Volume 12, Number 6—June 2006
Research
Co-infections of Adenovirus Species in Previously Vaccinated Patients
Cite This Article
Citation for Media
Abstract
Despite the success of the adenovirus vaccine administered to US military trainees, acute respiratory disease (ARD) surveillance still detected breakthrough infections (respiratory illnesses associated with the adenovirus serotypes specifically targeted by the vaccine). To explore the role of adenoviral co-infection (simultaneous infection by multiple pathogenic adenovirus species) in breakthrough disease, we examined specimens from patients with ARD by using 3 methods to detect multiple adenoviral species: a DNA microarray, a polymerase chain reaction (PCR)–enzyme-linked immunosorbent assay, and a multiplex PCR assay. Analysis of 52 samples (21 vaccinated, 31 unvaccinated) collected from 1996 to 2000 showed that all vaccinated samples had co-infections. Most of these co-infections were community-acquired serotypes of species B1 and E. Unvaccinated samples primarily contained only 1 species (species E) associated with adult respiratory illness. This study highlights the rarely reported phenomenon of adenoviral co-infections in a clinically relevant environment suitable for the generation of new recombinational variants.
Adenoviruses cause an estimated 8% of clinically relevant viral disease globally (1). Human adenoviruses (HAdVs) are divided into 51 serotypes (HAdV-1–HAdV-51) on the basis of type-specific antiserum-mediated neutralization of infectivity (determined primarily by the hexon coat protein and terminal knob portion of the fiber protein) (2) and into 6 species, also referred to as subgenera or subgroups (HAdV-A, B, C, D, E, and F) on the basis of hemagglutination inhibition and biochemical criteria (3–5). Species HAdV-B is further classified into subspecies B1 and B2 (3). In civilian populations, HAdV-B1 serotypes 3, 7, 16, and 21; HAdV-E serotype 4; and 1 member of subspecies HAdV-B2, serotype 14, cause outbreaks of illness ranging from mild febrile respiratory infections and conjunctivitis to potentially lethal disseminated infections in both adults and children (1,6). HAdV-C serotypes 1, 2, 5, and 6 cause locally endemic upper respiratory infections in infants and children (7,8) and occasional outbreaks in adults. Other HAdV species are usually not associated with respiratory disease in otherwise healthy humans.
HAdV seems to have found a particularly destructive niche in military training camps. HAdV-B1 serotypes 3, 7, and 21; HAdV-E serotype 4; and HAdV-B2 serotype 14 have caused severe outbreaks of acute respiratory disease (ARD) among military recruits in training centers (9,10). Before initiation of an HAdV vaccination program in 1971, outbreaks occurred regularly, and ≈1 of 6 recruits in affected camps required hospitalization (1). Systematic vaccination of recruits against the 2 most common agents of ARD in the military, HAdV serotypes 4 and 7, decreased HAdV-specific respiratory illness by 95% to 99% and overall respiratory illness rates by 50% to 60% (11–13). Despite this general efficacy, breakthrough infection (infection of vaccinated persons by the vaccine-targeted adenoviral serotypes) was still regularly reported (14). Production of the vaccine was suspended in 1996, at which point vaccination became sporadic until the existing stocks ran out in 1999. ARD rates quickly returned to prevaccine levels, with HAdV as the apparent causal agent. As a result, reintroduction of the vaccine is being actively pursued (15).
To explore the possibility that unique HAdV strains were causing ARD in vaccinated persons, throat swab samples were selected from the Naval Health Research Center population-based febrile respiratory illness surveillance collection from vaccinated (n = 21) and unvaccinated (n = 31) recruits who reported ARD from 1996 to 2000. Samples were chosen that had tested positive for serotypes 4 or 7 by culture and serotypic antibody neutralization. The gene coding for the primary adenoviral antigen, the hexon coat protein, was sequenced from these isolates. The sequence data suggested that the detectable serotype 4 and 7 strains apparently responsible for breakthrough infection were the same as those circulating in unvaccinated military and civilian populations (16). In this study, we reanalyze the same set of samples to identify co-infections with multiple HAdV strains and to address what role co-infections may play in breakthrough infection.
Sample Collection and Preparation
Samples were collected as throat swabs into viral transport medium from military recruits with ARD at a variety of training camps as previously described (16). The throat swab samples were cultured on A549 cells and tested by using standard serologic methods. Both original swabs and in vitro tissue culture fluid (ITCF) samples were stored at –80°C. Samples that initially tested positive for serotypes 4 or 7 by culture and microneutralization were chosen for analysis and grouped by previous vaccination status. DNA extracts from ITCF samples were collected and used in molecular assays. Collection details and symptom definitions were previously reported (16), and sample details are shown in Table 1 and Table 2. Initially, 13 unidentified (blinded) samples were sent by the Naval Health Research Center to the Naval Research Laboratory personnel for testing. After the initial 13 samples showed a high rate of respiratory HAdV co-infection, primarily in vaccinated persons, an additional 39 samples were tested in an unblinded fashion.
Microarray-based Genotyping
One microliter of purified DNA extract from each of the 52 ITCF samples was used as the template in 50-μL degenerate PCR amplifications targeting portions of the E1A, hexon, and fiber genes. The primers, degenerate polymerase chain reaction (PCR) amplification protocol, probes, and microarray fabrication techniques have been previously described (18). Once constructed, the spotted microarrays were blocked with a 3% bovine serum albumin–casein solution (BSA-C) for 15 min at room temperature, and the slides were outfitted with MAUI Mixer DC hybridization chambers (BioMicro Systems, Salt Lake City, UT, USA). Twenty-microliter hybridization reactions (13.6 μL biotinylated degenerate PCR amplicons, 2 μL 3% BSA-C, 4 μL 20× SSC (0.3 mol/L sodium citrate, 3.0 mol/L NaCl, pH 7.0), and 0.4 μL 10% sodium dodecyl sulfate [SDS]) were denatured for 3 min at 98°C and immediately applied to the microarrays. Hybridizations were performed for 2 h at 63°C in a MAUI Hybridization System (BioMicro Systems). Slides were then washed twice with 4× SSC-0.2% SDS buffer and 2× SSC buffer, and hybridization was detected by the sequential addition of Cy5-conjugated mouse antibiotin immunoglobulin G (IgG) (Jackson ImmunoResearch, West Grove, PA, USA) and Cy5-conjugated goat antimouse IgG (Jackson ImmunoResearch). Images were obtained with a ScanArray Lite confocal laser scanning system (Perkin-Elmer, Torrance, CA, USA) at a laser power of 60 to 80 and a photomultiplier tube gain of 60 to 80. The fluorescent signal from each microarray element was considered positive only when its quantified intensity was >3× that of known internal negative control elements. Each ITCF sample was subjected to 2 to 5 independent amplification and hybridization experiments. Hybridization patterns unique to specific serotypes were determined empirically with prototype strains (18). Although members of species HAdV-B1 often produced complex hybridization profiles (18), these profiles were unique, reproducible, and readily identifiable in both single infections and co-infections.
Adenovirus Consensus PCR–Enzyme-linked Immunosorbent Assay
We used a commercially available kit capable of typing adenoviruses to the species level to confirm the results obtained with microarray analyses. Briefly, the Adenovirus Consensus kit (Argene, North Massapequa, NY, USA) uses a PCR–enzyme-linked immunosorbent assay that amplifies a fragment from the adenovirus virus-associated (VA) RNA gene and subsequently detects and types the amplicon with species-specific biotinylated oligonucleotide probes in a colorimetric microwell format (19). Results obtained with the kit were interpreted according to the manufacturer's adenovirus typing protocol.
Adenovirus-specific PCR
The species-specific PCR amplification was performed with previously published primers (20) and a Multiplex PCR Kit (Qiagen, Valencia, CA, USA) according to the manufacturer's instructions (with 0.5× Q solution). These amplifications were performed in 25-μL reaction volumes at an annealing temperature of 52°C. In general, the PCRs were performed in an iCycler (Bio-Rad, Hercules, CA, USA) and analyzed by electrophoresis on 1.5% agarose gels. Monoplex PCR was performed under identical reaction conditions, except that the same primers were used in independent reactions. Sequencing reactions and microneutralization assays were performed as previously described (21,22). Serotype-specific PCR assays (Table 1 and Table 2) were verified as described (20,23–26), with occasional substitutions of polymerase type and annealing temperature adjustments.
Co-infection Separation
Limiting dilutions of ITCF sample 7151 were plated on A549 cells and allowed to adsorb for 16 hours, after which agarose overlays (0.4% agarose in Dulbecco minimal essential medium, 2% fetal bovine serum, 4 mmol/L glutamine) were added to each infected monolayer. Well-separated virus plaques were picked 5 days postinfection, placed into viral transport medium, and tested by PCR for HAdV-B and HAdV-C. A second round of plaque purification was performed on several plaque isolates that were treated with 0.05% Triton-X 100 to potentially disrupt virus clumps before their dilution and plating. After 6 hours of adsorption, the original inoculum was removed, and the monolayers were overlayed with agarose solution. The newly formed plaques were tested as described above.
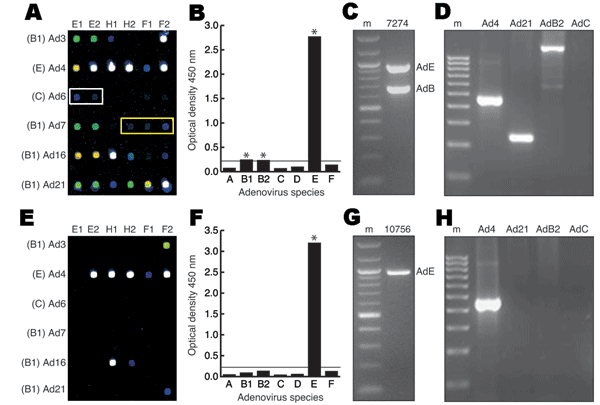
By using a new 70-mer spotted microarray (18), a PCR–enzyme-linked immunosorbent assay (19), and a species-specific multiplex PCR assay (20), we generated data profiles for each of the 52 tissue culture-amplified samples; the raw data from 2 of these samples are shown as representative examples (Figure). The microarray profile of vaccinated sample 7274 detected HAdV-4 (species E), HAdV-21 (species B1), HAdV-C, and HAdV-B2 according to previously validated hybridization patterns (18) (Figure, panel A). Except for detection of an apparent low-level HAdV-C co-infectant, the results of the Adenovirus Consensus kit (HAdV-B1, HAdV-B2, and HAdV-E) (Figure, panel B), multiplex and monoplex species-specific PCR (HAdV-B and HAdV-E) (Figure, panel C), and serotype-specific PCR (HAdV-4, HAdV-21, and HAdV-B2) (Figure, panel D) confirmed the microarray-based identification of multiple adenoviral strains in sample 7274.
In contrast, the microarray profile of unvaccinated sample 10756 detected a single serotype, HAdV-4 (Figure, panel E). The microarray-based finding was verified by results of the Adenovirus Consensus kit (Figure, panel F), multiplex species-specific PCR (Figure, panel G), and HAdV-4 serotype-specific PCR (Figure, panel H). The data profiles for all 52 samples assembled and compared in this manner are shown in Table 1 and Table 2. Dual, triple, and quadruple infections were found in all 21 of the vaccinated samples and in 14 of the 31 unvaccinated samples tested (Table 1 and Table 2).
Previously vaccinated persons showed a high rate of co-infection with both species commonly associated with ARD (HAdV-B1 and HAdV-E), whereas unvaccinated persons were primarily infected with HAdV-E. Since HAdV-4 and HAdV-7 are the 2 most common ARD-associated serotypes, that they were also the most commonly paired respiratory pathogenic co-infectants detected in vaccinated persons is not surprising. When the vaccine was used, the rates of other respiratory adenoviruses were much higher than when the vaccine was not used (16). However, these isolates were chosen for study because they yielded antigenic signals consistent with either HAdV-4 or HAdV-7 and were therefore expected to contain at least 1 of these 2 viruses as the highest titer adenoviral components (Table 1) (16).
The ability of the microarray to identify to the serotype level resulted in the detection of the greatest number of co-infections, despite its inability to detect members of species B2 when a co-infecting HAdV-7 was present (hybridization pattern interference) and members of species F that were not targeted (Table 3). Microarray-based identification of multiple ARD-associated serotypes from diverse HAdV-B1 species (serotypes 3, 7, and 21) was necessary because co-infections with these serotypes would not have been indicated or resolved by methods limited to species-level identification.
Although most apparent co-infections could be verified by each of the primary methods tested and by serotype-specific PCR (e.g., single infections: 10756, 60406, 20142; co-infections: 1212, 7151, 7274), some could not be verified (e.g., 60691, CHPPM2). Those co-infectant signals that could not be verified were usually weak positives. The strains responsible for these signals appeared to be subordinate co-infectants because the predominant serotype or species signals generated for the associated samples by the microarray, Adenovirus Consensus kit, and serotype-specific PCR were corroborated in every case and matched the results obtained from the sequencing experiments previously reported (16).
The microarray and Adenovirus Consensus kit use detection and signal amplification techniques that enhance assay sensitivity and thus render them more sensitive than traditional PCR/agarose gel visualization techniques, as shown by the number of triple and quadruple co-infections detected with these techniques (Table 3). Thus, attempting to corroborate these methods with the 3 PCR-based methods used was not completely successful. Nevertheless, most of the positive results from these tests were verified by comparing the microarray and Adenovirus Consensus kit results or by comparison with the results from independent methods such as microneutralization, hexon sequence analysis, serotype-specific PCR that uses primers not used in the multiplex tests, and PCR amplicon sequencing (Table 1, Table 2, and Table 4). These results suggest that these methods can identify and corroborate HAdV co-infections and that, in general, the HAdV load in ARD patients is more complex than previously thought.
To determine whether >1 replication-competent serotype or strain was present in the samples with evidence of co-infection, we attempted to physically separate the paired co-infectants in sample 7151 by plaque purification. Of 92 plaques picked from the initial plate, all tested positive for HAdV-C by PCR and 12 of 92 also tested positive for HAdV-B. Several of the plaques that retained both HAdV-B and HAdV-C signals were replaqued, and PCR testing of these plaques yielded only HAdV-C isolates. Further efforts that used a detergent to increase separation within the original ITCF sample 7151 and applied the agarose overlay more quickly (6 hours) to prevent interplaque contamination also yielded only HAdV-C plaques (data not shown).
We demonstrate the rarely reported phenomenon of co-infections with multiple adenoviral species. Two previous studies have noted rare instances of HAdV-C dual infections in small numbers (27,28). HAdV-C, although rarely associated with pharyngitis outbreaks in recruits (10), is usually seen in children (7,8) and can produce latent infections that last into young adulthood. This fact, combined with low incidence of co-infection (27,28), has led to the assertion that multistrain adenovirus co-infections are not common (28) or clinically relevant. The results from the population tested in this study suggest otherwise. Samples from vaccinated recruits showed a high rate of co-infection with multiple species of adenovirus associated with adult ARD (HAdV-E and HAdV-B1).
Many of the identified co-infectants in this study were species not generally associated with ARD in the military (HAdV-C, HAdV-B2, and HAdV-F). Although these species were not likely the cause of ARD observed in these patients, since they are not believed to cause ARD in adults and because they have a high potential for latent carriage (1,7,8,29), their presence sheds new light on the general complexity of the human adenoviral load. In addition, they remain viable reservoirs capable of genetic complementation or recombination with upper respiratory strains.
Recombination can generate new strains with unique and stable phenotypes. Intraspecies adenovirus recombination has been demonstrated in laboratory cell-culture co-infection studies (30–32). These recombination events can generate viable hybrids with intermediate or unique immunogenic and tropic properties. Evidence suggests recombination can generate hybrids in immunocompromised patients (29,33,34), possibly as a result of co-infection with normally isolated serotypes. Recombination, particularly intraspecies, seems to play a major role in the evolution of new, virulent strains of HAdV (1,17,35,36). The currently dominant pathogenic HAdV in US military recruits, a considerably diverged variant HAdV-4 strain (16), appears to be a recent recombinant between HAdV-4 and a HAdV-B1 serotype, probably HAdV-7 (37). Given that these 2 are the most common co-infectants seen in our sample set, this finding suggests that the observed dominance of co-infections in vaccinated persons may have contributed to the emergence of the new variant. In general, the understanding and control of situations that create or promote co-infection may be important considerations.
The HAdV vaccine, an enteric-coated live-virus tablet designed to transiently and selectively infect the gastrointestinal tract with normal respiratory HAdV strains, contains viable HAdV-4 and HAdV-7. Thus, we cannot assume whether the detected co-infectants arose from the vaccine itself or from community acquisition of circulating strains. Most HAdV-4 strains in this study are not the vaccine strain but rather a highly divergent variant that has recently been dominant in military training centers (GenBank strain Z-G 95-873). This identity was shown by sequence analysis of 1,500 bp of the hexon gene from many primary infectants identified in the same sample set that was analyzed here (16). The variant HAdV-4 isolates consistently differ from the vaccine strain by 32 base substitutions, including 9 coding changes, in this region (16) (Table 1). Hexon sequence analysis showed that many HAdV-7 co-infectants are HAdV-7d2. HAdV-7d2 is distinguished from the HAdV-7 vaccine strain (HAdV-7a) by a single coding polymorphism in the hexon sequence, but this polymorphism (protein L443Q or nucleotide T1328A in GenBank [16]) is specific to HAdV-7d and HAdV-7d2 and is not found in HAdV-7a, b, c, g, or h or in the vaccine strain (16,38,39). Three of the other HAdV-7 co-infectants (1856, 4185, and 7372) were shown to be HAdV-7h by fiber gene sequencing. The fiber gene of HAdV-7h appears to have been horizontally transferred from HAdV-3 and thus is highly diverged from the usual HAdV-7 fiber gene, as found in the vaccine strain (17). Thus, sequence analyses show that most, if not all, co-infectants are currently circulating HAdV-4 and HAdV-7 strains that are distinct from the vaccine strains (16) (Table 1).
Four lines of evidence support the idea that most of the apparent genetic complexity in the throat swab samples comes from multiple strains, as opposed to recombinants with mixed genetic characteristics. The first comes from the microarray data. The microarray tests for hybridization of 6 independent probes designed to match serotype-specific sequences in 3 genes (18). Since different species do not cross-react among the microarray probes, hybridization of genes from 1 species to the identifying probes for 2 species would require redundant presence of 2 different alleles in all 3 genes. Since both natural recombination in hosts (17) and artificially encouraged recombination in cell culture (30,32) strongly favor homologous recombination and generation of nonredundant hybrid strains, redundant characterization of paired, divergent alleles is inconsistent with a single recombinant genome.
The second line of evidence supporting co-infection with independent genomes comes from comparisons of relative co-infectant titers before and after potentially selective events, such as growth in tissue culture. PCR amplification of fiber gene sequences using species B- and E-specific primers was performed on serial dilutions of vaccinated sample 7274 before and after passage of the original ITCF through 2 additional cycles of growth in A549 cells. In this instance, the relative titers of HAdV-4 and HAdV-7, as measured by serial-dilution PCR, changed by 2 orders of magnitude (data not shown). The rapid drift in relative concentrations of PCR targets from paired co-infecting strains strongly suggests that the co-infectants' genomes are replicating independently and thus likely to be physically separate entities.
The third line of evidence supporting co-infectant independence comes from whole-genome sequencing efforts. Several molecular methods indicated that vaccinated sample 7151 harbored an HAdV-5/HAdV-21 co-infection (Table 1 and Table 2). The genome of the HAdV-5 co-infecting strain was sequenced and assembled into a contiguous sequence (GenBank no. AY601635) consistent with a published HAdV-5 genome (GenBank accession no. AY339865) (40), which suggested no recombination of foreign DNA. However, this effort also generated several orphan sequences that did not fit into the assembled sequence and were subsequently identified as genetically redundant HAdV-21 regions. Further amplification and sequencing of several genetically distant fragments from the same sample using HAdV-21-specific primers yielded ≈2 kb of HAdV-21 sequence. On the basis of the entire genome and partial PCR sequencing analyses, >2 co-infecting HAdV genomes are contained in sample 7151.
The fourth line of evidence comes from our attempts to physically separate paired co-infectants by plaque purification. Sample 7151, which contained the HAdV-5/HAdV-21 co-infection, was used initially because it contained relatively equal titers of both co-infectants. Although most of the plaques tested contained HAdV-5, some contained both HAdV-5 and HAdV-21. Although we were unable to identify plaques that contained only HAdV-21, our results demonstrate the physical independence of the co-infecting entities and the functional independence of HAdV-5. Our results also suggest that either the HAdV-21 co-infectant is functionally dependent on HAdV-5 or is effectively outgrown by HAdV-5 to a degree that prevents independent isolation. Similar attempts were made with a few samples that had HAdV-4/HAdV-7 co-infections, but these were generally biased in titer (104 in favor of HAdV-7) and, as expected, yielded only HAdV-7 in >300 plaques tested. The data demonstrated the functional independence of 1 co-infectant (HAdV-7) and physical independence of the co-infecting entities but could not conclusively demonstrate functional independence of the minor co-infectant.
Conventional clinical microbiologic methods, including microneutralization and hemagglutination inhibition, are comparative and designed to identify the primary HAdV serotype (or species) in a sample. Secondary infections are masked in these methods by the tests (e.g., microneutralization is reported as the strongest reaction, not the spectrum of reactions across all serotypes). Likewise, direct sequencing (16) may restrict identification to a single strain, particularly if 1 co-infectant is dominant. Restriction enzyme analysis methods are capable of resolving HAdV-C dual infections in which both serotypes are present in similar numbers (27). In contrast, when using sensitive molecular methods that can yield measurable signals from secondary (less numerous) co-infectants against the background of stronger signals produced by primary infecting strains, these methods may identify co-infections more than do conventional methods. In the case of respiratory infections, this finding has previously been documented (41).
Finally, each of the methods designed to test for multiple species or serotypes showed a higher number of HAdV (and accepted virulent HAdV species and serotypes) in vaccinated persons than in unvaccinated persons. HAdV vaccine was administered routinely to all trainees until supplies were exhausted, at which point adenovirus vaccination was stopped. Since trainees were vaccinated systematically, persons tended to be sampled at times when either all or no recruits were being vaccinated. Therefore, vaccinated samples collected and tested (from 1996 to 1998) are not concurrent with unvaccinated samples (collected from 1998 to 2000). Because of this sampling limitation, we could not confidently correlate HAdV co-infection with breakthrough infections in previously vaccinated persons. Thus, although this study highlights the previously underappreciated phenomenon of adenoviral co-infection, the conclusive examination of its relationship to vaccination must await reintroduction of HAdV vaccine (15).
Dr Vora is a research biologist at the Center for Bio/Molecular Science and Engineering, Naval Research Laboratory, Washington, DC. His primary research interest is development of microarray-based molecular diagnostics for human pathogenic microorganisms.
Acknowledgments
We thank Gregory C. Gray and Dan Blasiole for critical preceding work, including collection and initial analysis of strains that we reanalyzed, and Beth Metzgar and Valerie de Crecy-Lagard for critically reading the manuscript.
Partial support was provided by the Epidemic Outbreak Surveillance Consortium2 funded by the US Air Force Surgeon General Office, the Modernization Directorate, the Defense Threat Reduction Agency, and the Office of Naval Research.
References
- Rubin BA. Clinical picture and epidemiology of adenovirus infections. Acta Microbiol Hung. 1993;40:303–23.PubMedGoogle Scholar
- Hierholzer JC, Stone YO, Broderson JR. Antigenic relationships among the 47 human adenoviruses determined in reference horse antisera. Arch Virol. 1991;121:179–97. DOIPubMedGoogle Scholar
- Wadell G. Molecular epidemiology of human adenoviruses. Curr Top Microbiol Immunol. 1984;110:191–220.PubMedGoogle Scholar
- Rosen L. A hemagglutination-inhibition technique for typing adenoviruses. Am J Hyg. 1960;71:120–8.PubMedGoogle Scholar
- Hierholzer JC. Further subgrouping of the human adenoviruses by differential hemagglutination. J Infect Dis. 1973;128:541–50. DOIPubMedGoogle Scholar
- Schmitz H, Wigand R, Heinrich W. Worldwide epidemiology of human adenovirus infections. Am J Epidemiol. 1983;117:455–66.PubMedGoogle Scholar
- Brandt CD, Kim HW, Vargosko AJ, Jeffries BC, Arrobio JO, Rindge B, Infections in 18,000 infants and children in a controlled study of respiratory tract disease. I. Adenovirus pathogenicity in relation to serologic type and illness syndrome. Am J Epidemiol. 1969;90:484–500.PubMedGoogle Scholar
- Fox JP, Hall CE, Cooney MK. The Seattle Virus Watch. VII. Observations of adenovirus infections. Am J Epidemiol. 1977;105:362–86.PubMedGoogle Scholar
- Takafuji ET, Gaydos JC, Allen RG, Top FH Jr. Simultaneous administration of live, enteric-coated adenovirus types 4, 7 and 21 vaccines: safety and immunogenicity. J Infect Dis. 1979;140:48–53. DOIPubMedGoogle Scholar
- Van der veen J. The role of adenoviruses in respiratory disease. Am Rev Respir Dis. 1963;88:167–80.PubMedGoogle Scholar
- Gooch WM III, Mogabgab WJ. Simultaneous oral administration of live adenovirus types 4 and 7 vaccines: protection and lack of emergence of other types. Arch Environ Health. 1972;25:388–94.PubMedGoogle Scholar
- Griffin JP, Greenberg BH. Live and inactivated adenovirus vaccines: clinical evaluation of efficacy in prevention of acute respiratory disease. Arch Intern Med. 1970;125:981–6. DOIPubMedGoogle Scholar
- Top FH Jr, Dudding BA, Russell PK, Buescher EL. Control of respiratory disease in recruits with types 4 and 7 adenovirus vaccines. Am J Epidemiol. 1971;94:142–6.PubMedGoogle Scholar
- Gray GC, Goswami PR, Malasig MD, Hawksworth AW, Trump DH, Ryan MA, Adult adenovirus infections: loss of orphaned vaccines precipitates military respiratory disease epidemics. For the Adenovirus Surveillance Group. Clin Infect Dis. 2000;31:663–70. DOIPubMedGoogle Scholar
- Gaydos CA, Gaydos JC. Adenovirus vaccines. In: Plotkin SA, Orenstein WA, Offit PA, editors. Vaccines. Fourth ed. Philadelphia: W.B. Saunders; 2003, p. 863–85.
- Blasiole DA, Metzgar D, Daum LT, Ryan MA, Wu J, Wills C, Molecular analysis of adenovirus isolates from vaccinated and unvaccinated young adults. J Clin Microbiol. 2004;42:1686–93. DOIPubMedGoogle Scholar
- Kajon AE, Wadell G. Sequence analysis of the E3 region and fiber gene of human adenovirus genome type 7h. Virology. 1996;215:190–6. DOIPubMedGoogle Scholar
- Lin B, Vora GJ, Thach D, Walter E, Metzgar D, Tibbetts C, Use of oligonucleotide microarrays for rapid detection and serotyping of acute respiratory disease-associated adenoviruses. J Clin Microbiol. 2004;42:3232–9. DOIPubMedGoogle Scholar
- Vabret A, Gouarin S, Joannes M, Barranger C, Petitjean J, Corbet S, Development of a PCR-and hybridization-based assay (PCR adenovirus consensus) for the detection and the species identification of adenoviruses in respiratory specimens. J Clin Virol. 2004;31:116–22. DOIPubMedGoogle Scholar
- Xu W, McDonough MC, Erdman DD. Species-specific identification of human adenoviruses by a multiplex PCR assay. J Clin Microbiol. 2000;38:4114–20.PubMedGoogle Scholar
- Crawford-Miksza LK, Nang RN, Schnurr DP. Strain variation in adenovirus serotypes 4 and 7a causing acute respiratory disease. J Clin Microbiol. 1999;37:1107–12.PubMedGoogle Scholar
- Malasig MD, Goswami PR, Crawford-Miksza LK, Schnurr DP, Gray GC. Simplified microneutralization test for serotyping adenovirus isolates. J Clin Microbiol. 2001;39:2984–6. DOIPubMedGoogle Scholar
- Xu W, Erdman DD. Type-specific identification of human adenovirus 3, 7, and 21 by a multiplex PCR assay. J Med Virol. 2001;64:537–42. DOIPubMedGoogle Scholar
- Pring-Akerblom P, Trijssenaar FE, Adrian T, Hoyer H. Multiplex polymerase chain reaction for subgenus-specific detection of human adenoviruses in clinical samples. J Med Virol. 1999;58:87–92. DOIPubMedGoogle Scholar
- Mei YF, Wadell G. Hemagglutination properties and nucleotide sequence analysis of the fiber gene of adenovirus genome types 11p and 11a. Virology. 1993;194:453–62. DOIPubMedGoogle Scholar
- Basler CF, Horwitz MS. Subgroup B adenovirus type 35 early region 3 mRNAs differ from those of the subgroup C adenoviruses. Virology. 1996;215:165–77. DOIPubMedGoogle Scholar
- Fife KH, Ashley R, Shields AF, Salter D, Meyers JD, Corey L. Comparison of neutralization and DNA restriction enzyme methods for typing clinical isolates of human adenovirus. J Clin Microbiol. 1985;22:95–100.PubMedGoogle Scholar
- Adhikary AK, Inada T, Banik U, Numaga J, Okabe N. Identification of subgenus C adenoviruses by fiber-based multiplex PCR. J Clin Microbiol. 2004;42:670–3. DOIPubMedGoogle Scholar
- Hierholzer JC. Adenoviruses in the immunocompromised host. Clin Microbiol Rev. 1992;5:262–74.PubMedGoogle Scholar
- Meinschad C, Winnacker EL. Recombination in adenovirus. I. Analysis of recombinant viruses under non-selective conditions. J Gen Virol. 1980;48:219–24. DOIPubMedGoogle Scholar
- Boursnell ME, Mautner V. Recombination in adenovirus: crossover sites in intertypic recombinants are located in regions of homology. Virology. 1981;112:198–209. DOIPubMedGoogle Scholar
- Sambrook J, Williams J, Sharp PA, Grodzicker T. Physical mapping of temperature-sensitive mutations of adenoviruses. J Mol Biol. 1975;97:369–90. DOIPubMedGoogle Scholar
- de Jong PJ, Valderrama G, Spigland I, Horwitz MS. Adenovirus isolates from urine of patients with acquired immunodeficiency syndrome. Lancet. 1983;1:1293–6. DOIPubMedGoogle Scholar
- Kojaoghlanian T, Flomenberg P, Horwitz MS. The impact of adenovirus infection on the immunocompromised host. Rev Med Virol. 2003;13:155–71. DOIPubMedGoogle Scholar
- Davison AJ, Benko M, Harrach B. Genetic content and evolution of adenoviruses. J Gen Virol. 2003;84:2895–908. DOIPubMedGoogle Scholar
- Stone D, Furthmann A, Sandig V, Lieber A. The complete nucleotide sequence, genome organization, and origin of human adenovirus type 11. Virology. 2003;309:152–65. DOIPubMedGoogle Scholar
- Houng HH, Clavio S, Graham K, Kuschner R, Sun W, Russell KL, Emergence of a new human adenovirus type 4 (Ad4) genotype: identification of a novel inverted terminal repeated (ITR) sequence from a majority of Ad4 isolates from US military recruits. J Clin Virol. 2006;35:381–7. DOIPubMedGoogle Scholar
- Erdman DD, Xu W, Gerber SI, Gray GC, Schnurr D, Kajon AE, Molecular epidemiology of adenovirus type 7 in the United States, 1966–2000. Emerg Infect Dis. 2002;8:269–77. DOIPubMedGoogle Scholar
- Li Q, Wadell G. Genetic variability of hexon loops 1 and 2 between seven genome types of adenovirus serotype 7. Arch Virol. 1999;144:1739–49. DOIPubMedGoogle Scholar
- Sugarman BJ, Hutchins BM, McAllister DL, Lu F, Thomas BK. The complete nucleic acid sequence of the adenovirus type 5 reference material (ARM) genome. Bioprocessing Journal. 2003;2:27–32.
- Mitchell S, O'Neill HJ, Ong GM, Christie S, Duprex P, Wyatt DE, Clinical assessment of a generic DNA amplification assay for the identification of respiratory adenovirus infections. J Clin Virol. 2003;26:331–8. DOIPubMedGoogle Scholar
Figure
Tables
Cite This Article1These authors contributed equally to this article.
2The members of the Epidemic Outbreak Surveillance Consortium are Peter F. Demitry, Theresa Lynn Difato, Robb K. Rowley, Clark Tibbetts, Eric H. Hanson, Rosana R. Holliday, Curtis White, David A. Stenger, Donald Seto, Elizabeth A. Walter, Jerry Diao, Brian K. Agan, Kevin Russell, David Metzgar, Gary J.Vora, Baochuan Lin, Dzung Thach, Jing Su, Chris Olsen, Dong Xia, John Gomez, John McGraw, Linda Canas, Margaret Jesse, Mi Ha Yuen, Robert Crawford, Sue A. Worthy, Sue Ditty, John McGraw, Michael Jenkins, Zheng Wang, Cheryl J. James, Kathy Ward, Kenya Grant, and Kindra Nix.
Table of Contents – Volume 12, Number 6—June 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
David Metzgar, Naval Health Research Center, PO Box 85122, San Diego, CA 92186-5122, USA
Top