Volume 7, Number 6—December 2001
Research
Bioterrorism-Related Inhalational Anthrax: The First 10 Cases Reported in the United States
Cite This Article
Citation for Media
Abstract
From October 4 to November 2, 2001, the first 10 confirmed cases of inhalational anthrax caused by intentional release of Bacillus anthracis were identified in the United States. Epidemiologic investigation indicated that the outbreak, in the District of Columbia, Florida, New Jersey, and New York, resulted from intentional delivery of B. anthracis spores through mailed letters or packages. We describe the clinical presentation and course of these cases of bioterrorism-related inhalational anthrax. The median age of patients was 56 years (range 43 to 73 years), 70% were male, and except for one, all were known or believed to have processed, handled, or received letters containing B. anthracis spores. The median incubation period from the time of exposure to onset of symptoms, when known (n=6), was 4 days (range 4 to 6 days). Symptoms at initial presentation included fever or chills (n=10), sweats (n=7), fatigue or malaise (n=10), minimal or nonproductive cough (n=9), dyspnea (n=8), and nausea or vomiting (n=9). The median white blood cell count was 9.8 X 103 /mm3 (range 7.5 to 13.3), often with increased neutrophils and band forms. Nine patients had elevated serum transaminase levels, and six were hypoxic. All 10 patients had abnormal chest X-rays; abnormalities included infiltrates (n=7), pleural effusion (n=8), and mediastinal widening (seven patients). Computed tomography of the chest was performed on eight patients, and mediastinal lymphadenopathy was present in seven. With multidrug antibiotic regimens and supportive care, survival of patients (60%) was markedly higher (<15%) than previously reported.
Historically, human anthrax in its various forms has been a disease of those with close contact to animals or animal products contaminated with Bacillus anthracis spores. In the mid-1800s, inhalational anthrax related to the textile industry became known as woolsorters' disease (in England) (1) and ragpickers' disease (in Germany and Austria) because of the frequency of infection in mill workers exposed to imported animal fibers contaminated with B. anthracis spores. In the early 1900s, human cases of inhalational anthrax occurred in the United States in conjunction with the textile and tanning industries. In the last part of the 20th century, with improved industrial hygiene practices and restrictions on imported animal products, the number of cases fell dramatically (1,2); however, death rates remained high (>85%) (1,3). In 1979, in Sverdlovsk, former Soviet Union, an apparent aerosol release of B. anthracis spores from a military facility resulted in the largest outbreak of inhalational anthrax in the 20th century (3).
Before October 2001, the last case of inhalational anthrax in the United States had occurred in 1976 (1,4). Identification of inhalational anthrax in a journalist in Florida on October 4, 2001, marked the beginning of the first confirmed outbreak associated with intentional anthrax release in the United States (5,6). We describe the clinical presentation of the first 10 cases of this bioterrorism-related outbreak.
From October 4 to November 2, 2001, the Centers for Disease Control and Prevention (CDC) and state and local public health authorities reported 10 confirmed cases of inhalational anthrax and 12 confirmed or suspected cases of cutaneous anthrax in persons who worked in the District of Columbia, Florida, New Jersey, and New York (6,7). Epidemiologic investigation indicated that the outbreak resulted from intentional delivery of B. anthracis spores through mailed letters or packages.
Of the 10 inhalational cases, 7 occurred in postal employees in New Jersey and the District of Columbia who were likely exposed to letters known to be contaminated with B. anthracis spores. Two cases were in employees of a media company in Florida: one is believed to have received contaminated mail, the other to have sorted and distributed that mail. Case 10 was in a resident of New York, and the nature of her exposure to B. anthracis is currently unknown.
In the course of the US Public Health Service response to the outbreak, CDC, in conjunction with state and local health departments, collected clinical data from patients with confirmed inhalational anthrax as defined by the CDC surveillance case definition (8). Data were collected through a variety of methods, including interviews with patients and family members, physical examinations of patients, discussions with clinicians, reviews of medical records, and analyses of clinical and pathologic specimens.
Presumptive isolation of B. anthracis in these patients was confirmed by gamma phage lysis, presence of a capsule, detection of capsule and cell-wall antigens by direct fluorescent antibody, and B. anthracis-specific polymerase chain reaction (PCR). All isolates were confirmed by state and other laboratory facilities of the National Bioterrorism Laboratory Response Network and by the laboratories of the National Center for Infectious Diseases (NCID), CDC. Other tests performed at NCID for confirming the diagnosis of inhalational anthrax (8) included immunohistochemical testing of clinical specimens by using B. anthracis capsule and cell-wall antibody, B. anthracis-specific PCR, and serologic detection of immunoglobulin (Ig) G to B. anthracis protective antigen (PA).
Case 1
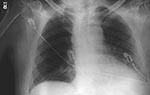
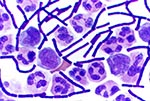
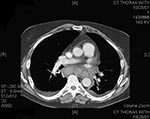
On October 2, 2001, a 63-year-old Caucasian photo editor working for a Florida newspaper awoke early with nausea, vomiting, and confusion and was taken to a local emergency room for evaluation. His illness, which started on September 27 during a trip to North Carolina, was characterized by malaise, fatigue, fever, chills, anorexia, and sweats. No history of headache, cough, chest pain, myalgias, dyspnea, abdominal pain, diarrhea, or skin lesions was reported. Past medical history included hypertension, cardiovascular disease, and gout. He did not smoke. On admission, the patient was alert and interactive but spoke nonsensically. Temperature was 39.2°C, and heart rate 109/min; blood pressure and respiratory rate were normal. Initial pulmonary, heart, and abdominal examinations were reported as normal. No nuchal rigidity was observed. He was not oriented to person, place, or time. Admission laboratory values included a normal total white blood cell (WBC) count, but the platelet count was low. Serum chemistries were normal, except for borderline hyponatremia and elevated total bilirubin. He had mild metabolic acidosis (Table 1). A chest X-ray showed a prominent superior mediastinum and a possible small left pleural effusion (Figure 1). Cerebrospinal fluid (CSF) analysis showed WBC count 4,750/µL (81% neutrophils), red blood cell count 1,375/µL, glucose 57 mg/dL (serum glucose 174 mg/dL), and protein 666 mg/dL. Microscopy examination of the CSF showed many gram-positive bacilli (Figure 2). B. anthracis was isolated from CSF after 7 hours of incubation and from blood cultures within 24 hours of incubation. The patient was admitted to the hospital with a diagnosis of meningitis. After a single dose of cefotaxime, he was started on multiple antibiotics, including ceftazidime, gentamicin, metronidazole, doxycycline, ampicillin, and trimethoprim-sulfamethoxazole. Shortly after admission, he had generalized seizures and was intubated for airway protection. On hospital day 2, penicillin G, levofloxacin, and clindamycin were begun; ampicillin, ceftazidime, and trimethoprim-sulfamethoxazole were discontinued. He remained febrile and became unresponsive to deep stimuli. His condition progressively deteriorated, with hypotension and worsening renal insufficiency. The patient died on October 5. Autopsy findings included hemorrhagic mediastinal lymphadenitis, and immunohistochemical staining showed disseminated B. anthracis in multiple organs.
Case 2
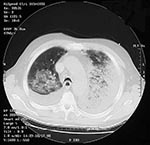
On September 24, a 73-year-old Hispanic man, the newspaper mailroom clerk who delivered mail to the patient in Case 1, had onset of fatigue. On September 28, nonproductive cough, intermittent fever, rhinorrhea, and conjunctivitis developed. From September 28 to October 1, he had gradual progression of cough, marked worsening of fatigue with lethargy, onset of exertional dyspnea, fever, and sweats. He had mild abdominal pain associated with vomiting, and his co-workers and family noted intermittent periods of confusion. He had no underlying chronic illnesses, with the exception of a transient ischemic attack in August 2001. He did not smoke. He was admitted to the hospital on October 1. Temperature was 38.5°C, heart rate 109/min, respiratory rate 20/min, and blood pressure 108/61 mm Hg. He had bilateral conjunctival injection and bilateral pulmonary rhonchi. Examination, including assessment of neurologic function, was otherwise unremarkable. No skin lesions were observed. Admission laboratory results included normal WBC count and serum chemistries, except for hypoalbuminemia, elevated hepatic transaminases, borderline hyponatremia, and increased creatinine. Arterial blood gas values showed hypoxia (Table 1). Blood cultures obtained on hospital day 2, after initiation of antibiotics, showed no growth. A chest X-ray showed left upper and lower lobe infiltrates consistent with pneumonia and a small left pleural effusion (Figure 3). No mediastinal widening was observed. The patient was initially given intravenous azithromycin; cefotaxime and ciprofloxacin were subsequently added. A nasal swab obtained on October 5 grew B. anthracis. Computed tomography (CT) of the chest showed bilateral effusions and multilobar pulmonary consolidation but no significant mediastinal lymphadenopathy (Figure 4). A left thoracentesis yielded serosanguinous fluid (Table 1) positive for B. anthracis DNA by PCR. Bronchoscopy showed bloody secretions in the right lower lobe and left lung, with severe mucosal hyperemia, mottling, and inflammation. Bacterial cultures of bronchial washings and pleural fluid did not grow. A transbronchial biopsy showed B. anthracis capsule and cell-wall antigens by immunohistochemical staining. Tests for Legionella spp., acid-fast bacteria, Pneumocystis carinii, Chlamydia spp., Leptospira, and Hantavirus and other viral pathogens were negative. The hospital course included an episode of supraventricular tachycardia with hypotension, maximum WBC count 26,800/ mm3, and recurrent left pleural effusion that required repeat thoracentesis and placement of a chest tube. The pleural fluid from the second thoracentesis was positive for B. anthracis DNA by PCR. A pleural fluid cytology preparation and pleural biopsy showed B. anthracis capsule and cell-wall antigens by immunohistochemical staining. In addition, serial serum samples demonstrated a >4-fold rise in levels of serum antibody (IgG) to the PA component of the anthrax toxins by enzyme-linked immunosorbent assay (ELISA). The patient's condition gradually improved, and he was discharged from the hospital October 23 on oral ciprofloxacin.
Case 3
On October 16, a 56-year-old African-American man, a US Postal Service mail sorter, noted low-grade fever, chills, sore throat, headache, and malaise. This was followed by minimal dry cough, chest heaviness, shortness of breath, night sweats, nausea, and vomiting. On October 19, when he arrived at a local hospital, he was afebrile and normotensive. Heart rate was 110/min, and he was not tachypneic. He was in no acute distress but had decreased breath sounds and rhonchi at the left base. No skin lesions were observed. His past medical history was unremarkable, and he did not smoke. Total WBC count was normal, but there was a slight left shift in the differential. Hepatic transaminase levels and bilirubin were elevated. Serum albumin was decreased, but serum chemistries and renal function were normal. Arterial blood gas values showed adequate oxygenation (Table 1). A chest X-ray showed a widened mediastinum (especially in the right paratracheal region), bilateral hilar masses, bilateral pleural effusions, and a small right lower lobe air space opacity. CT of the chest showed diffuse mediastinal edema; marked paratracheal, subcarinal, hilar, and azygo-esophageal recess adenopathy (the largest node measuring 4.2 cm in diameter); and bilateral moderate pleural effusions. Within 11 hours, admission blood cultures grew B. anthracis. Ciprofloxacin, rifampin, and clindamycin were initiated. On October 21, the patient had respiratory distress, which was treated with diuretics, systemic corticosteroids, and a therapeutic thoracentesis. During the course of hospitalization, the pleural effusion reaccumulated, necessitating two additional thoracenteses. All three pleural fluid specimens were hemorrhagic. The patient did not require a chest tube. Hematemesis developed, and several shallow gastric ulcers were noted on upper endoscopy. On October 28, the patient had signs of hemolytic anemia and thrombocytopenia, which was treated with plasmapheresis. Hematologic values subsequently improved, and the patient remains hospitalized in stable condition.
Case 4
On October 16, a 56-year-old African-American man, a US Postal Service worker, noted mild headache that was constant and global but not associated with visual changes, stiff neck, or other neurologic symptoms. Over the following 3 days, the headache worsened and was accompanied by low-grade fever, chills, sore throat, myalgias, nausea, malaise, drenching sweats, intermittent blurred vision, and photophobia. A mild dry cough, dyspnea on exertion, and pleuritic chest pain developed. When he arrived at the hospital on October 20, he was afebrile and normotensive, pulse was 127/min, and respiratory rate 20/min. He was in no acute distress, and physical examination was unremarkable, except for decreased breath sounds at both bases. His past medical history was unremarkable, and he did not smoke. Admission laboratory results were normal, except for elevated bilirubin and hepatic enzymes, low albumin, and hypoxia (Table 1). A noncontrasted head CT was normal, and CSF exhibited 4 WBC/µL (all lymphocytes), 20 RBC/µL, with normal glucose and protein. No organisms were seen on Gram stain of the CSF, and CSF culture did not grow. An anteroposterior chest X-ray showed a widened mediastinum, bilateral hilar masses, right pleural effusion, and bilateral perihilar air space disease. A noncontrasted chest CT scan showed diffuse mediastinal edema; bilateral pleural effusions; bibasilar air space disease; and marked paratracheal, subcarinal, hilar, and azygo-esophageal recess adenopathy. Admission blood cultures grew B. anthracis within 15 hours. Ciprofloxacin, rifampin, and clindamycin were begun. On October 22, signs of worsening respiratory distress developed, and on October 23, the patient underwent therapeutic thoracentesis, which yielded bloody pleural fluid, after which his condition improved. He also received systemic corticosteroids for bronchospasm. He required a second thoracentesis and was discharged from the hospital on November 9.
Case 5
On October 16, a 55-year-old African-American man, a District of Columbia US Postal Service employee, became ill. He had fever, intermittent diaphoresis, myalgias, and cough productive of green sputum but no shortness of breath, chest discomfort, gastrointestinal symptoms, or headache. When he visited his primary-care provider on October 18, he had temperature of 38.9°C, normal heart rate and blood pressure, and respiratory rate of 24/min. He had no other notable examination findings, but WBC count was slightly elevated (Table 1). His past medical history included diabetes mellitus and sarcoidosis. He did not smoke. No chest X-ray was performed. He was sent home with a diagnosis of a viral syndrome; no antibiotics were prescribed. On October 21, he arrived at the emergency department with worsening symptoms, including chest tightness, fatigue, chills, myalgias, nausea, vomiting, and shortness of breath. Temperature was 38.9°C, pulse 93 to 150/min and irregular, respiratory rate 20/min, and blood pressure 119/73 mm Hg. Signs of respiratory distress were observed. Examination findings included rales at the right base with diffuse wheezing and tachycardia. WBC count was 18,800/mm3 with a differential of 73% segmented neutrophils, 6% bands, 11% lymphocytes, and 8% monocytes. Hematocrit was 55%, and platelets were 141,000/mm3. Sodium was 130 mmol/L, potassium 5.3 mmol/L, chloride 99 mmol/L, and bicarbonate 14 mmol/L, with an anion gap of 17. Creatinine was 1.6 mg/dL, and glucose was 425 mg/dL. Aspartate aminotransferase (AST) was 76 IU/lL and alanine aminotransferase (ALT) was 77 IU/L. Coagulation studies were normal. Arterial pH was 7.42, PaCO2 25 mm Hg, PaO2 66 mm Hg, and O2 saturation 93% on 2 L of O2/min by nasal cannula. A chest X-ray showed right hilar and peritracheal soft tissue fullness with right middle and lower lobe infiltrates compatible with pneumonia and right pleural effusion. An electrocardiogram showed atrial fibrillation. The patient was intubated, ventilated, and administered levofloxacin, diltiazem, and insulin. Later on the day of admission, the patient became hemodynamically unstable, had cardiac arrest, and died. Blood cultures grew B. anthracis. Autopsy findings included hemorrhagic mediastinal lymphadenitis, and immunohistochemical staining showed evidence of disseminated B. anthracis.
Case 6
On October 16, a 47-year-old African-American man, a US Postal Service employee who worked at the same District of Columbia mail distribution center associated with cases 3, 4, and 5, had mild nonproductive cough, nausea, vomiting, and stomach cramps. On October 20, the patient had a syncopal episode at church but did not seek medical attention. Early in the morning of October 21, he arrived at an emergency department complaining of vomiting and profuse sweating. His past medical history included asthma and renal calculi. Therapy for asthma had not recently included corticosteroids. He was afebrile and had orthostatic hypotension. WBC count was slightly elevated, but he had normal serum chemistries and coagulation values. Serum glutamic pyruvic transaminase (SGPT) was slightly elevated (Table 1). A chest X-ray was initially read as normal, but later review noted an ill-defined area of increased density due to infiltrate or mass in the right suprahilar region. The patient was discharged after receiving intravenous hydration. On the morning of October 22, he visited the emergency department again, reporting myalgias, chills, dyspnea, continued vomiting, and another syncopal episode. His temperature was 35.6°C, blood pressure 76/48 mm Hg, heart rate 152/min, and respiratory rate 32/min. He was ill-appearing with mottled skin that was cool to the touch, and he was in respiratory distress. He had bilateral wheezing, tachycardia, and mildly distended abdomen with absent bowel sounds. WBC count was 31,200/mm3 with a differential of 78% segmented neutrophils, 2% bands, 14% lymphocytes and 3% monocytes. Sodium was 148 mmol/L, bicarbonate 18 mmol/L, anion gap 21, and creatinine 2.8 mg/dL. Serum glutamic oxalacetic transaminase (SGOT) was 47 IU/L, SGPT 33 IU/L, and alkaline phosphatase 197 IU/L. Prothrombin time was 13.3 seconds and partial thromboplastin time was 40 seconds. Penicillin, ceftriaxone, rifampin, and levofloxacin were begun. Respiratory distress developed, which required endotracheal intubation and mechanical ventilation. Soon thereafter signs consistent with peritonitis were observed. Arterial pH was 7.13, PaCO2 37 mm Hg, PaO2 106 mm Hg, and oxygen saturation 95% after intubation on 100% FiO2. A chest X-ray showed bilateral lung infiltrates concentrated within perihilar and infrahilar regions without pleural effusions. Chest and abdominal CT scans with intravenous contrast noted large bilateral pleural effusions, perihilar and suprahilar infiltrates, mediastinal edema, pneumomediastinum, ascites, air in the portal venous system, mesenteric edema, diffuse small bowel edema, and small collections of intramural air involving several parts of the jejunum. CT of the head was normal. The patient died within 6 hours of admission. Gram-positive bacilli were visible on the buffy coat blood smear, and blood cultures grew B. anthracis within 18 hours. Postmortem findings included prominent hemorrhagic mediastinal lymphadenitis and evidence of systemic B. anthracis infection by histopathologic and immunohistochemical tests.
Case 7
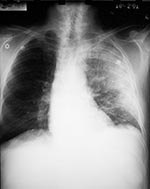
On the evening of October 22, a 59-year-old Caucasian man, contract employee at a U.S. State Department mail sorting facility that received mail from the District of Columbia postal facility associated with cases 3, 4, 5, and 6, became ill. He had drenching sweats, followed over the next 2 days by fatigue, severe myalgias, subjective fever, chills, headache, nausea, vomiting, abdominal pain, cough with scant white sputum, and substernal chest pain. He had no dyspnea or diarrhea. When he arrived at a local emergency room on October 24, temperature was 38.2°C, and heart rate 116/min, and respiratory rate and blood pressure were normal. A complete blood count was normal, and serum electrolytes showed hyponatremia and hypokalemia (Table 1). His past medical history was unremarkable, and he did not smoke. A chest X-ray was initially reported as normal. The patient was thought to have a viral syndrome and was discharged, but blood cultures were obtained and ciprofloxacin was prescribed. He took one dose that night, but vomiting, fatigue, and headache worsened. He also reported transient distortion in his left visual field, and his wife reported that he was intermittently confused. Blood cultures grew gram-positive bacilli after 17 hours of incubation; therefore, on October 25, he was called back to the hospital for admission. The blood isolate was subsequently identified as B. anthracis. At admission, his vital signs were as follows: temperature 38.2°C, heart rate 108/min, respiratory rate 20/min, blood pressure 121/60 mm Hg, and oxygen saturation 94% on room air. He appeared ill and had decreased breath sounds at the right base. The rest of the examination was unremarkable. Laboratory studies on admission included WBC count 9,500/mm3 with 81% segmented neutrophils, 9% lymphocytes, and 9% monocytes, hematocrit 48.1%, platelet count 196,000/ mm3, normal electrolytes and creatinine, SGOT 85 IU/L, SGPT 64 IU/L, alkaline phosphatase 141 IU/L, bilirubin 1.6 mg/dL, and albumin 3.0 mg/dL. On review, the initial chest X-ray showed mediastinal widening (Figure 5), and chest CT on the day of admission showed mediastinal adenopathy with evidence of hemorrhage, normal lung parenchyma, small bilateral pleural effusions, and a suspected small pericardial effusion (Figure 6). Intravenous penicillin and rifampin were added to the ciprofloxacin. His temperature rose to 39°C. Subsquently, vancomycin was added and penicillin was discontinued. On October 26, gastrointestinal bleeding developed, which required blood transfusion, endoscopic injection, and cautery of gastric and duodenal ulcers. On October 27, atrial fibrillation with variable ventricular response developed. On October 28, fever reached a maximum of 39.4°C and then decreased to 38.3°C. On October 30, WBC peaked at 31,300/mm3. On October 31, enlargement of the right pleural effusion required thoracentesis and removal of 900 cc of serosanguinous fluid (Table 1). The patient was discharged from the hospital on November 9.
Case 8
On October 14, a 56-year-old African-American woman who worked as a mail sorter in the Hamilton, New Jersey, US Postal Service facility, became ill with vomiting and diarrhea, followed the next day by subjective fever and chills unrelieved by aspirin. The vomiting and diarrhea improved, but over the next 2 days she had fevers to 38.4°C with shaking chills, headache, and fatigue. A nonproductive cough developed, along with mild shortness of breath, and anterior chest pain on inspiration. She had no sore throat or rhinorrhea. On October 19, because of persistent fever and worsening chest pain, she went to a local emergency room. Findings at that time included temperature 38.4°C, heart rate 120/min, blood pressure 159/95 mm Hg, and respiratory rate 18/min. She appeared ill with increased respiratory effort, had decreased breath sounds at both bases, and had a 0.5- to 1.0-cm healing scab on the anterior neck. Initial WBC was normal except for elevation in neutrophil band forms, and hematocrit and platelets were normal. Serum electrolytes, creatinine, and coagulation values were unremarkable, except for hyponatremia. Hepatic enzymes were elevated, and she was hypoxic (Table 1). Her past medical history included a transient ischemic attack. She did not smoke. A chest X-ray showed bibasilar infiltrates and a small right pleural effusion but no mediastinal widening. Initial differential diagnosis included atypical pneumonia versus inhalational anthrax, and the patient was begun on levofloxacin; rifampin was added the next day. Two days later, the fever persisted, dyspnea worsened, and large bilateral pleural effusions developed. Antibiotics were changed to ciprofloxacin, rifampin, and vancomycin. Right-sided thoracenteses were performed on October 21 and 22, and 750 cc and 650 cc of hemorrhagic fluid were removed, respectively. On October 23, a right chest tube was placed. A chest CT on October 22 showed mediastinal and cervical lymphadenopathy, bibasilar infiltrates, and large left pleural effusion. On October 25, the enlarging left-sided effusion required chest tube placement. On October 26, the fever resolved, transaminases became normal, and the dyspnea gradually improved. On October 30 and November 1, the chest tubes were removed. On November 5, the patient was discharged from the hospital. Blood for B. anthracis DNA by PCR was positive, as were immunohistochemistry studies for B. anthracis cell-wall and capsule antigens from pleural fluid cytology preparations.
Case 9
On October 15, a 43-year-old South Asian woman, also a mail sorter at the US Postal Service facility in Hamilton but at different mail sorting machines from those used by the patient in Case 8, became ill. She had intermittent fevers with chills, dry cough with chest discomfort and shortness of breath, myalgias, and fatigue. She also had nausea and vomiting but no abdominal pain or diarrhea. She complained of "head stuffiness" but no rhinorrhea or sore throat. She also had headache and was reported to be mildly confused. On October 16, when she visited her primary-care physician, she had a temperature of 38°C and was started on levofloxacin for bronchitis. Her past medical history was unremarkable, and she did not smoke. On October 18, she went to a local emergency room because of persistent symptoms. Vital signs were as follows: temperature 38.4°C, heart rate 120/min, respiratory rate 16/min, and blood pressure 141/85mm Hg, with oxygen saturation 92% on room air and 97% on 4 L of oxygen by nasal cannula. She appeared ill and had decreased breath sounds with egophony at the right base. WBC showed increase in neutrophil band forms; hematocrit and platelet count were normal. She had hyponatremia, but electrolytes, renal function, and coagulation values were otherwise normal. Hepatic enzymes were elevated (Table 1). Blood PCR for B. anthracis DNA obtained 2 days after initiation of antibiotics was negative. A chest X-ray showed right hilar opacity consistent with consolidation or mass, moderate right and minimal left pleural effusions. Antibiotics were changed to azithromycin and ciprofloxacin. Ciprofloxacin was discontinued 24 hours later. On October 19, a chest CT showed increased soft tissue in the mediastinum (thought to represent adenopathy), right hilar consolidation with possible underlying mass, and large right pleural effusion. Clindamycin and ceftriaxone were added, and thoracentesis was performed with removal of 500 cc of serosanguinous fluid (Table 1). Pleural fluid cytology preparation was positive for B. anthracis cell-wall and capsule antigens by immunohistochemical staining. On October 21, repeat thoracentesis was required, and 800 cc of fluid was removed. On October 22, bronchoscopy found edematous, erythematous mucosa. A transbronchial biopsy showed B. anthracis cell-wall and capsule antigens by immunohistochemical staining. Cultures of endobronchial samples, pleural fluid, and a nasal swab were all negative for B. anthracis, and no other pathogens were identified. On October 23, the fever resolved, other symptoms began to improve, treatment was changed to doxycycline, and the patient was discharged from the hospital on doxycycline on October 26.
Case 10
On October 25, a 61-year-old Asian woman who worked in the supply room of a New York City Hospital had onset of malaise and myalgias. Over the next 2 days, she also had fatigue, chills, chest pain, progressively worsening dyspnea, and cough productive of sputum, which was later blood-tinged; she denied fever. On October 28, 2001, when she went to a local hospital (other than her workplace), she was noted to be febrile in the emergency room and normotensive, but her heart rate was 110/min and respiratory rate 38/min, with room-air oxygen saturation of 92% by pulse oximetry. She was awake, alert, and completely oriented. She had prominent jugular venous distension at 60 degrees. She had a history of hypertension. She did not smoke. Chest examination showed rales heard to the apices bilaterally. Abdominal and cardiovascular examinations were normal except for tachycardia, and she had no peripheral edema. WBC count was slightly elevated; hematocrit and platelets were normal. Serum chemistries and coagulation studies were normal except for hyponatremia, elevated hepatic enzymes, and hypoalbuminemia. The serum lactate dehydrogenase level was 1,370 IU/L. The patient required oxygen delivery by a nonrebreather mask to maintain adequate oxygenation (Table 1). Chest X-ray was initially interpreted as showing pulmonary venous congestion with bilateral pleural effusions. Therapy for congestive heart failure was initiated, but an echocardiogram in the emergency department showed normal ejection fraction, no substantial wall motion abnormalities, and a small pericardial effusion. Therapy was changed to levofloxacin for atypical pneumonia. The patient's respiratory status worsened, and she was intubated. A CT scan of the chest showed mediastinal lymphadenopathy, massive mediastinal bleeding, thickened bronchial mucosa, and bilateral pleural effusions. Rifampin, gentamicin and subsequently nafcillin were added to her antimicrobial regimen. Early on October 29, 2001, bilateral chest tubes were placed, and 2.5 liters of serosanguinous fluid was drained from the right side and 1.0 liter from the left (Table 1). On the same day, bronchoscopy showed hemorrhagic mucosa throughout the entire tracheobronchial tree, friable and collapsible airways, and purulent secretions in multiple segments bilaterally. On October 30, the antimicrobial regimen was changed to ciprofloxacin, rifampin, clindamycin, and ceftazidime. Serial echocardiograms documented enlarging pericardial effusion, ultimately with tamponade. The patient's clinical condition progressively worsened, and she died on October 31, after attempted pericardiocentesis. Cultures of blood (after 20 hours) and pleural fluid grew B. anthracis. Autopsy findings included hemorrhagic mediastinitis; immunohistochemical stains confirmed the presence of B. anthracis in multiple organs.
The median age of patients with inhalational anthrax in this series was 56 years (range 43 to 73 years), 70% were male and, except for the patient in Case 10, all were known to or believed to have processed, handled, or received letters containing B. anthracis spores (postal workers [n=6], mail handlers or sorters [n=2], journalist [n=1]). The source of exposure to B. anthracis spores in Case 10, a hospital supply room worker, remains undefined. Four patients had underlying cardiovascular or cerebrovascular disease, one had a remote history of treated sarcoidosis and diet-controlled diabetes mellitus, and one had a history of asthma. None of the patients was a current or recent smoker.
The median incubation period from the time of exposure to onset of symptoms when known (n=6) was 4 days (range 4 to 6 days). Two of the patients (in Cases 8 and 9) are presumed to have been exposed on October 9, when a letter later known to be contaminated with spores of B. anthracis was processed in the postal facility where they worked. The same letter was routed through a second postal facility and processed on October 12, the presumed day of exposure for four additional cases of inhalational anthrax (Cases 3, 4, 5, and 6).
Patients sought care a median of 3.5 days (range 1 to 7 days) after onset of symptoms. Eight of 10 patients were in the initial phase of illness when they first sought care. Of these eight, six received antibiotics with activity against B. anthracis on the same day, and all six survived. Four patients, including one with meningitis, were exhibiting fulminant signs of illness when they first received antibiotics with activity against B. anthracis, and all four died. Table 2 summarizes the symptoms of the 10 patients with bioterrorism-related anthrax. Table 3 lists major initial clinical, laboratory and radiologic findings. The median initial WBC count was 9,800/mm3 (range 7,500 to 13,300/mm3). However, the median peak WBC count after presentation and during the course of the illness was 26,400/mm3 (range 11,900 to 49,600/mm3). The chest X-ray was abnormal in all patients, but only seven had mediastinal changes.
Pleural effusions were present in all 10 patients and often became large during hospitalization in those who survived. Seven patients required drainage of pleural fluid, three of these with chest tubes; pleural fluid was consistently hemorrhagic with relatively few leukocytes. Pulmonary infiltrates, which were seen in seven patients, involved the right and left lower lobes (one patient), left upper and lower lobes (one), right upper lobe (two), right lower lobe (one), right middle and right lower lobes (one), and bilateral perihilar areas (one). Eight patients had a chest CT performed; mediastinal lymphadenopathy was present in all but one patient.
Blood cultures were obtained before antibiotic therapy was initiated in seven patients, and B. anthracis was isolated in all seven. Blood cultures grew B. anthracis at a median of 18 hours (range 12 to 24 hours). Three patients had the first set of blood cultures obtained after initiation of antibiotic therapy; these cultures revealed no growth. One patient with blood cultures that grew B. anthracis had blood cultures repeated within 24 hours after initiating antibiotics with activity against B. anthracis, and the repeat cultures did not grow.
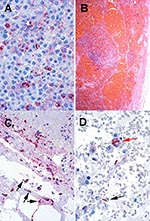
B. anthracis-specific immunohistochemical tests performed on pleural fluid cytology preparations or transbronchial biopsy specimens were positive in every case in which these tissues were available (three nonfatal cases) (Figure 7A). Three patients had atrial arrythmias (supraventricular tachycardia in Case 2 and atrial fibrillation in Cases 5 and 7), and three patients had pericardial effusion on CT scan. In one patient, pericardial tamponade was suspected. Six of 10 patients with inhalational anthrax have survived to date (death rate 40%). Five have been discharged from the hospital, and one is recovering in the hospital. Figure 8 shows the timeline from exposure (when known) to current status for these 10 patients. Autopsy findings in all four patients who died showed hemorrhagic mediastinal lymphadenitis and evidence of disseminated B. anthracis infection (Figure 7B-D)
Before this outbreak of bioterrorism-related anthrax, only 18 cases of inhalational anthrax had been reported in the United States in the 20th century (1,4,9-21). The most recent case was in 1976 (4). Most cases were related to exposure to animal products, primarily in textile mills processing goat hair, goat skins, or wool (1). Clinical characteristics in the 10 cases due to bioterrorism described here share similarities with previously reported cases but have important differences.
Inhalational anthrax has been described as a biphasic clinical illness characterized by a 1- to 4-day initial phase of malaise, fatigue, fever, myalgias, and nonproductive cough, followed by a fulminant phase of respiratory distress, cyanosis, and diaphoresis (1). Death follows the onset of the fulminant phase in 1 to 2 days (2,22). The symptoms of the initial phase of inhalational anthrax in the 10 cases caused by bioterrorism (Table 1) were similar to those of the 18 occupationally related cases previously described (1); however, profound, often drenching sweating, which was a prominent feature in the current cases, was not emphasized in earlier reports. Previous case reports have noted a brief period of improvement between the initial and fulminant phases (1), but this phenomenon was not observed in the current cases. Nausea and vomiting were also frequent symptoms of the initial phase in the current cases, suggesting early involvement of the gastrointestinal tract. Gastrointestinal lesions were observed in 39 of 42 total cases of fatal inhalational anthrax associated with the Sverdlovsk outbreak (23). Most lesions in these fatal cases appeared to represent hematogenous spread of B. anthracis to the submucosa of the gastrointestinal tract and did not involve Peyer's patches or, in most cases, mesenteric lymph nodes.
The nondistinctive nature of the initial phase of inhalational anthrax presents a diagnostic challenge. The chest X-ray appeared to be a sensitive indicator of disease in patients with bioterrorism-associated inhalational anthrax, as none of the 10 patients had an initially normal chest X-ray. Multiple abnormalities were noted on initial chest X-ray, including mediastinal widening, paratracheal fullness, hilar fullness, pleural effusions,and parynchemal infiltrates. Two patients had chest X-rays that were interpreted initially as normal, but abnormalities (mediastinal widening in one case and perihilar mass versus infiltrate in the other) were detected when the X-rays were reviewed by a radiologist. Pulmonary infiltrates or effusion were initially seen in two cases without evidence of mediastinal widening. Chest CT was helpful in further characterizing abnormalities in the lungs and mediastinum and was more sensitive than chest X-ray in revealing mediastinal lymphadenopathy.
The total WBC count was usually normal or only slightly elevated at the time of initial visit to a health-care provider for patients who sought medical care in the initial phase. However, an elevation in the proportion of neutrophils or band forms was frequently noted and was an early diagnostic clue. During the course of illness, WBC counts increased, sometimes markedly, in most patients.
Blood cultures grew B. anthracis even in the initial phase of the illness in all patients who had not received prior antibiotic therapy. Animal models suggest that primates with inhalational anthrax become bacteremic early in the course of the illness before the fulminant clinical phase develops (24). In contrast, blood cultures rapidly became sterile after initiation of antibiotic therapy, suggesting that prior antibiotic treatment may substantially decrease the sensitivity of blood cultures as a diagnostic test.
The diagnosis of anthrax was established in three patients without growth of B. anthracis from clinical specimens. In all three of these cases, proper cultures were obtained only after initiation of antibiotic therapy. The diagnosis in these patients was established by a history of exposure or occupational and environmental risk with a clinically compatible syndrome, by the identification of B. anthracis in pleural fluid, pleural biopsy, or transbronchial biopsy specimens by immunohistochemical staining with B. anthracis-specific cell wall and capsular antibodies, or by identifying B. anthracis DNA by PCR on pleural fluid or blood. Serologic data from ELISA available for one patient with inhalational anthrax also demonstrated a >4-fold increase in levels of serum antibody (IgG) to the PA component of anthrax toxins.
The survival of patients with inhalational anthrax in this series (60%) is higher than previously reported (<15%) (1,3). All patients received combination antimicrobial therapy with more than one agent active against B. anthracis. The apparent improvement in survival compared with previous cases suggests that the antibiotic combinations used in these patients may have therapeutic advantage over previous regimens. Limited data on treatment of the survivors suggest that early treatment with a fluoroquinolone and at least one other active drug (7) may improve survival. Other antimicrobial susceptibilities of the isolates associated with this outbreak have been published (7). B. anthracis isolates produce a cephalosporinase (7) that inhibits the antibacterial activity of cephalosporins such as ceftriaxone, and cephalosporins should not be used for treatment. Other explanations for the improved survival rate include earlier recognition and initiation of treatment, better supportive care, differences in the pathogenesis of bioterrorism-related anthrax, differences in susceptibility of the hosts, or a combination of the above.
Pleural effusions were a remarkably consistent clinical feature of inhalational anthrax in this series, occurring in all patients. The pleural effusions were often small on presentation, but in the surviving patients effusions were characterized by progressive enlargement and persistence. Drainage of the pleural cavity was required in seven patients. The characteristics of the pleural fluid in all patients were similar: hemorrhagic, with a high protein concentration and relatively few WBCs. Immunohistochemistry demonstrated large quantities of B. anthracis capsule and cell-wall antigens in pleural tissue or pleural fluid cell blocks.
Case 2 was remarkable in that X-ray findings were dominated by large and progressive pulmonary infiltrates, not by mediastinal widening. In fact, no mediastinal adenopathy was noted on chest CT. Transbronchial biopsies of the patient in Case 2 showed B. anthracis-specific capsular and cell-wall antigens in the lung parenchyma. Previous reports have noted bronchopneumonia and pulmonary hemorrhagic infarcts in patients with inhalational anthrax. Abramova et al. (23) described focal hemorrhage and necrotizing anthrax pneumonia in 11 of 42 patients who died with inhalational anthrax. Characteristics of the spore-containing aerosol or individual host factors may influence pathogenesis, so pulmonary infiltrates may be a more prominent manifestation in some patients with bioterrorism-related inhalational anthrax. Three patients had supraventricular arrythmias, and three had pericardial effusion on chest CT, one of which may have progressed to tamponade. Pericardial effusions may reflect toxin-related local edema or inflammation, hemorrhagic necrosis and infarct extending into the pericardium from the mediastinum, or hematogenous spread of B. anthracis.
Anthrax meningitis is a complication of inhalational anthrax and is characteristically hemorrhagic. Pathologic findings are a hemorrhagic leptomeningitis with edema and inflammatory infiltrates (2,23). Fifty-five percent of patients at Sverdlovsk who died of inhalational anthrax had evidence of meningeal involvement at autopsy. Cerebrospinal fluid was examined in only two patients in our series, and meningitis was documented in one. Three other patients had a history of intermittent confusion, but no meningeal signs were observed, and lumbar punctures were not performed. No predominant underlying diseases or conditions were noted in the patients with bioterrorism-related inhalational anthrax. One patient (Case 5) had a remote history of sarcoidosis but had been free of clinical illness or treatment associated with this diagnosis for the past 25 years. One other case of inhalational anthrax associated with underlying sarcoidosis has been reported (12). The patient in Case 5 also had diabetes mellitus, a condition present in one previously reported case (12). Smoking did not appear to be a risk factor for inhalational anthrax.
In summary, we describe the clinical presentation of the first 10 cases of bioterrorism-related inhalational anthrax in the United States. The clinical presentation in these patients was variable and often resembled a viral respiratory illness, but the interpretation of the initial symptoms in the context of a possible exposure to B. anthracis often led to an early diagnosis. In contrast to previous reports indicating a death rate >85% (1,3), our series suggests that survival may be markedly improved by combination antimicrobial therapy begun during the initial phase of the illness and by aggressive supportive care (e.g., drainage of pleural effusions). Newer methods of detection such as polymerase chain reaction, immunohistochemistry, and sensitive serologic tests are important adjunctive diagnostic modalities that aid in the diagnosis of B. anthracis infections. Further studies are needed to better define optimal antimicrobial regimens, explore the role of adjunctive therapies (e.g., immunoglobulin antitoxin, corticosteroids, and other toxin inhibitors), and better understand the pathogenesis of inhalational anthrax associated with intentional release.
Acknowledgment
We thank Sherrie Bruce, Rebecca Dixon, Anexis Lopez, Pat McConnon, Carmen Resurreccion, Kay Vydareny, the laboratories of the National Center for Infectious Diseases, the laboratories of affected states, the U.S. Department of Defense, and Epidemic Intelligence Service Officers for their assistance in preparing this article.
References
- Dixon TC, Meselson M, Guillemin J, Hanna P. Anthrax. N Engl J Med. 1999;341:815–26. DOIPubMedGoogle Scholar
- Meselson M, Guillemin J, Hugh-Jones M, Langmuir A, Popova I, Shelokov A, The Sverdlovsk anthrax outbreak of 1979. Science. 1994;266:1202–8. DOIPubMedGoogle Scholar
- Suffin SC, Carnes W, Kaufmann A. Inhalation anthrax in a home craftsman. Hum Pathol. 1978;9:594–7. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Notice to readers: ongoing investigation of anthrax--Florida, October 2001. MMWR Morb Mortal Wkly Rep. 2001;50:877.
- Centers for Disease Control and Prevention. Update: investigation of bioterrorism-related anthrax and interim guidelines for clinical evaluation of persons with possible anthrax. MMWR Morb Mortal Wkly Rep. 2001;50:941–8.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: investigation of bioterrorism-related anthrax and interim guidelines for exposure management and antimicrobial therapy, October 2001. MMWR Morb Mortal Wkly Rep. 2001;50:909–19.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: investigation of anthrax associated with intentional exposure and interim public health guidelines, October 2001. MMWR Morb Mortal Wkly Rep. 2001;50:889–93.PubMedGoogle Scholar
- Albrink WS, Brooks S, Biron R, Kopel M. Human inhalation anthrax: a report of three fatal cases. Am J Pathol. 1960;36:457–71.PubMedGoogle Scholar
- Case records of the Massachusetts General Hospital, case #14032: an acute infection with bloody fluid in the right pleural space. N Engl J Med. 1928;198:148–53.
- Brachman PS, Plotkin S, Bumford F, Atchison M. An epidemic of inhalation anthrax: the first in the twentieth century. II. Epidemiology. Am J Hyg. 1960;72:6–23.PubMedGoogle Scholar
- Brachman P, Pagano J, Albrink W. Two cases of fatal inhalation anthrax, one associated with sarcoidosis. N Engl J Med. 1961;265:203–8. DOIGoogle Scholar
- Fletcher J. Human anthrax in the United States: a descriptive review of case reports, 1955-1999. Rollins School of Public Health. Atlanta: Emory University; 2000.
- Gold H. Anthrax: a report of one hundred seventeen cases. AMA Arch Intern Med. 1955;96:387–96.PubMedGoogle Scholar
- LaForce FM, Bumford B, Feeley J, Stokes S, Snow D. Epidemiologic study of a fatal case of inhalation anthrax. Arch Environ Health. 1969;18:798–805.PubMedGoogle Scholar
- LaForce F. Woolsorter's disease in England. Bull NY Acad Sci. 1978;54:956–63.
- Levinsky W, Anderson T, Richardson G. Inhalation anthrax meningitis and bacillemia: a case report. In: Proceedings of the Symposium on Anthrax in Man. Philadelphia, Pennsylvania; 1954 Oct 9. p. 96-103.
- Plotkin SA, Brachman P, Utell M, Bumford F, Atchison M. An epidemic of inhalation anthrax, the first in the twentieth century, I. Clinical features. Am J Med. 1960;29:992–1001. DOIPubMedGoogle Scholar
- Krane S. Cases from the medical grand rounds of the Massachusetts General Hospital, case 388: sinusitis due to anthrax. Am Pract Dig Treat. 1957;8:1628–36.
- Brooksher W, Briggs J. Pulmonary anthrax: report of a case. JAMA. 1920;74:323–4.
- Cowdery JA. Primary pulmonary anthrax with septicemia. Arch Pathol (Chic). 1947;43:396–9.PubMedGoogle Scholar
- Brachman P, Kaufmann A. Anthrax. In: Evans A, Brachman P, editors. Bacterial infections of humans. New York: Plenum Medical Book Company; 1998. p. 95-107.
- Abramova FA, Grinberg L, Yampolskaya O, Walker D. Pathology of inhalational anthrax in forty-two cases from the Sverdlovsk outbreak of 1979. Proc Natl Acad Sci U S A. 1993;90:2291–4. DOIPubMedGoogle Scholar
- Albrink WS, Goodlow R. Experimental inhalation anthrax in the chimpanzee. AM JPatho. 1959;35:1055–65.PubMedGoogle Scholar
Figures
Tables
Cite This Article1Members of the team who contributed to the work presented in this manuscript are J. Aguilar, M. Andre, K. Baggett, B. Bell, D. Bell, M. Bowen, G. Carlone, M. Cetron, S. Chamany, B. De, C. Elie, M. Fischer, A. Hoffmaster, K. Glynn, R. Gorwitz, C. Greene, R. Hajjeh, T. Hilger, J. Kelly, R. Khabbaz, A. Khan, P. Kozarsky, M. Kuehnert, J. Lingappa, C. Marston, J. Nicholson, S. Ostroff, T. Parker, L. Petersen, R. Pinner, N. Rosenstein, A. Schuchat, V. Semenova, S. Steiner, F. Tenover, B. Tierney, T. Uyeki, S. Vong, D. Warnock, C. Spak, D. Jernigan, C. Friedman, M. Ripple, D. Patel, S. Pillai, S. Wiersma, R. Labinson, L. Kamal, E. Bresnitz, M. Layton, G. DiFerdinando, S. Kumar, P. Lurie, K. Nalluswami, L. Hathcock, L. Siegel, S. Adams, I. Walks, J. Davies-Coles, M. Richardson, K. Berry, E. Peterson, R. Stroube, H. Hochman, M. Pomeranz, A. Friedman-Kien, D. Frank, S. Bersoff-Matcha, J. Rosenthal, N. Fatteh, A. Gurtman, R. Brechner, C. Chiriboga, J. Eisold, G. Martin, K. Cahill, R. Fried, M. Grossman, and W. Borkowsky.
Table of Contents – Volume 7, Number 6—December 2001
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
John A. Jernigan, NCID, CDC, Mailstop E68, 1600 Clifton Road, N.E., Atlanta, GA 30333 USA; fax: 404-498-1244
Top