Volume 14, Number 7—July 2008
Dispatch
AIDS Patient Death Caused by Novel Cryptococcus neoformans × C. gattii Hybrid
Cite This Article
Citation for Media
Abstract
Interspecies hybrids of Cryptococcus neoformans and C. gattii have only recently been reported. We describe a novel C. neoformans × C. gattii hybrid strain (serotype AB) that was previously described as C. gattii and that caused a lethal infection in an AIDS patient from Canada.
Cryptococcus neoformans and C. gattii are pathogenic yeasts that may cause meningoencephalitis. C. neoformans primarily infects immunocompromised patients and occurs worldwide, whereas C. gattii primarily mainly infects otherwise healthy persons and has been thought to occur in subtropical regions (1–3). However, the recent outbreak of infection with C. gattii on Vancouver Island, British Columbia, Canada (4), expansion of this outbreak to mainland Canada and the Pacific Northwest region of the United States (5), and identification of C. gattii isolates in Europe (6) show that C. gattii can also occur in temperate climates. Molecular techniques can distinguish 7 haploid genotypic groups within C. neoformans and C. gattii (7–9; F. Hagen and T. Boekhout, unpub. data).
Recently, 3 serotype BD C. neoformans var. neoformans × C. gattii hybrids were isolated from 2 HIV-negative patients in the Netherlands (10). We describe a novel C. neoformans var. grubii serotype A × C. gattii serotype B hybrid that was isolated from an HIV-positive person.
Strain CBS10496 was isolated from a 31-year-old AIDS patient from Montreal, Quebec, Canada, who had traveled to Mexico ≈15 months before cryptococcosis was diagnosed. The patient died despite extensive antifungal treatment with ketoconazole and amphotericin B (11). CBS10496 has been identified as C. gattii serotype B (cited as C. neoformans var. gattii) (11). Reference isolates used in this study are listed in the Table.
The ploidy of CBS10496 was determined by using flow cytometry (10) with the sequenced haploid strains CBS8710 and CBS10510 as references. Nuclei were visualized after staining with 4′,6-diamidino-2-phenylindole (10). Coloration of colonies grown on canavanine-glycine-bromthymol blue (CGB) medium (12) was determined after incubation at 24°C for 6 and 15 days. The serotype of CBS10496 was determined by using the CryptoCheck serotyping kit (Iatron Laboratories, Tokyo, Japan).
Ten colonies of CBS10496 were used for DNA extraction (10). DNA of these colonies was used for amplified fragment length polymorphism (AFLP) analysis (7). The partial sequence of 6 nuclear regions was determined for reference isolates CBS10488–CBS10490, CBS1622, CBS6992, and the putative hybrid isolate CBS10496. Selected nuclear regions were those for internal transcribed spacer (ITS) region, intergenic spacer region, laccase (CNLAC1), 2 RNA polymerase II subunits (RPB1 and RPB2), and translation elongation factor 1α (TEF1α) (9,10). Mating types and serotype were determined as described (10,13).
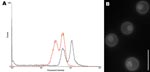
DNA content of CBS10496 was compared with that of CBS8710 and CBS10510. The G1 peak of reference strains was located at positions 31.6 (CBS8710) and 31.1 (CBS10510), and the G2 peak was located at positions 65.8 (CBS8710) and 56.4 (CBS10510). The G1 peak of CBS10496 was located at position 57.5, and the G2 peak was located at position 115.7. Thus, the G1 peak of CBS10496 coincided with the G2 peak of the haploid strains (Figure 1, panel A), which indicates that CBS10496 has ≈2× more DNA than haploid strains. We concluded that CBS10496 is diploid or aneuploid. Staining with 4′,6-diamidino-2-phenylindole showed that cells of CBS10496 were monokaryotic (Figure 1, panel B).
Reaction of CBS10496 on CGB medium was negative, which corresponds to C. neoformans (12). The CryptoCheck serotyping kit serum factors 5 (corresponding to serotype B) and 7 (corresponding to serotype A) agglutinated, which indicated that CBS10496 is a serotype AB strain.
The AFLP fingerprint obtained by analysis of colonies of CBS10496 did not match any of the previously defined AFLP genotypes. The fingerprint of CBS10496 was compared with AFLP fingerprints of reference strains CBS8710 and CBS9172, which are AFLP1/VNI, and E566 and CBS10510, which are AFLP4/VGI. The AFLP fingerprint of CBS10496 contained fragments characteristic of AFLP1/VNI and AFLP4/VGI (Figure 2), which indicated that genetic material from these 2 genotypes was present in this isolate.
Two alleles representing AFLP1/VNI and AFLP4/VGI were found when fragments of RPB1, RPB2, CNLAC1, and intergenic spacer region of CBS10496 were cloned and sequenced. However, after 30 clones were sequenced, only 1 allele was obtained for TEF1α, i.e., AFLP4/VGI, and ITS, i.e., AFLP1/VNI. Our results indicate that genetic material from AFLP1/VNI and AFLP4/VGI was present in CBS10496, although only 1 allele was obtained for TEF1α and ITS. All sequences were submitted to GenBank (accession nos. DQ286656–DQ286676 and EF102027–EF102072).
Amplification of CBS10496 in a PCR with the MATα and the MATα serotype A–specific primer pair resulted in an amplicon. When MATa and the MATa serotype A–specific PCRs were conducted, no amplicon was obtained. These findings indicate that CBS10496 has a MATα serotype A background. All reference strains yielded amplicons with the expected primer pairs. In addition, CBS10510, a MATα serotype B strain, was amplified with the MATα specific primer pair, and E566, a MATa serotype B strain, yielded an amplicon with the MATa-specific PCR. These results indicate that a C. gattii and a MATα serotype A background are present in CBS10496. Because the mating type of the C. gattii background within CBS10496 was unknown, 30 MATα clones of CBS10496 were sequenced to determine whether a MATα serotype B allele could be identified. However, all clones were MATα serotype A; no MATα serotype B clones were found.
Our results indicated that CBS10496 is a monokaryotic, diploid, or aneuploid strain with the novel AB serotype. AFLP and sequence analysis showed that the isolate contained fragments of C. neoformans var. grubii (AFLP1/VNI) and C. gattii (AFLP4/VGI). We conclude that this isolate is a novel aneuploid hybrid of C. neoformans var. grubii (serotype A, AFLP1/VNI) and C. gattii (serotype B, AFLP4/VGI).
CBS10496 had been identified as C. gattii on the basis of a weak positive reaction on CGB medium (11). Our results indicated that CBS10496 was negative on CGB medium. Although a negative response on CGB medium has been shown for other C. neoformans × C. gattii hybrids (10,14), weak and delayed positive reactions on CGB medium may occur in C. neoformans × C. gattii hybrid isolates (10,14). CBS10496 was previously identified as a serotype B strain (11). Inconsistent serotyping results have been reported for other hybrids (10,15) and may result from differences in specificity and potency among different batches of factor serum. All C. neoformans × C. gattii hybrids discovered have originated from clinical sources (13; F. Hagen and T. Boekhout, unpub. data).
We expected that CBS10496 would have 2 mating-type loci. However, only a serotype A MATα background was observed. Although an amplicon was obtained with C. gattii–specific mating-type primers, the C. gattii background could not be linked to a mating type. We hypothesize that the serotype AB C. neoformans × C. gattii hybrid CBS10496 was formed by mating of a MATa serotype B strain with a MATα serotype A strain and subsequent loss of the MATa serotype B allele. Detection of single ITS and TEF1α alleles in CBS10496 further supports our findings because it indicates that other alleles were also lost. Loss of genetic material has been observed in other hybrids, such as serotype AD and BD hybrids (14), and seems to be a normal process in cryptococcal hybrids.
Our results show that the C. gattii parent of the serotype AB hybrid belongs to the AFLP4/VGI genotype, as was the case for serotype BD hybrids (10). The C. gattii parental sequence of all known serotype BD C. neoformans × C. gattii hybrid isolates was identical to sequences of AFLP4/VGI strains CBS1622 and CBS6992 in all regions studied (9). Detection of 1 specific C. gattii–AFLP4/VGI subgroup in all isolated C. neoformans × C. gattii hybrids may indicate that this subgroup preferentially forms interspecies hybrids.
Dr Bovers participated in this study while conducting doctoral research at the Centraalbureau voor Schimmelcultures–Fungal Biodiversity Centre in Utrecht. She is currently working at the Netherlands Commission on Genetic Modification in Bilthoven. Her research interests include the epidemiology and phylogeny of pathogenic yeasts.
Acknowledgment
The research of M.B. and F.H. is supported by the Odo van Vloten Foundation. E.E.K. is supported by the Renewal Fund of the Royal Netherlands Academy of Arts and Sciences.
References
- Speed B, Dunt D. Clinical and host differences between infections with the two varieties of Cryptococcus neoformans. Clin Infect Dis. 1995;21:28–34.PubMedGoogle Scholar
- Sorrell TC. Cryptococcus neoformans variety gattii. Med Mycol. 2001;39:155–68. DOIPubMedGoogle Scholar
- Kwon-Chung KJ, Bennett JE. Epidemiological differences between the two varieties of Cryptococcus neoformans. Am J Epidemiol. 1984;120:123–30.PubMedGoogle Scholar
- Kidd SE, Hagen F, Tscharke RL, Huynh M, Bartlett KH, Fyfe M, A rare genotype of Cryptococcus gattii caused the cryptococcosis outbreak on Vancouver Island (British Columbia, Canada). Proc Natl Acad Sci U S A. 2004;101:17258–63. DOIPubMedGoogle Scholar
- MacDougall L, Kidd SE, Galanis E, Mak S, Leslie MJ, Cieslak PR, Spread of Cryptococcus gattii in British Columbia, Canada, and detection in the Pacific Northwest, USA. Emerg Infect Dis. 2007;13:42–50.PubMedGoogle Scholar
- Viviani MA, Cogliati M, Esposto MC, Lemmer K, Tintelnot K, Colom Valiente MF, European Confederation of Medical Mycology (ECMM) Cryptococcosis Working Group. Molecular analysis of 311 Cryptococcus neoformans isolates from a 30-month ECMM survey of cryptococcosis in Europe. FEMS Yeast Res. 2006;6:614–9. DOIPubMedGoogle Scholar
- Boekhout T, Theelen B, Diaz M, Fell JW, Hop WC, Abeln EC, Hybrid genotypes in the pathogenic yeast Cryptococcus neoformans. Microbiology. 2001;147:891–907.PubMedGoogle Scholar
- Meyer W, Castañeda A, Jackson S, Huynh M, Castañeda E. the IberoAmerican Cryptococcal Study Group. Molecular typing of IberoAmerican Cryptococcus neoformans isolates. Emerg Infect Dis. 2003;9:189–95.PubMedGoogle Scholar
- Bovers M, Hagen F, Kuramae EE, Boekhout T. Six monophyletic lineages identified within Cryptococcus neoformans and Cryptococcus gattii by multi-locus sequence typing. Fungal Genet Biol. 2007;45:400–21. DOIPubMedGoogle Scholar
- Bovers M, Hagen F, Kuramae EE, Diaz MR, Spanjaard L, Dromer F, Unique hybrids between fungal pathogens Cryptococcus neoformans and Cryptococcus gattii. FEMS Yeast Res. 2006;6:599–607. DOIPubMedGoogle Scholar
- St-Germain G, Noël G, Kwon-Chung KJ. Disseminated cryptococcosis due to Cryptococcus neoformans variety gattii in a Canadian patient with AIDS. Eur J Clin Microbiol Infect Dis. 1988;7:587–8. DOIPubMedGoogle Scholar
- Kwon-Chung KJ, Polacheck I, Bennett JE. Improved diagnostic medium for separation of Cryptococcus neoformans var. neoformans (serotypes A and D) and Cryptococcus neoformans var. gattii (serotypes B and C). J Clin Microbiol. 1982;15:535–7.PubMedGoogle Scholar
- Barreto de Oliveira MT, Boekhout T, Theelen B, Hagen F, Baroni FA, Lazera MS, Cryptococcus neoformans shows a remarkable genotypic diversity in Brazil. J Clin Microbiol. 2004;42:1356–9. DOIPubMedGoogle Scholar
- Kwon-Chung KJ, Varma A. Do major species concepts support one, two or more species within Cryptococcus neoformans? FEMS Yeast Res. 2006;6:574–87. DOIPubMedGoogle Scholar
- Viviani MA, Wen H, Roverselli A, Caldarelli-Stefano R, Cogliati M, Ferrante P, Identification by polymerase chain reaction fingerprinting of Cryptococcus neoformans serotype AD. J Med Vet Mycol. 1997;35:355–60. DOIPubMedGoogle Scholar
Figures
Table
Cite This Article1These authors contributed equally to this article.
2Current affiliation: Netherlands Commission on Genetic Modification, Bilthoven, the Netherlands.
Table of Contents – Volume 14, Number 7—July 2008
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Ferry Hagen, Centraalbureau voor Schimmelcultures–Fungal Biodiversity Center, Uppsalalaan 8, NL-3584CT Utrecht, the Netherlands;
Top