Volume 20, Number 9—September 2014
CME ACTIVITY - Synopsis
Confirmed Bacillus anthracis Infection among Persons Who Inject Drugs, Scotland, 2009–2010
Cite This Article
Citation for Media
Introduction

Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint providership of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at http://www.medscape.org/journal/eid; (4) view/print certificate.
Release date: August 15, 2014; Expiration date: August 15, 2015
Learning Objectives
Upon completion of this activity, participants will be able to:
• Distinguish patient factors associated with a higher risk for mortality from injection anthrax
• Evaluate variables at patient presentation associated with a higher risk for death from injection anthrax
• Assess medical treatment variables associated with a higher risk for death from injection anthrax
• Analyze hospital events associated with a higher risk for mortality from injection anthrax.
CME Editor
Karen L. Foster, Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: Karen L. Foster has disclosed no relevant financial relationships.
CME Author
Charles P. Vega, MD, Clinical Professor of Family Medicine, University of California, Irvine. Disclosure: Charles P. Vega, MD, has disclosed the following financial relationships: served as an advisor or consultant for McNeil Pharmaceuticals.
Authors
Disclosures: Malcolm Booth, MBChB, FRCA, MPhil, FFICM; Lindsay Donaldson, MB ChB, FRCA, FFICM; Xizhong Cui, MD, PhD; Junfeng Sun, PhD; Stephen Cole, BSc(Hons), MBChB, FRCA, FFICM, FRCP; Susan Dailsey, MBChB; Andrew Hart, BSc, MBChB, MD, PhD, MRCS, AFRCS, FRCS(plast); Neil Johns, MBBS, BSc; Paul McConnell, MBChB(Hons), FRCA, EDIC, FFICM; Tina McLennan, MBChB, FRCA; Benjamin Parcell, MBChB, FRCPath; Henry Robb, MBChB, FRCA, MS, FFICM; Charles Wallis, MBChB, FRCA, FFICM; and Peter Q. Eichacker, MD, have disclosed no relevant financial relationships. Benjamin Shippey, FRCPE, FFICM, FRCA, BM, BS, BMedSci, has disclosed the following relevant financial relationships: served as an advisor or consultant for and received grants for clinical research from Aircraft Medical. Malcolm Sim, BSc(Hons), MBChB, FRCA, FRCP, FFICM, EDIC, has disclosed the following relevant financial relationships: received grants for clinical research from Roche/Wyeth Pharmaceuticals Inc.
Abstract
In Scotland, the 2009 outbreak of Bacillus anthracis infection among persons who inject drugs resulted in a 28% death rate. To compare nonsurvivors and survivors, we obtained data on 11 nonsurvivors and 16 survivors. Time from B. anthracis exposure to symptoms or hospitalization and skin and limb findings at presentation did not differ between nonsurvivors and survivors. Proportionately more nonsurvivors had histories of excessive alcohol use (p = 0.05) and required vasopressors and/or mechanical ventilation (p<0.01 for each individually). Nonsurvivors also had higher sequential organ failure assessment scores (mean + SEM) (7.3 + 0.9 vs. 1.2 + 0.4, p<0.0001). Antibacterial drug administration, surgery, and anthrax polyclonal immune globulin treatments did not differ between nonsurvivors and survivors. Of the 14 patients who required vasopressors during hospitalization, 11 died. Sequential organ failure assessment score or vasopressor requirement during hospitalization might identify patients with injectional anthrax for whom limited adjunctive therapies might be beneficial.
Bacillus anthracis infection in humans has typically been classified as cutaneous, gastrointestinal, or inhalational on the basis of the bacterium’s route of entry (1). However, in Scotland, United Kingdom, during 2009–2010, a total of 47 patients had confirmed B. anthracis soft tissue infection related to injection of contaminated heroin (2,3). This form of B. anthracis infection appears to be distinct from cutaneous disease and has been termed “injectional” anthrax (2–5). In addition to confirmed cases, 35 probable and 37 possible cases in Scotland, 5 confirmed cases in England, and 2 confirmed cases in Germany also were identified. This initial outbreak ended in late 2010, but since the summer of 2012, new cases have been reported in the United Kingdom and Europe (3,4). Although 1 case of injectional anthrax was recognized in Norway in 2001, the patients in 2009–2010 constitute the first large outbreak of this newly recognized and poorly characterized form of anthrax (5).
Health Protection Scotland (HPS) has published epidemiologic analyses of the 2009–2010 outbreak (3,6,7). Among other findings, analysis suggested associations between longer injecting histories, opioid substitution therapy, and alcohol use and risk for B. anthracis infection in persons who inject drugs (PWID) (6). Several case reports from the outbreak also have been published (8–12), but they did not include systematic examinations of the physical, laboratory, and surgical findings or of therapies administered. Notably, although 13 of the 47 persons from Scotland who had confirmed cases died, no published report has compared findings in survivors and nonsurvivors. Such a comparison is needed for the prognosis and management of future cases. We therefore sent a questionnaire regarding these issues to clinicians who had treated PWIDs in whom B. anthracis infection was confirmed in Scotland during the outbreak.
Approval
We used data collected during routine hospital care of patients. Patient identifiers were removed from data before analysis. Because of the retrospective nature of the study and the anonymity of data, the West of Scotland Research Ethics Service (Glasgow, Scotland, UK) and the Office of Human Subjects Research from the Clinical Center at the National Institutes of Health (Bethesda, MD, USA) exempted the study from formal review.
Data Collection
We developed an electronic questionnaire that requested information in several areas. These were: general information (i.e., age, sex, and medical and drug histories); current illness; data at the time the patient sought care, including skin and limb findings, vital signs, laboratory findings, and diagnosis (i.e., was anthrax infection or sepsis initially suspected?); medical and surgical treatments at the time the patient sought care or later (including antibacterial drugs; need for hemodynamic, respiratory, renal replacement, or blood product support; and use of anthrax immune globulin, a polyclonal antibody produced by Cangene [Winnipeg, MB, Canada] and made available by the Centers for Diseases Control and Prevention [Atlanta, GA, USA]; microbiological data supporting the diagnosis of anthrax infection and the time at which the diagnosis was confirmed; surgical findings; other procedures performed during hospitalization; levels of organ injury based on the sequential organ failure assessment (SOFA) score; and outcomes, including survival, time in the intensive care unit (ICU), and total time in hospital.
During this anthrax outbreak, HPS identified 13 hospitals to which the 47 patients with confirmed anthrax were admitted (3). In March 2012, two of the authors (L.D. and M.G.B.), who were members of the HPS anthrax outbreak control team, asked physicians known to have treated persons with confirmed anthrax to complete the questionnaire. The questionnaire was sent to these physicians in early April 2012. In June 2012, physicians who had not yet returned it were asked to do so. Data from all questionnaires completed by the end of August 2012 were analyzed. We received no additional questionnaires after August 2012. Contributors were subsequently contacted to clarify missing or unclear responses.
Data Analysis
A variable was reported on only if >50% (i.e., >14) of questionnaires provided definitive data. SOFA score was calculated as previously described for each patient for whom data were obtained within the initial 24 hours after they sought care (13). Measures of time were expressed as medians (interquartile range [IQR]). We used Wilcoxon rank sum test to compare these times between survivors and nonsurvivors. Categorical data (i.e., chief complaints, types of initial surgery and gross tissue findings, skin and limb findings, and treatments) were analyzed with Fisher exact or χ2 test, where applicable, and continuous data (i.e., laboratory data, age, vital signs, and SOFA score) were examined with 1-way ANOVA (analysis of variance).
Initial Findings
We received data on 27 confirmed cases from the outbreak: 16 of the 33 survivors and 11 of the 14 nonsurvivors from 10 of 13 hospitals that admitted PWID with outbreak-associated anthrax. The median (IQR) times (days) from exposure to onset of symptoms and from onset of symptoms to hospital admission were 1 (0–4) and 2 (2–4), respectively, and these did not differ significantly between survivors (1 [0–4.5] and 3 [2–5] days) and nonsurvivors (1 (0–2] and 2 [2– 2] days) (p = 0.90 and p = 0.19, respectively) (Figure 1).
Mean (+ SEM) age of patients was 34.5 + 1.7 years. Nonsurvivors tended to be older than survivors (38.2 + 2.8 vs. 31.9 + 1.9, p = 0.07). The proportion who were male did not differ significantly (9 [56%] survivors vs. 9 [82%] nonsurvivors, p = 0.23). Although history of excessive alcohol use was higher in nonsurvivors than in survivors (4 [80%] of 5 vs. 3 [23%] of 13, p = 0.05), tobacco use and suspected injection sites (arm, groin, buttock, or leg) and routes (intravenous or intramuscular) of contaminated drug injection did not differ significantly (p>0.19 for all, data not shown). HIV infection status was available for only 10 patients (4 nonsurvivors), and hepatitis C infection status was available for only 12 patients (5 nonsurvivors). One survivor was HIV positive, and 4 survivors and 5 nonsurvivors had histories of hepatitis C virus infection. Although the questionnaire requested other medical history (e.g., hepatitis B virus infection status), these data were not provided in sufficient numbers for analysis.
Eleven (69%) survivors and 2 (18%) nonsurvivors had only localized skin or limb symptoms (i.e., pain, swelling, erythema, exudate); 5 (31%) survivors and 4 (36%) nonsurvivors had both localized and generalized symptoms (i.e., fever, confusion, seizures, abdominal pain, fatigue, malaise, sweating, headache), and 0 survivors and 5 (45%) nonsurvivors had only generalized symptom (Table 1). The proportion of nonsurvivors who had only a localized or only a generalized complaint was less (p = 0.02) and greater (p = 0.006) than the proportion of survivors.
At the time they sought care, nonsurvivors had lower temperatures and systolic blood pressures and higher respiratory rates than survivors (p<0.01) (Figure 2), but other vital signs (mean + SEM), including diastolic blood pressure (68 + 4 vs. 61 + 5 mm Hg), heart rate (111 + 5 vs. 118 + 8 beats per minute), and capillary refill (2.6 + 0.4 vs. 3.6 + 0.5 seconds) did not differ significantly. Four nonsurvivors, but only 1 survivor, had a temperature <36°C when they sought care, whereas 1 nonsurvivor and 7 survivors had temperatures >38°C. Also, whereas 4 nonsurvivors had systolic blood pressures <90 mm Hg, no survivor did. The Glasgow coma score recorded during the initial 24 hours was lower for nonsurvivors than for survivors (p = 0.008) (Figure 2). The proportion of survivors and nonsurvivors for whom specific skin and limb findings were available did not differ significantly (p>0.19 for all) (Table 2).
Nonsurvivors had lower serum sodium, corrected calcium, albumin, and platelet levels and higher bilirubin, percentage circulating neutrophils, hemoglobin concentration, international normalized ratio, prothrombin time, partial thromboplastin time, and C-reactive protein levels (p<0.05) (all shown in Figure 3 except prothrombin time, which was median [IQR] 12 [11–13] vs.15 [13.5–16.1]). For a greater proportion of nonsurvivors than survivors, arterial blood gases were measured when they sought care (10 [91%] vs. 6 [38%], p = 0.008). In patients with for whom the following values were measured, nonsurvivors had lower bicarbonates and greater base deficits (p = 0.02). Other laboratory data did not differ significantly between survivors and nonsurvivors (Table 2).
Clinical Impression, Treatment, and SOFA Score at Presentation
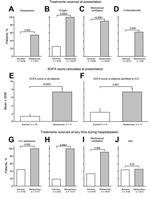
Sepsis was noted in a greater proportion of nonsurvivors than survivors at presentation (7 [70%] vs. 3 [19%], p = 0.02), but B. anthracis infection was not (5 [46%] vs. 12 [75%], p = 0.22). All patients were initially treated with at least 1 antibacterial drug (Table 3). Neither type nor number (3.8 + 0.3 vs. 3.6 + 0.7, respectively) of antibacterial drugs received differed significantly between survivors and nonsurvivors (p>0.06). Among survivors, 75% received ciprofloxacin and 88% received clindamycin; among nonsurvivors, 55% and 64% received these drugs, respectively. More nonsurvivors than survivors received vasopressors, oxygen support, mechanical ventilation, and corticosteroids (all p<0.002) (Figure 4) but not undergo surgery (5 [46%] nonsurvivors vs. 11 [69%] survivors, p = 0.26) (Table 4). SOFA score calculated within the first 24 hours after patients sought care was higher for nonsurvivors than for survivors and for only those requiring ICU admission (p<0.003) (Figure 4).
Confirmation of B. anthracis Infection
The basis for the microbiological diagnosis of B. anthracis for the 47 confirmed anthrax cases has been published (3). For the 27 cases reported here, the median (IQR) time (days) to confirmation was 3 (1.5–9.0) and did not differ between nonsurvivors and survivors (3.0 [2.0–7.0] vs. 3.0 [1.0–9.0], respectively, p = 0.83) (Figure 1). Results of tests confirming B. anthracis infection did not differ significantly between survivors and nonsurvivors (p>0.05 for all) (Table 2). We had insufficient data to determine how often bacteria other than B. anthracis were found in blood or tissue samples.
Hospital Course
Proportionately more nonsurvivors than survivors received ICU care (11 [100%] vs. 7 [44%], p = 0.003) (Figure 4). The median (IQR) time (days) survivors remained in the ICU and hospital were 2.0 (1.0–13.3) and 21.5 (4.5–35.5), respectively. For nonsurvivors, median time from hospital admission to death was 2.1 (0.63–4) days. During hospitalization, proportionately more nonsurvivors received vasopressors (p = 0.0001) (Figure 4). More nonsurvivors also required mechanical ventilation (p = 0.005). Among all patients, 11 (79%) of the 14 who received vasopressors at any time died. Twelve (44%) of the 27 patients received anthrax immune globulin. We found no significant difference in the proportions of survivors and nonsurvivors who received anthrax immune globulin (p = 0.93, Figure 4) or in the median (IQR) time (days) to treatment (3 [1–3] vs. 1 [1–1]), respectively; p = 0.13).
We found no significant difference in the proportions of survivors and nonsurvivors who required surgery during hospitalization (11 [69%] vs. 5 [46%]; p = 0.26) or who required >1 surgery (7 [64%] vs. 4 (80%); p = 0.52) or in the median (IQR) time (days) to initial surgery (0 [0–1] vs. 0 [0–0.125]; p = 0.53) (Figure 1). Even when we examined data from the first week of hospitalization only, survivors and nonsurvivors did not differ significantly in the proportion having surgery (7 [44%] survivors vs. 4 [36%] nonsurvivors; p = 0.56). However, nonsurvivors more often bled excessively during surgery (4 [80%] nonsurvivors vs. 1 [8%] survivor; p = 0.01) (Table 4). One survivor had an arm amputated above the elbow, and 4 received skin grafts.
Receipt of packed erythrocytes, fresh frozen plasma, cryoglobulin, and platelets; renal replacement therapy; and pleural or peritoneal drainage did not differ significantly between survivors and nonsurvivors (Table 2). For 9 (36%) of 25 patients for whom information was available, cardiac function was assessed: echocardiography for 4 patients, troponin measures for 3, and lithium dilution cardiac output and pulse contour cardiac output for 1 each. Of these, 3 nonsurvivors were noted to have evidence of myocardial dysfunction on the basis of echocardiography, lithium dilution cardiac output, or pulse contour cardiac output; 1 survivor had an elevated troponin level. Causes of death for the 7 patients for whom autopsies were reported were as follows; multiple system organ failure caused by B. anthracis sepsis, 2 patients; necrotizing fasciitis related to B. anthracis, 1; sepsis and hemorrhagic meningitis with B. anthracis infection, 2; subarachnoid hemorrhage, 1; and myocardial infarction, 1.
Our review of 27 confirmed cases of B. anthracis infection in PWID compares clinical findings in survivors and nonsurvivors of this newly described form of infection. Although duration of symptoms and time to seeking hospital care did not differ between survivors and nonsurvivors, the severity of illness did. Most survivors reported localized symptoms related to the injection site, and none required vasopressor therapy or mechanical ventilation. In contrast, most nonsurvivors had generalized symptoms and evidence of sepsis, which required both vasopressor support and mechanical ventilation. Nonsurvivors also had lower systolic blood pressures and Glasgow coma scores; higher respiratory rates; worsened base deficits; higher levels of hemoglobin (consistent with hemoconcentration) and C-reactive protein; higher international normalized ratio; and lower sodium and albumin levels and platelet counts. During hospitalization, all nonsurvivors required vasopressor and ICU support, whereas only 3 and 7 survivors, respectively, required these. SOFA scores were substantially higher in nonsurvivors than survivors. Thus, assessing the need for aggressive cardiopulmonary support or determining a score like SOFA for patients with injectional anthrax can help identify those for whom prognosis is particularly poor and more aggressive therapy is needed.
Possibly consistent with prior analysis showing an association between excessive alcohol use and risk for B. anthracis infection in PWID, we found a higher incidence of excessive alcohol use among nonsurvivors than survivors (6). Increased bilirubin and decreased albumin levels in nonsurvivors might in part have reflected preexisting alcoholic liver disease. Although age did not differ significantly between survivors and nonsurvivors, the latter tended to be older, a finding consistent with analysis of inhalational B. anthracis infection (14).
Differences in outcome between survivors and nonsurvivors did not appear related to variation in treatment. All patients received antibacterial drugs from the time they sought care, and the types and numbers of antibacterial drugs administered did not differ. Also, the proportion of patients who had >1 surgeries and the time from admission to initial surgery did not differ. Finally, similar proportions of survivors and nonsurvivors received anthrax immune globulin, and the median time to treatment for these groups did not differ.
The most common skin and limb findings were localized edema, pain, and erythema. Although these findings are consistent with soft tissue infection, their presence did not differ between survivors and nonsurvivors (15). Thus, skin and limb findings and even the need for surgery did not appear to predict worsened prognoses. Notably absent in most patients was the eschar formation classically associated with cutaneous B. anthracis infection (16,17). This absence combined, with the frequent need for surgery and the overall high death rate despite receipt of antibacterial drugs, supports the observation that the pathogeneses of injectional and cutaneous anthrax differ (3,4–6).
In general, the severity of soft tissue infection and its requirement for surgery varies from mild to severe on the basis of the depth of tissue involvement and tissue necrosis (15). The B. anthracis soft tissue infections reviewed here reflect this range of disease. Two survivors had symptoms consistent with cellulitis, were treated only with antibacterial drugs, and were discharged within 1–2 days. However, 10 survivors and 5 nonsurvivors required debridement, fasciotomy, or laparotomy on >1 occasion; 10 had evidence of tissue necrosis. Even among the 5 survivors who did not have surgery, 3 required hospitalization for >7 days, which suggests severe infection. Without additional data about co-existing conditions, we cannot determine whether outcomes from soft tissue infection in PWID differ between B. anthracis and other bacteria.
B. anthracis has a cell wall that elicits a robust host inflammatory response and the endothelial dysfunction, shock, and organ injury with which this response is associated (1,18–21). However, B. anthracis also releases lethal and edema toxins that can produce this same dysfunction but through very different mechanisms than the cell wall (22,23). Whether, as a result of these diverse nontoxin and toxin components, the manifestations of soft tissue infection with B. anthracis differ from those of other bacteria is unclear. Several differences between nonsurvivors and survivors, such as reduced systolic blood pressure and sodium and worsened acidosis, are associated with worsened outcome with other types of soft tissue infection (24–26). However, of patients who required vasopressor treatment, almost 80% died. This death rate is high, even for patients identified with septic shock on the basis of need for vasopressors. However, this finding is consistent with the 2001 US outbreak of inhalational B. anthracis in which all patients in whom shock developed died (27). Also in the current review, nonsurvivors bled more during surgery, possibly because of an increase in international normalized ratio and a decrease in platelets. Although excessive bleeding is not typically associated with soft tissue infection, it is associated with inhalational and gastrointestinal B. anthracis infection (28,29).
B. anthracis lethal and edema toxins inhibit components in the innate and adaptive immune responses (22,23). This inhibition might contribute to infection, as well as suppress signs typically associated with an activated host inflammatory response (3). However, although temperature was lower in nonsurvivors than in survivors, circulating leukocyte counts, percentage of neutrophils, and C-reactive protein were higher and in ranges approaching or consistent with invasive soft tissue infection caused by other bacteria (30,31). Whether toxin production interferes with host defense and influences the features and course of injectional B. anthracis infection requires further study.
Several lines of evidence suggest that B. anthracis lethal toxin produces direct cardiac dysfunction (22). Whether such dysfunction contributes to clinical B. anthracis infection is unclear because there are few measures of cardiac function in patients. Although 4 of the 9 patients in the current review were described as having evidence of cardiac dysfunction, these data were limited. More comprehensive investigation of cardiac function is necessary during future outbreaks of B. anthracis.
This study has limitations. First, data were collected >2 years after patients sought care, were not obtainable for some questions on patients we included in the analysis, and were unavailable for 20 of the 47 confirmed cases. However, these 27 patients included 11 of the 14 nonsurvivors from the outbreak and probably were fairly representative of nonsurvivors. Second, data were limited regarding co-morbidities, particularly HIV infection and viral hepatitis status, which might have influenced outcome. Information about other co-morbidities (e.g., diabetes, heart disease, and chronic lung disease) might have been informative as well. Third, although 1 survivor and 1 nonsurvivor described headache, using the Glasgow coma scale to assess neurologic status might not have captured other patients with this symptom, a possible manifestation of underlying meningeal infection. Fourth, comparisons of therapies administered to survivors and nonsurvivors later than when they initially sought care might have been confounded by patients’ length of hospitalization; however, data were insufficient to analyze the influence of this variable. Finally, autopsy findings were available only for 7 patients.
The 2009–2010 outbreak of B. anthracis infection among PWID in Scotland was considered over at the end of 2010 (3). However, during June 2012–December 2013, thirteen new cases were reported in the United Kingdom and Europe (4,32). The death rate for these patients has been close to 50%. Findings from the patients in the current review, combined with findings from newer cases, emphasize the need to better understand the pathogenesis and management of this recently identified form of B. anthracis infection.
Drs Booth and Donaldson are critical care physicians at Glasgow Royal Infirmary, Glasgow, Scotland. Their research interests include improving the treatment of invasive infection in critically ill patients.
Acknowledgments
We appreciate the editorial assistance of Kelly Byrne during the preparation of this manuscript.
This work was supported by the Intramural Research Program of the National Institutes of Health and the National Institutes of Health Clinical Center.
None of the authors have commercial or other associations that might pose a conflict of interest.
References
- Sweeney DA, Hicks CW, Cui X, Li Y, Eichacker PQ. Anthrax infection. Am J Respir Crit Care Med. 2011;184:1333–41 and. DOIPubMedGoogle Scholar
- Booth MG, Hood J, Brooks TJ, Hart A. Anthrax infection in drug users. Lancet. 2010;375:1345–6 and. DOIPubMedGoogle Scholar
- Health Protection Scotland. An outbreak of anthrax among drug users in Scotland, December 2009 to December 2010. A report on behalf of the National Anthrax Outbreak Control Team [cited 2013 May 30]. http://www.documents.hps.scot.nhs.uk/giz/anthrax-outbreak/anthrax-outbreak-report-2011-12.pdf
- Grunow R, Klee SR, Beyer W, George M, Grunow D, Barduhn A, Anthrax among heroin users in Europe possibly caused by same Bacillus anthracis strain since 2000. Euro Surveill. 2013;18:20437.
- Ringertz SH, Hoiby EA, Jensenius M, Maehlen J, Caugant DA, Myklebust A, Injectional anthrax in a heroin skin-popper. Lancet. 2000;356:1574–5 . DOIPubMedGoogle Scholar
- Palmateer NE, Ramsay CN, Browning L, Goldberg DJ, Hutchinson SJ. Anthrax infection among heroin users in Scotland during 2009–2010: a case–control study by linkage to a national drug treatment database. Clin Infect Dis. 2012;55:706–10 . DOIPubMedGoogle Scholar
- Price EP, Seymour ML, Sarovich DS, Latham J, Wolken SR, Mason J, Molecular epidemiologic investigation of an anthrax outbreak among heroin users, Europe. Emerg Infect Dis. 2012;18:1307–13. DOIPubMedGoogle Scholar
- Beaumont G. Anthrax in a Scottish intravenous drug user. J Forensic Leg Med. 2010;17:443–5 and. DOIPubMedGoogle Scholar
- Jallali N, Hettiaratchy S, Gordon AC, Jain A. The surgical management of injectional anthrax. J Plast Reconstr Aesthet Surg. 2011;64:276–7. DOIPubMedGoogle Scholar
- Johns N, Cooper D, Terrace J. An unusual case of peritonitis in an intravenous drug user. Gastroenterology. 2011;141:435–6, 780–1. DOIPubMedGoogle Scholar
- Knox D, Murray G, Millar M, Hamilton D, Connor M, Ferdinand RD, Subcutaneous anthrax in three intravenous drug users: a new clinical diagnosis. J Bone Joint Surg Br. 2011;93:414–7. DOIPubMedGoogle Scholar
- Parcell BJ, Wilmshurst AD, France AJ, Motta L, Brooks T, Olver WJ. Injection anthrax causing compartment syndrome and necrotising fasciitis. J Clin Pathol. 2011;64:95–6. DOIPubMedGoogle Scholar
- Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, The SOFA (sepsis-related organ failure assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996;22:707–10. DOIPubMedGoogle Scholar
- Holty JE, Bravata DM, Liu H, Olshen RA, McDonald KM, Owens DK. Systematic review: a century of inhalational anthrax cases from 1900 to 2005. Ann Intern Med. 2006;144:270–80. DOIPubMedGoogle Scholar
- Ebright JR, Pieper B. Skin and soft tissue infections in injection drug users. Infect Dis Clin North Am. 2002;16:697–712. DOIPubMedGoogle Scholar
- Doganay L, Welsby PD. Anthrax: a disease in waiting? Postgrad Med J. 2006;82:754–6 . DOIPubMedGoogle Scholar
- Doganay M, Metan G, Alp E. A review of cutaneous anthrax and its outcome. J Infect Public Health. 2010;3:98–105. DOIPubMedGoogle Scholar
- Cui X, Su J, Li Y, Shiloach J, Solomon S, Kaufman JB, Bacillus anthracis cell wall produces injurious inflammation but paradoxically decreases the lethality of anthrax lethal toxin in a rat model. Intensive Care Med. 2010;36:148–56. DOIPubMedGoogle Scholar
- Langer M, Malykhin A, Maeda K, Chakrabarty K, Williamson KS, Feasley CL, Bacillus anthracis peptidoglycan stimulates an inflammatory response in monocytes through the p38 mitogen-activated protein kinase pathway. PLoS ONE. 2008;3:e3706. DOIPubMedGoogle Scholar
- Popov SG, Villasmil R, Bernardi J, Grene E, Cardwell J, Popova T, Effect of Bacillus anthracis lethal toxin on human peripheral blood mononuclear cells. FEBS Lett. 2002;527:211–5. DOIPubMedGoogle Scholar
- Qiu P, Li Y, Shiloach J, Cui X, Sun J, Trinh L, Bacillus anthracis cell wall peptidoglycan but not lethal or edema toxins produces changes consistent with disseminated intravascular coagulation in a rat model. J Infect Dis. 2013;208:978–89. DOIPubMedGoogle Scholar
- Hicks CW, Cui X, Sweeney DA, Li Y, Barochia A, Eichacker PQ. The potential contributions of lethal and edema toxins to the pathogenesis of anthrax associated shock. Toxins. 2011;3:1185–202.
- Moayeri M, Leppla SH. Cellular and systemic effects of anthrax lethal toxin and edema toxin. Mol Aspects Med. 2009;30:439–55. DOIPubMedGoogle Scholar
- Dworkin MS, Westercamp MD, Park L, McIntyre A. The epidemiology of necrotizing fasciitis including factors associated with death and amputation. Epidemiol Infect. 2009;137:1609–14. DOIPubMedGoogle Scholar
- Frazee BW, Fee C, Lynn J, Wang R, Bostrom A, Hargis C, Community-acquired necrotizing soft tissue infections: a review of 122 cases presenting to a single emergency department over 12 years. J Emerg Med. 2008;34:139–46. DOIPubMedGoogle Scholar
- Yaghoubian A, de Virgilio C, Dauphine C, Lewis RJ, Lin M. Use of admission serum lactate and sodium levels to predict mortality in necrotizing soft-tissue infections. Arch Surg. 2007;142:840–6, discussion 4–6. DOIPubMedGoogle Scholar
- Sherer K, Li Y, Cui X, Eichacker PQ. Lethal and edema toxins in the pathogenesis of Bacillus anthracis septic shock: implications for therapy. Am J Respir Crit Care Med. 2007;175:211–21 . DOIPubMedGoogle Scholar
- Abramova FA, Grinberg LM, Yampolskaya OV, Walker DH. Pathology of inhalational anthrax in 42 cases from the Sverdlovsk outbreak of 1979. Proc Natl Acad Sci U S A. 1993;90:2291–4. DOIPubMedGoogle Scholar
- Grinberg LM, Abramova FA, Yampolskaya OV, Walker DH, Smith JH. Quantitative pathology of inhalational anthrax I: quantitative microscopic findings. Mod Pathol. 2001;14:482–95. DOIPubMedGoogle Scholar
- Chan T, Yaghoubian A, Rosing D, Kaji A, de Virgilio C. Low sensitivity of physical examination findings in necrotizing soft tissue infection is improved with laboratory values: a prospective study. Am J Surg. 2008;196:926–30, discussion 30. DOIPubMedGoogle Scholar
- Su YC, Chen HW, Hong YC, Chen CT, Hsiao CT, Chen IC. Laboratory risk indicator for necrotizing fasciitis score and the outcomes. ANZ J Surg. 2008;78:968–72. DOIPubMedGoogle Scholar
- Holzmann T, Frangoulidis D, Simon M, Noll P, Schmoldt S, Hanczaruk M, Fatal anthrax infection in a heroin user from southern Germany, June 2012. Euro Surveill. 2012;17:20204 .PubMedGoogle Scholar
Figures
Tables
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the “Register” link on the right hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/about-ama/awards/ama-physicians-recognition-award.page. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title:
Confirmed Bacillus anthracis Infection among Persons Who Inject Drugs, Scotland, 2009–2010
CME Questions
1. You are seeing a 33-year-old man in the emergency department. He complains of severe pain and redness on his left upper extremity after injecting heroin into an antecubital vein yesterday. A brief examination of the area reveals 8 cm of erythema, induration, and calor, with a central necrotic area. You consider whether this patient has injection anthrax; you also consider his prognosis. Which one of the following patient factors was most associated with a higher risk for mortality related to injection anthrax in the current study?
A. Younger age
B. Male sex
C. Excessive alcohol use
D. Cigarette smoking
2. Which one of the following variables at patient presentation was most associated with a higher risk for mortality in the current study of patients with injection anthrax?
A. Temperature less than 36˚C
B. Heart rate greater than 115 beats per minute
C. Respiratory rate less than 12 breaths per minute
D. Skin lesions on the upper extremity vs lower extremity
3. Which one of the following statements regarding treatment and mortality outcomes related to injection anthrax in the current study is most accurate?
A. Rates of initial diagnosis with Bacillus anthracis infection were similar among nonsurvivors and survivors
B. Compared with survivors, nonsurvivors were less likely to receive antibiotics at hospital admission
C. Compared with survivors, nonsurvivors were less likely to receive ciprofloxacin
D. Nonsurvivors and survivors were equally likely to require surgery
4. Which one of the following hospital events was more common among nonsurvivors vs survivors in the current study?
A. Application of anthrax immune globulin
B. Excessive bleeding during surgery
C. Longer duration until initial surgery
D. Initiation of renal replacement therapy
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
1These authors contributed equally to this article.
Related Links
Table of Contents – Volume 20, Number 9—September 2014
| EID Search Options |
|---|
|
|
|
|
|
|



Please use the form below to submit correspondence to the authors or contact them at the following address:
Peter Q. Eichacker, Critical Care Medicine Department, National Institutes of Health, Bldg 10, Rm 2C145, Bethesda, MD 20892, USA
Top