Volume 13, Number 8—August 2007
Letter
Mycobacterium cosmeticum, Ohio and Venezuela
Cite This Article
Citation for Media
To the Editor: Mycobacterium cosmeticum is a rapidly growing nontuberculous mycobacteria species that was first described in November 2004. The first strains were obtained from cultures of a sink drain in a nail salon in Atlanta, Georgia, USA, and from a granulomatous lesion of a female mesotherapy patient in Venezuela (1).
Among 3 additional isolates of M. cosmeticum obtained from July 2003 through November 2004, one was obtained from a 77-year-old man who was admitted to Ohio hospital A on September 22, 2004, with fever, exacerbation of chronic obstructive pulmonary disease, and urosepsis. Underlying medical conditions included diabetes, discitis, hyperlipidemia, coronary artery disease, and coal worker’s pneumoconiosis. He had received intravenous antimicrobial agents (rifampin and daptomycin) through a Groshong catheter that had been inserted to treat discitis. A routine blood culture was performed according to standard methods (2), and the catheter was removed. A diagnosis of catheter-associated bacteremia (CAB) was made, but the patient’s overall condition improved without antibacterial drug therapy, and he was discharged 4 days after admission. The culture specimen yielded only mycobacteria and was sent on to ARUP Laboratories, where it was identified as M. cosmeticum by 16S rDNA sequence analysis. The isolate was then sent to the Centers for Disease Control and Prevention (CDC) Mycobacteriology Laboratory Branch (Atlanta, GA, USA) and designated OH1.
A 43-year-old woman with a diagnosis of non-Hodgkin lymphoma, who had received regular central venous catheterizations, was admitted to Ohio hospital B on August 20, 2004. A left subclavian catheter was inserted, and a routine blood specimen for culture was subsequently obtained on the day of admission. Before admission, the woman had been receiving acyclovir and cefepime. She received chemotherapeutic agent injections, platelet infusion, and an autologous stem cell transplant 6 days after admission. The blood culture was positive only for rapidly growing mycobacteria, and the final diagnosis was CAB. However, no symptoms of infection were observed, and no antimycobacterial drug therapy was administered. She was discharged without complications after the transplant was received and the catheter removed. The bacterial isolate was forwarded to CDC’s Special Bacteriology Reference Laboratory, where it was identified as M. cosmeticum by 16S rDNA sequence analysis, sent on to the CDC Mycobacteriology Laboratory Branch, and designated OH2.
A 36-year-old man with AIDS was admitted to hospital C in Caracas, Venezuela, in June 2003 with dyspnea, fever, and expectoration. A sputum sample was positive by acid-fast bacillus smear and culture, yielding both M. cosmeticum (designated VZ1) and M. scrofulaceum on Middlebrook 7H10 agar (Remel Co., Lenexa, KS, USA). At the time the sputum was obtained, the patient was receiving only trimethoprim-sulfamethoxazole, but he experienced respiratory arrest and died ≈6 weeks later.
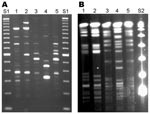
The 3 isolates were confirmed to be M. cosmeticum by high-performance liquid chromatography mycolate analyses and by PCR restriction analysis of a 440-bp segment of hsp65 (1). The relationship of these isolates to the only documented strains of M. cosmeticum was evaluated by analysis of large restriction fragments with pulsed-field gel electrophoresis (1) and by repetitive element PCR (3). Banding patterns for isolates OH1 and OH2 were different from one another as well as from isolate VZ1 and the 2 control strains. Typing patterns for isolate VZ1, however, matched the control strain from Venezuela (ATCC BAA-878T), which indicates that these 2 isolates are likely a common strain (Figure).
Of the >125 recognized Mycobacterium species, ≈50 are etiologic agents of human disease (4). The type strain of M. cosmeticum (ATCC BAA-878T) was associated with a soft-tissue infection in which the source was postulated to be environmental contamination of an unknown substance administered to the patient by injection as part of a weight loss regimen. This strain and isolate VZ1 were isolated from clinics in Caracas, Venezuela; both were found to be a common strain, but no other factors suggest that these represent an epidemic cluster. Although the Venezuelan patient from whom isolate VZ1 was obtained exhibited symptoms consistent with mycobacterial pulmonary disease, M. cosmeticum involvement cannot be proven because an additional Mycobacterium species, M. scrofulaceum, was isolated from the patient’s sputum. Because each of these organisms is found in the aqueous environment, they may represent colonization or may have been transiently present in the patient. Nonetheless, additional nontuberculous mycobacteria species have been reported to cause pulmonary disease, and the involvement of M. cosmeticum in this case cannot be excluded.
Successful treatment of CAB infections caused by rapidly growing mycobacteria has most often been achieved by removing the catheter with or without the use of antimicrobial drug therapy (4). Criteria to support a true bloodstream infection were met by one of the patients in Ohio. These criteria include the absence of a source for bacteremia alternative to M. cosmeticum OH1 and the resolution of the febrile syndrome after removal of the device. The second patient had no symptoms when the blood culture was obtained; thus, the clinical significance of M. cosmeticum in this case is unclear.
When all identified strains of M. cosmeticum are considered, this species is clearly present in diverse geographic regions and in healthcare institutions. These findings suggest that it may be widely distributed in the environment and should be regarded, along with other rapidly growing mycobacteria species, as a potential pathogen.
Acknowledgment
We thank Lena Fischer of the Ohio Department of Health for technical contributions.
References
- Cooksey RC, de Waard JH, Yakrus MA, Rivera I, Chopite M, Toney SR, Mycobacterium cosmeticum sp. nov., a novel rapidly growing species isolated from a cosmetic infection and from a nail salon. Int J Syst Evol Microbiol. 2004;54:2385–91. DOIPubMedGoogle Scholar
- Pfyffer GE, Brown-Elliott BA, Wallace RJ Jr. Mycobacterium: general characteristics, isolation, and staining procedures. In: Murray PR, Baron EJ, Pfaller MA, Jorgensen JH, Yolken RH, editors. Manual of clinical microbiology. Washington: ASM Press; 2003. p. 532–59.
- Dombek PE, Johnson LK, Zimmerley ST, Sadowsky MJ. Use of repetitive DNA sequences and the PCR to differentiate Escherichia coli isolates from human and animal sources. Appl Environ Microbiol. 2000;66:2572–7. DOIPubMedGoogle Scholar
- Wagner D, Young LS. Nontuberculous mycobacterial infections: a clinical review. Infection. 2004;32:257–70. DOIPubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 13, Number 8—August 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Robert C. Cooksey, Division of Tuberculosis Elimination, Centers for Disease Control and Prevention, 1600 Clifton Rd NE, Mailstop F08, Atlanta, GA 30333, USA;
Top