan infections in D. marsupialis were detected by xenodiagnosis or hemoculture (33). For xenodiagnosis, 20 first-instar uninfected nymphs of Dipetalogaster maximus took a blood meal from each adult D. marsupialis captured in the field. Thirty days later, the feces of the triatomines were examined by microscopy for flagellates. Any parasitic flagellates in the feces of triatomines or in hemocultures were subjected to passage in weanling mice. This procedure consisted of intraperitoneal injection of a saline dilution of the metacyclic flagellates into mice. Two weeks later, trypomastigote forms of the parasite were identified in the blood of the mice, then 100 µL of infected blood was seeded in blood-agar slants, the supernatant of which yielded parasitic forms, which were used for mass production in nutrient-rich liver infusion tryptose medium. One isolate from R. pictipes (Rp1) and three isolates from D. marsupialis (Dm1, Dm2, and Dm3) were characterized.
Phenotypic and Genotypic Characterization of T. cruzi-like Isolates
Specific antibodies in sera from Chagas disease patients with parasitologically confirmed T. cruzi infection were used as phenotypic markers for the counterpart herein called T. cruzi-like parasitic infection. Binding of antibodies to epimastigote forms grown in liver infusion tryptose (LIT) medium and to amastigote forms in sections of murine tissues was detected by indirect immunofluorescence assay (30,34). Epimastigote and amastigote forms of the archetype Berenice stock of T. cruzi were used as positive controls. For negative controls, parasitic forms from both sources were treated with sera of T. cruzi antibody-negative persons.
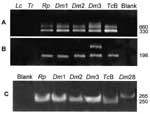
We extracted DNA of parasitic forms from each of three flagellate protozoa derived from D. marsupialis and one isolate of R. pictipes. The epimastigote forms grown in LIT were used for extraction of nuclear and kinetoplast DNA, essentially as described (35). DNA samples were analyzed by polymerase chain reaction (PCR) with specific primers for the constant regions of minicircles of kDNA and for highly repetitive sequences of nuclear DNA of T. cruzi, as described (35-37). In addition, we used the rDNA nested set of primers D71/72A, which can amplify sequences of 125 and 110 bp, respectively, from type II or I parasites (38-41). The reactions were run in parallel with 100 pg of protozoan flagellates Rp1, Dm1, Dm2, amd Dm3. As positive controls, we used 100 pg of DNA from T. cruzi Berenice (Type 1) and Dm28 (Type 2). Negative controls were 100 pg of DNA from Leishmania braziliensis (42) and T. rangeli (43,44).
Formalin-killed epimastigote forms of T. cruzi-like flagellates from Rp1, Dm1, Dm2, Dm3, and Dm4 and the Berenice stock of T. cruzi were used. The probes consisted of a nuclear DNA sequence PCR amplified with primer sets TcZ1/2 (37). The probe was labeled with biotin according to the manufacturer's protocol. Cells fixed in glass slides were hybridized with selected DNA probes and stained with fluorescein-labeled streptavidin (Sigma, St. Louis).
Each of 10 D. marsupialis trapped in the wild and BALB/c mice receiving T. cruzi-like parasites isolated from triatomines were subjected to histopathologic study. The animals were euthanized, and tissue samples from organs and tissues were fixed in 10% formalin. Three representative sections of skeletal muscles, heart, esophagus, small and large intestine, liver, kidney, spleen, and lung were stained with hematoxylin and eosin for examination by microscopy. An average of six sections from the scent glands of the marsupials was taken for histopathologic study.
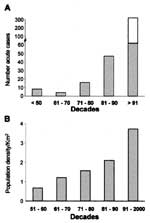
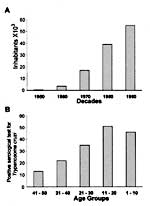
By indirect immunofluorescence test for anti-T. cruzi antibodies in human blood collected on filter paper (30), 212 (0.83%) of persons tested had specific antibodies for T. cruzi infections (Figure 3). Positive serologic results in young populations indicate recent transmission and acute infection. Forty-six children <10 years of age (0.18% of the total study population) were antibody-positive for T. cruzi and were considered acutely infected.
Our results, which show seroprevalence of T. cruzi infections in the absence of hematophagous bugs or their vestiges (excreta and molted skins) in houses, prompted us to search for triatomines in the ecosystem where the population was infected or continues to be at risk. The strategy for capture of triatomines consisted in surveillance of houses by residents (household members captured bugs in the house and placed them in plastic containers) or in dissection of palm trees in backyards of houses (16,45-49). This householder-assisted surveillance and capture method yielded 52 triatomine bugs (36 R. pictipes and 16 R. neglectus). Triatomine excreta and molted skins in these houses were neither reported by inhabitants nor detected by field workers. Adult triatomines were captured in houses only during the rainy season. We also captured 133 triatomines in 23 palm trees cut down in backyards in five villages. Careful dissection of these trees allowed detection of different developmental stages of triatomines in clefts of palm frond-sheets (Figure 4, Tables 2, 3, 4).
Remains of animal species (e.g., nests, hair, feathers) on which triatomine bugs prey were identified in palm trees in backyards in five villages. Twice when a tree was cut down, adult opossums (Didelphis, Figure 4) ran out of the fronds into the forest. Nests of marsupials and birds were easily detected on dissection of palm fronds and crowns. In addition to opossums and birds, we identified molds and captured and identified different species of various taxa of invertebrate and vertebrate animals (50-60) in the 23 palm trees (Table 5, Figure 5). Molds were found in stipes, fronds, and crowns, and insects in roots, stipes, inflorescence, fruits, fronds, crowns, and leaves. The clefts formed by frond sheets were particularly rich in Amphibia, Arachnida, and Hemiptera. Triatomines were detected at the bottom of clefts where marsupials built their nests. Bird nests were found in the fronds and crowns where abundant species of insects were available for predation.
Scarcity of blood flagellates in marsupials precluded detection by direct microscopy. However, the metacyclic flagellates recovered by xenodiagnosis were subinoculated in weanling mice. Two weeks after injection, trypomastigote forms of the parasitic protozoan morphologically indistinguishable from T. cruzi were detected in blood of the mice. To define and further characterize these isolates, we used phenotypic and genotypic molecular characterizations. In the first group, antibodies in sera of chronic Chagas disease patients reacted indistinctly with antigenic determinants in the surface of T. cruzi Berenice and with isolates Dm1, Dm2, and Dm3 from D. marsupialis and with Rp1 from R. pictipes.
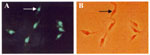
Genotypic kDNA and nuclear DNA (nDNA) markers were used to genetically characterize these wild flagellate protozoan isolates. PCR amplification of template DNA from each of these T. cruzi isolates showed that kDNA primers S35/36 (35-37) amplified cruzi, a laboratory standard for virulent T. cruzi. In addition, when we used PCR with mini-exon intergenic spacer primers TC/TC1/TC2 (38) and rDNA primers D71/72 (39-41), amplification resulted in the same bands, using template DNA from wild T. cruzi Dm 28c, Dm1, Dm2, Dm3, and Rp1 and from Berenice T. cruzi isolated from a patient with acute Chagas disease (Figure 6). These molecular features allowed classification of wild isolates of T. cruzi as phylogenetic type I, while Berenice was T. cruzi II (11,38-41). These results were further confirmed by in situ hybridization of wild T. cruzi with a biotinilated 198-bp sequence derived from Berenice template DNA (Figure 7), which was amplified with specific nDNA primers Tcz1/2 (37).
Several authors (7,8,17-19,12) described T. cruzi-like flagellate protozoans (instead of T. cruzi) in the blood of various classes of mammals and in the feces of triatomine species from the Amazon Basin. We detected blood flagellates in 9 of 10 Didelphidae captured in backyards of houses in Paço do Lumiar county. In addition, we isolated flagellates from feces of R. pictipes, which showed the same morphologic features described for T. cruzi. In biological characterizations, these flagellates induced low-level parasitemia in laboratory mice. However, histopathologic lesions in marsupials and laboratory mice were similar to those described in Chagas disease patients. Furthermore, nuclear DNA markers displayed all features of the T. cruzi standards Berenice and Dm 28c T. cruzi, which differed from those shown by T. rangeli and L. braziliensis. These data confirm the parasitic flagellates present in triatomines and mammals in this ecoregion as T. cruzi.
Birds are refractory to T. cruzi infections (33), but several authors (54-60) describe marsupials as important wildlife reservoirs of this parasitic protozoon. We used traps with fruit baits to capture 12 marsupials, which underwent karyotyping and parasitologic and pathologic examinations. The karyotyping confirmed these mammals as Didelphis marsupialis. Nine of 10 trapped marsupials had protozoan blood flagellates isolated by xenodiagnosis or hemoculture. Histopathologic study of heart sections from these infected animals showed typical myocarditis, characterized by mononuclear cell infiltrates and target cell lysis. Inflammatory infiltrates were seen in skeletal muscles, esophagus, and small and large intestines. Histopathologic study of representative tissue sections taken from the only opossum that did not have the parasite detected showed no histopathologic alterations (data not shown).
American trypanosomiasis has been considered an ancient zoonosis in which insect vectors and mammal hosts sympatrically occupy vast areas of South America (33). Wild T. cruzi infections and the bug vectors are syntopically adapted to mammalian host habitats under natural equilibrium. T. cruzi infections and human Chagas disease occur over a large geographic area, limited by parallels 42° N to 42°S (33). The Tocantins moist forest, much of which is subject to severe disturbance of the environmental equilibrium, provided conditions for disease outbreaks (61-64). The growing human populations encroaching on that natural ecoregion are fed upon by the triatomine vector R. pictipes, and cases of acute T. cruzi infections in humans have exponentially increased in the last three decades.
In this study, we describe a trophic network of five levels comprising different species dwelling in palm tree microhabitats. A single class of top predator mammal (Didelphidae) was found in the study area. The absence of other taxa of top wild predators upon which bugs feed may contribute to peridomiciliar and domiciliar invasion during the wet season. This observation contrasts with earlier descriptions of seven families of mammals, belonging to Primates, Edentates, Marsupials, Carnivores, Rodents, and Chiroptera classes (10,55,57), which were hosts for triatomines in relatively undisturbed ecoregions. Elimination of a single class of invertebrate or vertebrate animals in a trophic network may be a major risk factor leading to more triatomine species entering houses and initiating a new cycle of transmission of T. cruzi infection.
In our study, a child <10 years of age with a positive immunofluorescence test (see methods) was considered a host of acute T. cruzi infection. Considering the age-specific prevalence of T. cruzi infections in adults (30) and the fact that for each acute case that is clinically identified an estimated 20 to 100 others are unrecognized (34), autochthonous human Chagas disease in the Amazon Basin may reach 7,860 to 39,300 cases. The latter figure is consistent with serologic evidence of T. cruzi infection in the Brazilian Amazon region presented in the national report on Chagas disease (65). The characteristics of transmission of infections described here do not indicate a need for insecticide spraying in the Amazon region, for the cycle of transmission of T. cruzi is deeply embedded in a natural trophic network comprising wild animals belonging to several classes and trophic levels.
Risk factors associated with the possibility of emergence of endemic Chagas disease in the Amazon Basin have been described (10,12,17,19,25,54,55,62-64). First, the broadleaf moist rain forest ecosystem may be invaded by triatomine species (T. infestans and T. rubrofasciata), which are considered completely adapted to human domiciles, or by other triatomines (P. megistus and T. brasiliensis, T. pseudomaculata, and T. sordida), which can be found in different ecosystems but frequently enter and colonize houses (64). Second, several Amazon Basin triatomine species (R. pictipes, R. prolixus, R. neglectus, R. nausutus, T. vitticeps, T. rubrovaria and others) can adapt to human dwellings, where they could become important vectors of the T. cruzi infections (63-69). We found no vestiges of triatomine colonization in houses or their surroundings in our study area. We hypothesize that starving adult R. pictipes and R. neglectus may leave their natural shelters at night to feed on human hosts, probably attracted by light in the houses.
Factors associated with triatomines flying from palm tree to houses need to be clarified. The scarcity of birds and mammals during the wet season may be an important factor associated with anthropic predation and the presence of T. cruzi-infected insects in houses in the rainy season. Domiciliation of triatomines may not be required for an increasing endemicity of Chagas disease in the Amazon Basin (13,14,19). For example, sylvatic Rhodnius brethesi recently bit people harvesting palm fibers in Barcelos in the northwestern part of the State of Amazonas, 490 km upriver from Manaus, leading to an acute case of Chagas disease (13). Blood samples from residents of this locality tested by immunofluorescence assay had 12.5% positivity for anti-T. cruzi antibodies (70). These observations suggest that to prevent transmission of T. cruzi infections to humans in the Amazon Basin, new strategies are needed, which will not necessarily be similar to those used in controlling endemic Chagas disease in other ecosystems, such as the Cerrado and Caatinga ecosystems in Brazil (1,63,66,67,68,69).
Alternatively, the enormous task of controlling emerging Chagas disease in the Amazon Basin should rely initially on an information, education, and communication program, which encourages control measures by the householder (e.g., use of screens, bed nets, insecticide-treated fabrics, and vegetation management) (69-73). Such a program for prevention of contact with triatomines should be conducted directly in communities, elementary schools, and churches and social clubs, reinforced by social marketing and mass media communications. Further studies are also needed for identifying new and integrated (chemical and nonchemical) strategies required for controlling T. cruzi vectors in the Amazon Basin, which may not necessarily be similar to those already shown to be partially effective in controlling the domestic vectors of endemic Chagas disease in other ecosystems in the Americas.
Dr. Teixeira is professor of pathology at the medical school of the University of Brasilia. His research interests focus on endemic infections by kinetoplastic flagellates and the pathogeneses of Chagas disease and cutaneous and visceral leishmaniasis.
Acknowledgment
We thank the National Laboratory for Taxonomy of Triatomines, Fundação Instituto Oswaldo Cruz, Rio de Janeiro, for performing the precipitin test.
References
- Dinerstein E, Olson DM, Graham DJ, Webster AL, Primm SA, Bookbinder MP, A conservation assessment of the terrestrial ecoregions of Latin America and the Caribbean. Ed. Washington: The World Wildlife Fund/The World Bank; 1995.
- La Foret en Jeu. L' Extractivisme en Amazonie Centrale. In: Emperaire L, editor. L' Orston: Institut Français de Recherche Scientifique pour le Développement en Coopéracion; Paris: Unesco; 1996. p. 116-59.
- Pindorama. São Paulo: Mercedes-Benz do Brazil; 1993.
- Kahn F, Corradini L. Palmeiras da Amazonia. Ed. Cendotec. Brasilia: Centro Franco Brasileiro de Documentação Científica; 1994. Contato, special volume. p. 11-6.
- Mapeamento e levantamento do potencial das ocorrências de babaçuais. Brasília: Núcleo de Comunicação Social da Secretaria de Tecnologia Industrial, Ministério da Indústria e do Comércio; 1982. p. 11-62.
- Moomen H. Emerging infectious diseases--Brazil. Emerg Infect Dis. 1998;4:351–4. DOIPubMedGoogle Scholar
- Brasil RP, Silva AR, Arbarelli A, Vale JF. Distribuição e infecção de triatomineos por Trypanosoma tipo cruzi na Ilha de São Luis, Maranhão. Rev Soc Bras Med Trop. 1985;18:257–60.
- Miles MA. Souza de AA, Póvoa M. Chagas diseases in the Amazon Basin. III. Ecotopes of ten triatomine bug species (Hemiptera: Reduvidae) from the vicinity of Belém, Pará State, Brazil. J Med Entomol. 1981;18:266–78.PubMedGoogle Scholar
- Miles MA, Arias JR, Souza AA. Chagas disease in the Amazon Basin. V. Periurban palms as habitats of Rhodnius robustus and Rhodnius pictipes triatominae vectors of Chagas disease. Mem Inst Oswaldo Cruz. 1983;78:391–8. DOIPubMedGoogle Scholar
- Lainson R, Shaw JJ, Fraiha H, Miles MA, Draper CC. Chagas disease in the Amazon Basin: Trypanosoma cruzi infections in sylvatic mammals, triatomine bugs and man in the State of Pará, north Brazil. Trans R Soc Trop Med Hyg. 1979;73:193–204. DOIPubMedGoogle Scholar
- Recommendations from a satellite meeting. International Symposium to commemorate the 90th anniversary of the discovery of Chagas disease. Mem Inst Oswaldo Cruz. 1999;94:429–32. DOIPubMedGoogle Scholar
- D'Alessandro A, Barreto P, Saravia N, Barreto M. Epidemiology of Trypanosoma cruzi in the oriental plains of Colombia. Am J Trop Med Hyg. 1984;33:1084–95.PubMedGoogle Scholar
- Coura JR, Barret TV, Naranjo CM. Ataque de populações humanas por triatomineos silvestres no Amazonas: uma nova forma de transmissão da infecção Chagásica? Rev Soc Bras Med Trop. 1994;27:251–3.PubMedGoogle Scholar
- Valente VC, Valente SA, Noireau F, Carrasco HJ, Miles MA. Chagas disease in the Amazon Basin: association of Panstrongylus geniculatus (Hemiptera: Reduviidae) with domestic pigs. J Med Entomol. 1998;35:99–103.PubMedGoogle Scholar
- Amunárriz MU, Chico ME, Guderian RH. Chagas disease in Ecuador: a sylvatic focus in the Amazon Region. J Trop Med Hyg. 1991;94:145–9.PubMedGoogle Scholar
- Whitlaw JT, Chaniotis BN. Palm trees and Chagas' in Panama. Am J Trop Med Hyg. 1978;27:873–81.PubMedGoogle Scholar
- Almeida FB. Triatomíneos da Amazonia. Encontro de três espécies naturalmente infectadas por trypanosoma semelhante ao cruzi no Estado do Amazonas (Hemiptera, Reduviidae). Acta Amáz (Manaus). 1971;1:71–5.
- Coura JR. Doença de Chagas como endemia na amazonia brasileira: risco ou hipótese? Rev Soc Bras Med Trop. 1990;23:67–70.PubMedGoogle Scholar
- Fraiha Neto H, Valente SA, Valente VC, Pinto AYN. Doença de Chagas endemica na Amazonia. Rev Acad Med Pará, Belém. 1995;6:1–132.
- Chagas C. Infection naturelle des singes du Pará (Crysotrix sciureus) par Trypanosoma cruzi. C R Soc Biol (Paris). 1924;9:873–6.
- Raccurt CP. Trypanosoma cruzi in French Guinea: review of accumulated data since 1940. Med Trop. 1996;56:79–87.PubMedGoogle Scholar
- Shaw JJ, Lainson R, Fraiha H. Considerações sobre a epidemiologia dos primeiros casos autóctones de doença de Chagas registrados em Belém, Pará, Brasil. Rev Bras Saúde Pública, São Paulo. 1969;3:153–7.
- Ferraroni JJ, Melo JAN, Camargo ME. Molestia de Chagas na Amazônia. Ocorrência de seis casos suspeitos, autóctones, sorologicamente positivos. Acta Amazon. 1977;3:438–9.
- Silva AR, Mendes JR, Mendonca ML, Cutrin RN, Brazil RP. Primeiros casos agudos autoctones da doença de Chagas no Maranhão e inquérito soro-epidemiologico da população. Rev Soc Bras Med Trop. 1985;18:269–70.
- Rodrigues CI, Souza AA, Terceros R, Valente S. Doença de Chagas na Amazonia: I Registro de oito casos autóctones em Macapa. Rev Soc Bras Med Trop. 1988;21:193–7.PubMedGoogle Scholar
- Acosta HMS, Ferreira CS, Carvalho ME. Human infection with Trypanosoma cruzi in Nasca, Peru: a seroepidemiologic survey. Rev Inst Med Trop Sao Paulo. 1997;39:107–12. DOIPubMedGoogle Scholar
- Chico MH, Sandoval C, Guevara AE, Calvopiña MH, Cooper PJ, Reed SG, Chagas disease in Ecuador: evidence for disease transmission in an indigenous population in the Amazon Region. Mem Inst Oswaldo Cruz. 1997;92:317–20. DOIPubMedGoogle Scholar
- Corredor-Arjona A, Moreno CA, Agudelo CA, Bueno M, López MC, Cáceres E, Prevalence of Trypanosoma cruzi and Leishmania chagasi infection and risk factors in a Colombian indigenous population. Rev Inst Med Trop Sao Paulo. 1999;41:229–4. DOIPubMedGoogle Scholar
- Spellerberg IF, Hardes S. Biological conservation. Cambridge: Cambridge University Press, 1992.
- Williams-Blangero S, VandeBerg J, Blangero J, Teixeira ARL. Genetic epidemiology of seropositivity for Trypanosoma cruzi infection in rural Goias, Brazil. Am J Trop Med Hyg. 1997;57:538–43.PubMedGoogle Scholar
- Pedreira de Freitas JL, Siqueira AF, Ferreira OA. Investigações epidemiológicas sobre triatomíneos de hábitos domésticos e silvestres com auxílio da reação de precipitina. Rev Inst Med Trop Sao Paulo. 1960;2:90–9.
- Siqueira AF. Estudos sobre a reação da precipitina aplicada à identificação de sangue por triatomineos. Rev Inst Med Trop Sao Paulo. 1960;2:41–53.
- Teixeira ARL. The Stercorarian trypanosomes. In: Immune responses in parasitic infections: immunology, immunopathology and immunoprophylaxis. EJL Soulsby, editor. Boca Raton (FL): CRC Press, 1987. p. 125-45.
- Teixeira ARL, Teixeira G, Macedo V, Prata A. Acquired cell-mediated immunodepression in acute Chagas disease. J Clin Invest. 1978;62:1132–8. DOIPubMedGoogle Scholar
- Sturm NR, Degrave W, Morel CM, Simpson L. Sensitive detection and schizodeme classification of Trypanosoma cruzi by amplification of kinetoplast minicircle DNA sequences: use in the diagnosis of Chagas disease. Mol Biochem Parasitol. 1984;33:205–14. DOIPubMedGoogle Scholar
- Avila HA, Sigman DS, Coohen LM, Millikan RC, Simpson L. Polymerase chain reaction amplification of Trypanosoma cruzi kinetoplast minicircle DNA isolated from whole blood lysates: diagnosis of chronic Chagas disease. Mol Biochem Parasitol. 1991;48:211–22. DOIPubMedGoogle Scholar
- Moser DR, Kirchhoff LV, Donelson J. Detection of Trypansoma cruzi by DNA amplification using the polymerase chain reaction. J Clin Microbiol. 1989;27:1477–82.PubMedGoogle Scholar
- Fernandes O, Sturm NR, Campbell D. The mini-exon gene: a genetic marker for zymodeme III of Trypanosoma cruzi. Mol Biochem Parasitol. 1998;95:129–33. DOIPubMedGoogle Scholar
- Briones MR, Souto RP, Stolf BS, Zingales B. The evolution of two Trypanosoma cruzi subgroups inferred from rRNA genes can be correlated with the interchange of American mammalian faunas in the Cenozoic and has implications to pathogenicity and host specificity. Mol Biochem Parasitol. 1999;104:219–32. DOIPubMedGoogle Scholar
- Souto RP, Zingales B. Sensitive detection and strain classification of Trypanosoma cruzi by amplification of a ribosomal RNA sequence. Mol Biochem Parasitol. 1993;63:45–52. DOIPubMedGoogle Scholar
- Souto RP, Vargas N, Zingales B. Trypanosoma rangeli: discrimination from Trypanosoma cruzi based on a variable domain from the large subunit ribosomal RNA gene. Exp Parasitol. 1999;91:306–14. DOIPubMedGoogle Scholar
- Jorg ME. La modificación del biotopo peri-habitacional en la profilaxis de la enfermedad de Chagas. Rev Soc Bras Med Trop. 1989;22:91–5.PubMedGoogle Scholar
- Vexenat AC, Santana JM, Teixeira ARL. Cross-reactivity of antibodies in human infections by the kinetoplastid protozoa Trypanosoma cruzi, Leishmania chagasi and Leishmania (viannia) braziliensis. Rev Inst Med Trop Sao Paulo. 1996;38:177–85. DOIPubMedGoogle Scholar
- Guhl F, Hudson L, Marinkelle CJ, Jaramillo CA, Bridge C. Clinical Trypanosoma rangeli infection as a complication of Chagas disease. Parasitology. 1987;94:475–84. DOIPubMedGoogle Scholar
- Romaña CA, Pizarro JC, Rodas E, Guilbert E. Palm trees as ecological indicators of risk areas for Chagas disease. Trans R Soc Trop Med Hyg. 1999;93:594–5. DOIPubMedGoogle Scholar
- Barreto MP, Siqueira AF, Correa FMA, Ferrioli F, Cavalheiro JR. Estudos sobre reservatórios e vetores silvestres do Trypanosoma cruzi. VII: investigacões sobre a infecção natural de gambás por Tripanossomos semelhantes ao "T. cruzi.". Rev Bras Biol. 1964;24:289–300.PubMedGoogle Scholar
- Barreto MP, Siqueira AF. Filho, Cavalheiro JR. Estudos sobre reservatórios e vetores silvestres do Trypanosoma cruzi. XI: Observações sobre um foco da tripanosomose Americana no Município de Ribeirão Preto, São Paulo. Rev Inst Med Trop Sao Paulo. 1966;8:103–12.PubMedGoogle Scholar
- Barreto MP, Alburquerque RDR, Funayama GK. Estudos sobre reservatórios e vetores silvestres do Trypanosoma cruzi. XXXVI: investigações sobre triatomineos de palmeiras no Municipio De Uberaba, MG, Brasil. Rev Soc Bras Biol. 1969;29:577–88.
- Barreto MP, Ribeiro RD, Rocha GM. Estudos Sobre reservatórios e vetores silvestres do Trypanosoma cruzi. LXIX: Inquérito preliminar sobre triatomíneos silvestres na região do triângulo Mineiro MG, Brasil. Rev Bras Biol. 1978;38:633–7.PubMedGoogle Scholar
- Hawksworth DL, Kirk PM, Sutton DN. Dictionary of the fungi. 8th edition. International Mycological Institute. Cambridge: University Press; 1995. p. 76, 272.
- Fungos em plantas no Brasil. Empresa Brasileira de Pesquisa Agropecuária, Centro Nacional de Pesquisa de Recursos Genéticos e Biotecnologia, Ministério da Agricultura e do Abastecimento. Brasília: Serviço de Produção de Informação; 1988. p. 171.
- Edwards PJ, Wratten SD. Ecologia das interações entre insetos e plantas. Vol. 27.São Paulo: Editora Pedagógica e Universitária Ltda; 1981.
- Kempf WW. Levantamento das formigas da mata amazônica, nos arredores de Belém do Pará, Brasil. Studia entomológica. Rio de Janeiro: Vozes Ltda; 1970. p. 321-43.
- Naiff MF, Naiff RD, Barret TV. Vetores selváticos de doença de Chagas na área urbana de Manaus (AM): atividade de vôo nas estações secas e chuvosas. Rev Soc Bras Med Trop. 1998;31:103–5. DOIPubMedGoogle Scholar
- Miles MA, Arias JR, Naiff RD, Souza AA, Povoa MM, Lima JAN, Vertebrate hosts and vectors of Trypanosoma rangeli in the Amazon Basin of Brazil. Am J Trop Med Hyg. 1983;32:1251–9.PubMedGoogle Scholar
- Helmut S. Birds in Brazil. A natural history. Princeton (NJ): Princeton University Press;1993.
- Deane L. Animal reservoirs of Trypanosoma cruzi in Brazil. Rev Bras Malariol Doencas Trop. 1964;16:28–48.PubMedGoogle Scholar
- Deane MP, Jansen AM. Another Trypanosoma, distinct fron T. cruzi, multiples in the lumen of the anal glands of the opossum Didelphis marsupialis. Mem Inst Oswaldo Cruz. 1986;81:131–2. DOIPubMedGoogle Scholar
- Jansen AM, Madeira F, Carreira JC. Acosta-Medina, Deane MP. Trypanosoma cruzi in the opossum Didelphis marsupialis: a study of the correlations and kinetics of the systemic and scent gland infections in naturally and experimentally infected animals. Exp Parasitol. 1997;86:37–44. DOIPubMedGoogle Scholar
- Odun EP. Ecologia. Biblioteca Pioneira de Biologia Moderna. São Paulo: Livraria Pioneira; 1977. p. 19-148.
- Morse SS. Factors in the emergence of infectious diseases. Emerg Infect Dis. 1995;1:7–15. DOIPubMedGoogle Scholar
- Valente AS, Valente VC, Fraiha Neto A. Considerations on the epidemiology and transmission of Chagas disease in the Brazilian Amazon. Mem Inst Oswaldo Cruz. 1999;94:395–8. DOIPubMedGoogle Scholar
- Silveira AC, Rezende DF. Epidemiologia e controle da transmissão vetorial da doença de Chagas no Brasil. Rev Soc Bras Med Trop 1994;27(Supl III):11-22.
- Silveira AC, Vinhaes MC. Elimination of vector-borne transmission of Chagas disease. Mem Inst Oswaldo Cruz. 1999;94:405–11. DOIPubMedGoogle Scholar
- Camargo ME, Silva GR, Castilho EA, Silveira AC. Inquérito sorológico da prevalência de infecção chagasica no Brasil, 1985-1980. Rev Inst Med Trop Sao Paulo. 1984;26:192–204.PubMedGoogle Scholar
- Dias JCP. A doença de Chagas e seu controle na América Latina. Uma análise de possibilidades. Rio de Janeiro. Cad Saude Publica. 1993;9:201–9. DOIPubMedGoogle Scholar
- Moncayo A. Chagas disease: epidemiology and prospects for interruption of transmission in the Americas. Wld Hlth Statist Quart. 1992;45:276–9.PubMedGoogle Scholar
- Valente VC. Potential for domestication of Panstrongylus geniculatus in the municipality of Muaná, Marajó Island, State of Pará, Brazil. Mem Inst Oswaldo Cruz. 1999;94:399–400. DOIPubMedGoogle Scholar
- Feitosa VR. Implantação de um sistema de vigilância epidemológica (VE) de doença de Chagas na Amazônia. Ver Soc Brás Méd Trop. 1995;28:84–7.
- Coura JR, Junqueira ACV, Boia MN, Fernandes O. Chagas disease: from bush to huts and houses. Is it the case of the Brazilian Amazon? Mem Inst Oswaldo Cruz. 1999;94:379–84. DOIPubMedGoogle Scholar
- Bos R. The importance of peridomestic environmental management for the control of the vectors of Chagas disease. Rev Argent Microbiol. 1988;20:58–62.PubMedGoogle Scholar
- Kroeger A, Ordonez-Gonzalez J, Behrend M, Alvarez G. Bednet impregnation for Chagas disease control: a new perspective. Trop Med Int Health. 1999;4:194–8. DOIPubMedGoogle Scholar
- Wood E, de Licastro SA, Casabe N, Picollo MI, Alzogaray R, Nicolas Zerba E. A new tactic for Triatoma infestans control: fabrics impregnated with beta-cypermethrin. Rev Panam Salud Publica. 1999;6:1–7. DOIPubMedGoogle Scholar
Figures
Tables
Cite This Article1In this study, a major ecosystem is defined as a set of ecoregions of comparable dynamics, response characteristics to disturbance, species diversity, and conservation needs. An ecoregion is a geographically distinct set of natural communities with similar species, ecologic dynamics, environmental conditions, and ecologic interactions critical for long-term persistence (1).
Table of Contents – Volume 7, Number 1—February 2001
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Antonio R.L. Teixeira, Chagas' Disease Multidisciplinary Research Laboratory, Faculty of Medicine, University of Brasília. P.O. Box 04536. Postcode 70.919.970, Brasília, DF, Brazil; fax: 55+61 273-4645
Top