Volume 19, Number 10—October 2013
CME ACTIVITY - Research
Increased Incidence of Invasive Fusariosis with Cutaneous Portal of Entry, Brazil
Cite This Article
Citation for Media
Introduction

Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 70% minimum passing score and complete the evaluation at www.medscape.org/journal/eid; (4) view/print certificate.
Release date: September 18, 2013; Expiration date: September 18, 2014
Learning Objectives
Upon completion of this activity, participants will be able to:
• Distinguish the most common underlying diagnosis of patients with invasive fusariosis in the current study
• Analyze the clinical presentation of patients with invasive fusariosis
• Assess the laboratory diagnosis and management of invasive fusariosis
• Evaluate trends in the incidence of fusariosis.
CME Editor
Shannon O’Connor, ELS, Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: Shannon O’Connor has disclosed no relevant financial relationships.
CME Author
Charles P. Vega, MD, Health Sciences Clinical Professor; Residency Director, Department of Family Medicine, University of California, Irvine. Disclosure: Charles P. Vega, MD, has disclosed no relevant financial relationships.
Authors
Disclosures: Marcio Nucci, MD, has disclosed the following relevant financial relationships: served as an advisor or consultant for Astellas, Pfizer, Merck, and Gilead; served as a speaker or a member of a speakers bureau for Pfizer, Merck, and Gilead; received grants for clinical research from Pfizer and Merck. Andrea G. Varon; Marcia Garnica; Tiyomi Akiti; Gloria Barreiros; Beatriz Trope, MD, PhD; and Simone A. Nouér, MD, have disclosed no relevant financial relationships.
Abstract
Invasive fusariosis (IF) is an infection with Fusarium spp. fungi that primarily affects patients with hematologic malignancies and hematopoietic cell transplant recipients. A cutaneous portal of entry is occasionally reported. We reviewed all cases of IF in Brazil during 2000–2010, divided into 2 periods: 2000–2005 (period 1) and 2006–2010 (period 2). We calculated incidence rates of IF and of superficial infections with Fusarium spp. fungi identified in patients at a dermatology outpatient unit. IF incidence for periods 1 and 2 was 0.86 cases versus 10.23 cases per 1,000 admissions (p<0.001), respectively; superficial fusarial infection incidence was 7.23 versus 16.26 positive cultures per 1,000 superficial cultures (p<0.001), respectively. Of 21 cases of IF, 14 showed a primary cutaneous portal of entry. Further studies are needed to identify reservoirs of these fungi in the community and to implement preventive measures for patients at risk.
Invasive fusariosis (IF) is a mycosis caused by infection with Fusarium spp. fungi that affects primarily patients with hematologic malignancies and hematopoietic cell transplant (HCT) recipients (1,2). In these severely immunosuppressed patients, IF is typically disseminated and involves pneumonia, metastatic skin lesions, and positive blood cultures (3). The usual portal of entry is the airways, and IF is thought to be acquired by the inhalation of aerosols of fusarial conidia. However, the skin at sites of tissue breakdown may be a portal of entry (4). In a review of 232 published cases of IF in immunosuppressed patients, primary skin lesions represented the likely portal of entry in 16 (11%) of 147 patients with disseminated disease (5).
In 2007, we observed an increase in the incidence of IF in our hospital in Brazil: 5 cases of disseminated IF and 2 cases of locally invasive disease were diagnosed in a 7-month period. In addition to this apparent increase in incidence, we observed that all patients had a primary skin lesion on the lower limbs. All case-patients were housed in 4 rooms of the hematology unit. Because of the increasing incidence and the cutaneous primary lesions, environmental sampling of air, water, and water-related structures of the hematology unit were conducted; we also performed molecular analysis of patient and environmental isolates. We describe the incidence, clinical presentation, and outcome of these cases and compare these results with cases of superficial Fusarium spp. infection among outpatient dermatologic patients at the same hospital.
The University Hospital, Federal University of Rio de Janeiro, Rio de Janeiro, Brazil, is a tertiary-care teaching hospital with ≈450 beds and a hematology unit that has 5 double-bed rooms without high-efficiency particulate air filtration and 8 single-bed rooms equipped with filters and positive pressure. Cases of IF were identified during daily visits to the hematology ward, by review of a database of episodes of febrile neutropenia (active since 1986), and by review of the hospital’s mycology laboratory and pathology registries. Typically, the diagnosis of IF is confirmed by blood culture and/or by direct exam, culture, and histopathology of metastatic skin lesions, when present (3).
Cases of superficial infections caused by Fusarium spp. were identified by reviewing the Mycology Laboratory database of cultures of dermatologic patients, a database that contains description of the type of lesion from which direct examination and culture were performed, as well as the results of direct exam and culture. These infections were diagnosed in outpatients who attended the hospital’s dermatology clinic, and the diagnosis required the presence of a superficial lesion with positive culture of the lesion.
We reviewed the records of all patients in whom IF was diagnosed during 2000–2010, obtaining detailed information on demographics, underlying disease and treatment, comorbidities, presence of neutropenia, receipt of corticosteroids and other immunosuppressive agents, clinical manifestations of IF, diagnosis, treatment, and outcome. All patients had been hospitalized for the treatment of an underlying hematologic condition and had fusariosis develop in the context of immunosuppression caused by the underlying disease and its treatment. IF was defined as the isolation of Fusarium spp. from any sterilized biologic material, such as blood or skin biopsy, or from respiratory secretions in patients with typical clinical signs, including fever and metastatic skin lesions (1). A cutaneous portal of entry was defined when the clinical manifestations (and the diagnosis) of IF were preceded by the occurrence of localized skin lesions (such as cellulitis at sites of onychomycosis and intertrigo) with positive culture for Fusarium spp. The cases of IF were classified as proven or probable, according to the modified criteria of the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group Consensus Group (6). Superficial infections caused by Fusarium spp. in immunocompetent patients from the dermatology clinic were defined when Fusarium spp. was recovered from a skin lesion (usually onychomycosis and intertrigo). No changes in the population at risk, standards of collection and processing of biologic material, and diagnostic capabilities in the mycology laboratory occurred during the study period.
For the purpose of estimating changes in the incidence of IF, we split the study period into 2 periods: 2000–2005 (period 1) and 2006–2010 (period 2). We calculated the incidence of IF for the 2 periods using total admissions in the hematology unit as denominator and expressing the rates as number of cases per 1,000 admissions. The incidence of superficial infections caused by Fusarium spp. was expressed as number of positive cultures per 1,000 superficial cultures processed. Incidence densities between different periods were compared by the χ2 test using Epi Info software version 6.04d (Centers for Disease Control and Prevention, Atlanta, GA, USA). We considered p values <0.05 as statistically significant.
During 2000–2010, a total of 21 cases of IF were diagnosed in patients in the hematology unit at the hospital (Table). Acute myeloid leukemia (AML) was the most frequent underlying disease (42.9%); 12 patients (57.1%) were HCT recipients (8 allogeneic, 4 autologous). Neutropenia (81.0%), receipt of corticosteroids (76.2%), and graft-versus-host disease (6 of 8 allogeneic HCT recipients) were the most frequent predisposing factors. The IF diagnosis was confirmed by blood culture alone in 7 cases, blood culture plus culture and histopathology of biopsy of a metastatic skin lesion in 4, culture and histopathology of skin biopsy in 7, culture of synovial fluid in 2, and culture of sinus aspirate in 1. Among the 20 patients who received treatment, deoxycholate amphotericin B (10 patients) and voriconazole (7 patients) were the most frequent agents used as primary therapy. The overall 30-day and 60-day survival rates for the 21 patients were 33.3% and 28.6%, respectively.
A cutaneous portal of entry was identified in 14 (66.7%) of the 21 cases: onychomycosis with periungueal cellulitis in 6 cases, onychomycosis with interdigital intertrigo in 1 case, intertrigo with lymphangitis in 2 cases, intertrigo alone in 4 cases, and ulcer in 1 case (Figure 1, panels A–D). Some of the lesions of intertrigo evolved to necrosis and tissue loss (Figure 1, panel E). The median time from admission to recognition of a cutaneous portal of entry was 11 days (range −11 to 65).
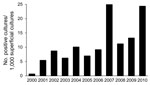
Figure 2 shows the incidence of IF during the full study period. The incidence was 0.86 cases per 1,000 admissions in period 1 and 10.23 cases per 1,000 admissions in period 2 (p<0.001). The incidence of IF with a cutaneous portal of entry was 0.43 per 1,000 admissions in period 1 and 6.99 per 1,000 admissions in period 2 (p<0.001).
Figure 3 shows the incidence of superficial infection caused by Fusarium spp. in patients from the dermatology outpatient clinic. The incidence (positive cultures per 1,000 superficial cultures) was 7.23 in period 1 and 16.26 in period 2 (p<0.001).
Isolates from hematologic and dermatologic patients showed similar species distribution, with a predominance of F. solani species complex (FSSC) species 2 (69% and 74%, respectively). By contrast, most environmental isolates from the initial investigation were identified as F. oxysporum species complex, with very few isolates of FSSC species 2, suggesting that most of the clinical cases of IF we identified would not be traceable to a specific environmental source in the hospital ward (7).
We observed an increase in the incidence of IF over time among patients in the hematology ward of our hospital; a cutaneous portal of entry was evident in most cases. We also observed an increase in the incidence of superficial infections caused by Fusarium spp. in outpatient nonhematologic patients at the same hospital during the same period.
The increase in the incidence of IF from period 1 (2000–2005) to period 2 (2006–2010) was >10-fold and showed a clear upward trend. Although IF is considered an emerging invasive fungal disease, affecting mostly patients with hematologic malignancies (3), its incidence is usually low. An epidemiologic study conducted in 18 hospitals in Italy reported 15 cases of IF among 11,802 patients with hematologic malignancies; patients with AML had the highest incidence (13 cases in 3,012 patients) (8). Another Italian study reported 3 cases among 1,249 allogeneic HCT recipients (9). In the United States, a large prospective study performed in 21 centers (1,194 allogeneic HCT recipients) reported only a few cases of infection caused by non–Aspergillus spp. molds, with a <0.3% 1-year cumulative incidence (10). By contrast, a prospective study conducted in 8 centers in Brazil during 2007–2009 reported 23 episodes of IF in 937 hematologic patients (2.4% overall incidence rate) (11); this study found a 1-year cumulative incidence of 5.2%, 3.8%, and 0.6% among allogeneic HCT recipients, AML patients, and autologous HCT recipients, respectively. No center effect was observed to account for this high incidence.
We also found a high incidence of a cutaneous portal of entry for IF, which is in sharp contrast with what had been previously reported. Similar to invasive aspergillosis, IF is thought to be acquired by inhalation of conidia from the air but occasionally has a cutaneous portal of entry (4). In a review of 259 published cases of IF, a cutaneous portal of entry was reported for only 11% of cases, and these were nearly all restricted to onychomycosis as the primary lesion (5). By contrast, 14 (66.7%) of the 21 IF cases in our study had a cutaneous portal of entry. Molecular typing of isolates recovered from sites of invasive disease (blood, synovial fluid) and from the lesions in the feet thought to be the portal of entry was performed for 4 cases and showed the same species for 3 (7).
Our results showed that interdigital intertrigo was as common as onychomycosis, occurring in 7 of the 14 cases with a cutaneous portal of entry. Onychomycosis is usually caused by Candida spp. and dermatophytes (12), but fusarial onychomycosis is a known clinical entity (13). Furthermore, recent studies have suggested that nondermatophyte fungi (including Fusarium spp.) are emerging as causes of onychomycosis (14–16). By contrast, interdigital intertrigo is rarely caused by Fusarium spp. (17).
The increased incidence of IF observed in 2007 at our hospital and the unique aspect of a cutaneous portal of entry in most cases raised the possibility that the patients could have acquired IF by contact with contaminated water in the hospital. Our hypothesis was that patients had been admitted with subtle skin breakdowns that became colonized by Fusarium spp. after contact with the hospital water, and local infection and dissemination subsequently developed. In support of this hypothesis were the findings of Anaissie et al., who reported that Fusarium spp. were recovered from 57% of water samples and 88% of water-related structures in a hospital in the United States; molecular studies of the isolates revealed a close relatedness between patient and environmental isolates (18). However, our environmental investigation showed that, although Fusarium spp. were present in the hospital water system, most isolates from patients belonged to the FSSC 2, whereas environmental isolates belonged to the F. oxysporum species complex (7). These results suggest that the infection did not have a nosocomial origin.
Concomitant to the increase in IF, we recorded an increase in the growth of Fusarium spp. from superficial infections in outpatients, from 7.23 positive cultures for period 1 to 16.26 positive cultures per 1,000 superficial cultures for period 2. Considering this apparent emergence of fusarial superficial infections in the community, the immunocompromised patients served as sentinels for the detection of this problem (19).
A limitation of our study is the denominator used to calculate the incidence. Because infection was acquired in the community, the appropriate denominator would be population based. However, because obtaining such a denominator would be difficult, we used a hospital-based denominator to approximate the incidence.
Our findings may have implications for future research, in particular, determining the environmental reservoirs of Fusarium spp. in the community that promoted the emergence of superficial fusariosis in immunocompetent patients. Fusarium spp. are widely found in the environment and are pathogens of various plants, including tomatoes, soybeans, and various grains (20). One possibility for an increase in Fusarium spp. in the environment is agricultural activities. For example, the Cerrado area is a large (≈2 million m2) territory that encompasses 10 states of Brazil. During the past 15–20 years, the area underwent a great deal of change in its composition, with a massive replacement of the native vegetation with monoculture, typically soybeans and pasture (21). A study evaluating the fungal diversity of the region found a great loss of fungal richness and diversity in the soybean plantation and pasture areas compared with native vegetation, with a concentration of ascomycetes (22). Other questions that require future research include assessment of the frequency and clinical significance of baseline skin colonization with Fusarium spp. in immunosuppressed patients and evaluation of preventive measures to reduce the incidence of this devastating disease.
In conclusion, we observed an increase in the incidence of IF in our hematology ward, with a cutaneous portal of entry, and of superficial fusariosis in immunocompetent outpatients. Future studies are needed to identify reservoirs of Fusarium spp. in the community, as well as preventive measures for patients at high risk for IF.
Dr Nucci is associate professor in the Department of Internal Medicine and head of the Mycology Laboratory of the University Hospital, Federal University of Rio de Janeiro, Brazil. His primary research interests are supportive care in hematologic patients, infections in hematologic patients and hematopoietic cell transplant recipients, and nosocomial fungal infections.
References
- Nucci M, Anaissie EJ, Queiroz-Telles F, Martins CA, Trabasso P, Solza C, Outcome predictors of 84 patients with hematologic malignancies and Fusarium infection. Cancer. 2003;98:315–9. DOIPubMedGoogle Scholar
- Nucci M, Marr KA, Queiroz-Telles F, Martins CA, Trabasso P, Costa S, Fusarium infection in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2004;38:1237–42 and. DOIPubMedGoogle Scholar
- Nucci M, Anaissie E. Emerging fungi. Infect Dis Clin North Am. 2006;20:563–79 and. DOIPubMedGoogle Scholar
- Girmenia C, Arcese W, Micozzi A, Martino P, Bianco P, Morace G. Onychomycosis as a possible origin of disseminated Fusarium solani infection in a patient with severe aplastic anemia. Clin Infect Dis. 1992;14:1167 and. DOIPubMedGoogle Scholar
- Nucci M, Anaissie E. Cutaneous infection by Fusarium species in healthy and immunocompromised hosts: implications for diagnosis and management. Clin Infect Dis. 2002;35:909–20 and. DOIPubMedGoogle Scholar
- De Pauw B, Walsh TJ, Donnelly JP, Stevens DA, Edwards JE, Calandra T, Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis. 2008;46:1813–21 and. DOIPubMedGoogle Scholar
- Scheel CM, Hurst SF, Barreiros G, Akiti T, Nucci M, Balajee SA. Molecular analyses of Fusarium isolates recovered from a cluster of invasive mold infections in a Brazilian hospital. BMC Infect Dis. 2013;13:49 and. DOIPubMedGoogle Scholar
- Pagano L, Caira M, Candoni A, Offidani M, Fianchi L, Martino B, The epidemiology of fungal infections in patients with hematologic malignancies: the SEIFEM-2004 study. Haematologica. 2006;91:1068–75 .PubMedGoogle Scholar
- Pagano L, Caira M, Nosari A, Van Lint MT, Candoni A, Offidani M, Fungal infections in recipients of hematopoietic stem cell transplants: results of the SEIFEM B-2004 study—Sorveglianza Epidemiologica Infezioni Fungine Nelle Emopatie Maligne. Clin Infect Dis. 2007;45:1161–70 and. DOIPubMedGoogle Scholar
- Kontoyiannis DP, Marr KA, Park BJ, Alexander BD, Anaissie EJ, Walsh TJ, Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001–2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis. 2010;50:1091–100 and. DOIPubMedGoogle Scholar
- Nucci M, Garnica M, Gloria AB, Lehugeur DS, Dias VC, Palma LC, Invasive fungal diseases in haematopoietic cell transplant recipients and in patients with acute myeloid leukaemia or myelodysplasia in Brazil. Clin Microbiol Infect. 2012. [Epub ahead of print].
- Thomas J, Jacobson GA, Narkowicz CK, Peterson GM, Burnet H, Sharpe C. Toenail onychomycosis: an important global disease burden. J Clin Pharm Ther. 2010;35:497–519 and. DOIPubMedGoogle Scholar
- Nucci M, Anaissie E. Fusarium infections in immunocompromised patients. Clin Microbiol Rev. 2007;20:695–704 and. DOIPubMedGoogle Scholar
- Gupta AK, Ryder JE, Baran R, Summerbell RC. Non-dermatophyte onychomycosis. Dermatol Clin. 2003;21:257–68 and. DOIPubMedGoogle Scholar
- Guilhermetti E, Takahachi G, Shinobu CS, Svidzinski TI. Fusarium spp. as agents of onychomycosis in immunocompetent hosts. Int J Dermatol. 2007;46:822–6 and. DOIPubMedGoogle Scholar
- Godoy-Martinez P, Nunes FG, Tomimori-Yamashita J, Urrutia M, Zaror L, Silva V, Onychomycosis in Sao Paulo, Brazil. Mycopathologia. 2009;168:111–6 and. DOIPubMedGoogle Scholar
- Calado NB, Sousa F Jr, Gomes NO, Cardoso FR, Zaror LC, Milan EP. Fusarium nail and skin infection: a report of eight cases from Natal, Brazil. Mycopathologia. 2006;161:27–31 and. DOIPubMedGoogle Scholar
- Anaissie EJ, Kuchar RT, Rex JH, Francesconi A, Kasai M, Muller FM, Fusariosis associated with pathogenic Fusarium species colonization of a hospital water system: a new paradigm for the epidemiology of opportunistic mold infections. Clin Infect Dis. 2001;33:1871–8 and. DOIPubMedGoogle Scholar
- Rubin RH. The compromised host as sentinel chicken. N Engl J Med. 1987;317:1151–3 and. DOIPubMedGoogle Scholar
- Osborne LE, Stein JM. Epidemiology of Fusarium head blight on small-grain cereals. Int J Food Microbiol. 2007;119:103–8 and. DOIPubMedGoogle Scholar
- Brannstrom C, Jepson W, Filippi AM, Redo D, Xu Z, Ganesh S. Land change in the Brazilian Savanna (Cerrado), 1986−2002: comparative analysis and implications for land-use policy. Land Use Policy. 2008;25:579–95. DOIGoogle Scholar
- de Castro AP, Quirino BF, Pappas G Jr, Kurokawa AS, Neto EL, Kruger RH. Diversity of soil fungal communities of Cerrado and its closely surrounding agriculture fields. Arch Microbiol. 2008;190:129–39 and. DOIPubMedGoogle Scholar
Figures
Table
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 70% passing score) and earn continuing medical education (CME) credit, please go to www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/category/2922.html. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title:
Increased Incidence of Invasive Fusariosis with Cutaneous Portal of Entry, Brazil
CME Questions
1. What was the most common underlying disease among patients with invasive fusariosis in the current study?
A. Multiple myeloma
B. Non-Hodgkin lymphoma
C. Acute myeloid leukemia
D. Chronic myeloid leukemia
2. Which of the following statements regarding the presentation of invasive fusariosis is most accurate?
A. Two-thirds of cases were notable for a cutaneous portal of entry of Fusarium spp.
B. All cases of cutaneous entry were associated with onychomycosis
C. Most cutaneous portals of entry were identified within 48 hours
D. Neutropenia was rare among cases of invasive fusariosis
3. Which of the following statements regarding the laboratory identification and management of invasive fusariosis in the current study is most accurate?
A. All patients had a positive blood culture
B. Fluconazole was the most common treatment used
C. The 30-day survival rate was 33.3%
D. Infecting Fusarium spp. were derived from the hospital’s water system
4. What were the trends in the rates of invasive and cutaneous fusariosis between 2000 and 2010?
A. Increased incidence of both invasive and cutaneous fusariosis
B. Increased incidence of invasive fusariosis; reduced incidence of cutaneous fusariosis
C. Reduced incidence of invasive fusariosis; increased incidence of cutaneous fusariosis
D. Reduced incidence of both invasive and cutaneous fusariosis
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
Related Links
Table of Contents – Volume 19, Number 10—October 2013
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Marcio Nucci, Hospital Universitário Clementino Fraga Filho, UFRJ Rua Prof Rodolpho Paulo Rocco 255–21941-913, Rio de Janeiro, Brazil
Top