Volume 19, Number 7—July 2013
Letter
Avian Influenza A(H7N9) Virus Infections, Shanghai, China
Cite This Article
Citation for Media
To the Editor: On March 31, 2013, the National Health and Family Planning Commission of China notified the World Health Organization of 3 cases of human infections with avian influenza A(H7N9) virus. These cases were caused by a novel virus that was identified by laboratory testing at the China Centers for Disease Control and Prevention (CDC) on March 29 (1).
As of April 19, 2013, a total of 91 laboratory-confirmed human cases (17 deaths) of infection with avian influenza A(H7N9) virus were reported in 4 provinces in China (2). We report clinical features of 2 infected adults who died, 2 critically ill infected adults who recovered, and 1 infected child who had a mild case during this outbreak in Shanghai, China.
A 3.5-year-old boy had fever (39.5°C) for 3 days and mild rhinorrhea starting on March 31. He was admitted to a district pediatric outpatient clinic on April 1. At admission, the child was given oseltamivir for 5 days, even though signs and symptoms had resolved. Nasopharyngeal swab samples were positive by real-time PCR for avian influenza A(H7N9) virus. All symptoms resolved uneventfully by April 3, and CDC was notified that avian influenza A(H7N9) virus was identified in his respiratory sample. The patient was discharged on day 11 after illness onset.
The 4 adult patients were given diagnoses of severe pneumonia with shortness of breath, dyspnea, and marked hypoxia (Table). Duration from disease onset to severe illness was 5–7 days. At admission, the 4 patients with severe cases had decreased peripheral blood leukocyte counts and increased levels of aspartate aminotransferase; 3 had increased levels of lactate dehydrogenase (Table).
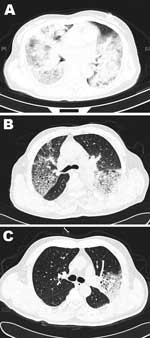
All 4 adult patients had radiologically confirmed pneumonia and bilateral patchy alveolar opacities or diffused lobar consolidation with or without pleural effusion (Figure, Appendix). Findings on chest radiographs for severe cases requiring mechanical ventilation were consistent with those for acute respiratory distress syndrome.
Among the 4 severe cases in adults, a 52-year-old woman (patient 1) and a 49-year-old man (patient 2) died from acute respiratory distress syndrome and multiple organ failure on days 14 and 10, respectively, after disease onset and 1–2 days after progression to respiratory failure. Two other patients showed improvement and were virus negative 6 and 4 days after antiviral treatment. After 23–24 days of treatment in an intensive care unit, the 2 patients with severe cases recovered and were discharged (Table).
The 2 patients who died were given methylprednisolone. Of the 2 patients who recovered, 1 was given a low dose of methylprednisolone for 1 week and the other was not given methylprednisolone. Although it is difficult to assess the role of glucocorticoids in treatment because of limited number of cases, caution is advised because of possible serious adverse events, including death, as reported for human infection with influenza A(H1N1) virus (4).
One of the adult patients reported exposure to poultry. The family of the child patient raised chickens and ducks, but these animals had no apparent disease, and cloacal swab specimens were negative for avian influenza A(H7N9) virus. One patient who died (patient 2) had frequent occupational exposure to poultry. Sixteen contacts of the child and 45 contacts of the 4 adult patients were monitored, and routine virologic sampling was performed. One contact (husband of patient 1) of a patient who died (Table) became febrile and was positive for avian influenza A(H7N9) virus on April 12 (day 24 after disease onset for patient 1); as of the date of this report, he was receiving treatment in an intensive care unit. However, it is difficult to tell if this is a case of human-to-human transmission or if both persons were exposed to infectious poultry. All remaining contacts had no symptoms and were negative for virus by PCR.
Several features of this avian influenza A(H7N9) outbreak are distinct from those of previous avian influenza outbreaks. Human infection with this virus showed a case-fatality rate of 18.7% (17/91), but this rate is not as high as that for avian influenza A(H5N1) virus (case-fatality rate 59%) (5).
Avian influenza A(H7N9) virus infection seems to cause more severe human illness than do other subgroups of H7 influenza A viruses (subtypes H7N2, H7N3, and H7N7), which are usually associated with poultry outbreaks but cause mild disease in humans. However, infection with avian influenza A(H7N7) virus resulted in the death of a veterinarian during an outbreak in the Netherlands (6). In the 5 patients reported here, avian influenza A(H7N9) virus caused fatal disease in 2 adult patients 52 and 49 years of age, who had other medical conditions. Older age has been reported to confer higher risk for developing more severe influenza-associated outcomes (7).
In conclusion, these cases indicated that avian influenza A(H7N9) virus might not be as virulent as avian influenza A(H5N1) virus in humans. Avian influenza A(H7N9) virus does not appear to cause obvious disease in poultry and causes mild disease in children. More severe disease in adults occurred among those had concurrent diseases or were immunodeficient.
References
- Gao R, Cao B, Hu Y, Feng Z, Wang D, Hu W, Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med. 2013 [Epub ahead of print].PubMedGoogle Scholar
- World Health Organization. Human infection with influenza A (H7N9) virus in China [cited 2013 Apr 22]. ww.who.int/csr/don/2013_04_19/en/index.html
- Yang F, Wang J, Jiang L, Jin JL, Shao LY, Zhang Y, A fatal case caused by novel H7N9 avian influenza A virus in China. Emerging Microbes and Infections. 2013;2:e19 .DOIGoogle Scholar
- Brun-Buisson C, Richard JC, Mercat A, Thiébaut AC, Brochard L. REVA-SRLF A/H1N1v 2009 Registry Group. Early corticosteroids in severe influenza A/H1N1 pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med. 2011;183:1200–6. DOIPubMedGoogle Scholar
- Update on human cases of influenza at the human–animal interface. Wkly Epidemiol Rec. 2013;88:137–44.PubMedGoogle Scholar
- World Health Organization. Avian influenza: assessing the pandemic threat, 2005, Table 3, Documented human infections with avian influenza viruses [cited 2013 Apr 22]. www.who.int/influenza/resources/documents/h5n1_assessing_pandemic_threat/en
- Van Kerkhove MD, Vandemaele KA, Shinde V, Jaramillo-Gutierrez G, Koukounari A, Donnelly CA, Risk factors for severe outcomes following 2009 influenza A (H1N1) infection: a global pooled analysis. PLoS Med. 2011;8:e1001053. DOIPubMedGoogle Scholar
Figure
Table
Cite This Article1These authors contributed equally to this article.
Related Links
Table of Contents – Volume 19, Number 7—July 2013
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Wenhong Zhang, Department of Infectious Diseases, Huashan Hospital and Key Laboratory of Medical Molecular Virology, Ministry of Education and Health, Fudan University, Shanghai 200040, China
Top