Volume 22, Number 2—February 2016
Research
Association between Landscape Factors and Spatial Patterns of Plasmodium knowlesi Infections in Sabah, Malaysia
Cite This Article
Citation for Media
Abstract
The zoonotic malaria species Plasmodium knowlesi has become the main cause of human malaria in Malaysian Borneo. Deforestation and associated environmental and population changes have been hypothesized as main drivers of this apparent emergence. We gathered village-level data for P. knowlesi incidence for the districts of Kudat and Kota Marudu in Sabah state, Malaysia, for 2008–2012. We adjusted malaria records from routine reporting systems to reflect the diagnostic uncertainty of microscopy for P. knowlesi. We also developed negative binomial spatial autoregressive models to assess potential associations between P. knowlesi incidence and environmental variables derived from satellite-based remote-sensing data. Marked spatial heterogeneity in P. knowlesi incidence was observed, and village-level numbers of P. knowlesi cases were positively associated with forest cover and historical forest loss in surrounding areas. These results suggest the likelihood that deforestation and associated environmental changes are key drivers in P. knowlesi transmission in these areas.
Since the initial description of a large cluster of human infections with the zoonotic malaria Plasmodium knowlesi in Malaysian Borneo in 2004, increasing numbers of P. knowlesi cases have been identified throughout Southeast Asia (1,2). Although most persons infected with P. knowlesi respond to treatment, infection can cause severe and fatal disease (3). Understanding the distribution of P. knowlesi malaria and risk factors associated with this disease is critical for designing appropriate public health interventions.
Carried by long- and pig-tailed macaques (Macaca fascicularis and M. nemestrina), the P. knowlesi parasite has a geographic range limited by the distribution of mosquito vectors and simian hosts (2). Within this range, risk for P. knowlesi infection in humans is highly variable. Although sporadic P. knowlesi cases have been reported in several Southeast Asia countries, P. knowlesi is the most common cause of human malaria Malaysian Borneo, the portion of the country that lies on the island of Borneo (1,2). In the state of Sabah, suspected P. knowlesi notifications increased from 2% (59/2,741) of total malaria notifications in 2004 to 62% (996/1,606) in 2013 (4,5).
Molecular studies indicate that zoonotic P. knowlesi is not a newly emergent malaria species and is likely to predate human settlement in Southeast Asia (6). P. knowlesi was first described in macaques in the 1930s, and the first naturally acquired human case was reported in 1965 in peninsular Malaysia (7,8). However, true incidence and effects of P. knowlesi are poorly understood because of its frequent misidentification by microscopy as other human malaria species and because of limited availability of P. knowlesi–specific molecular diagnostic capabilities. P. knowlesi appears microscopically similar to the human malaria species P. malariae but can also be misdiagnosed as P. falciparum or P. vivax (3). The extent to which improved detection has contributed to recent increases in numbers of human cases is difficult to determine; however, the rise in P. knowlesi relative to other malaria species strongly suggests that P. knowlesi transmission has increased in Malaysian Borneo (4,5).
Land use changes, such as deforestation and agricultural expansion, have been proposed as the main drivers of this apparent emergence (3). Deforestation and related forest activities have been associated with changes in malaria vector populations and related disease incidence globally (9). Changes in vegetation, microclimate, and soil composition can affect the species composition and abundance of mosquito populations (10). In Malaysia, studies have implicated the primarily exophagic Anopheles leucosphyrus group of mosquitoes as the main vector of P. knowlesi and have found relatively high biting rates in farm edges bordering forests and forest areas (11–14). Human-disturbed environments have been associated with changes in behavior of nonhuman primates and their increased contact with humans (15). Fragmentation of existing habitats can also increase the frequency of disease transmission by creating transition areas with increased spatial overlap among human, mosquito, and wildlife populations or by altering vector ecology (16,17). The effects of these changes at forest edges have been described for malaria and other vector-borne zoonotic diseases (18,19) but not for P. knowlesi. A previous mathematic modeling study highlighted the potential for increased transmission resulting from increased spatial overlap among people, macaques, and mosquitoes at forest edges (20).
Despite these apparent links between land use and P. knowlesi transmission, detailed environmental risk factors for P. knowlesi infections in humans are unknown. Although variability of P. knowlesi risk has been reported at a regional scale, patterns of P. knowlesi transmission have not been described at a subdistrict spatial scale (2,21). Furthermore, studies that formally evaluate associations between P. knowlesi and characteristics of environment and landscape are lacking.
After obtaining approval from the Medical Research and Ethics Committee of the Ministry of Health in Malaysia, we examined the changing incidence of P. knowlesi in Kudat and Kota Marudu districts in northwestern Sabah, Malaysia, on the island of Borneo, an area with relatively high P. knowlesi transmission (22). Our aim was to describe the spatial and temporal patterns of P. knowlesi incidence within these districts and to explore potential associations between village-level incidence and deforestation and other environmental factors. Clarifying these relationships is vital to predicting and responding to future disease outbreaks and understanding the underlying mechanisms of P. knowlesi emergence.
Study Site and Population
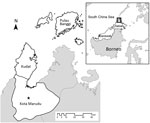
This study was conducted in the districts of Kudat and Kota Marudu in northwestern Sabah, Malaysia (07.38°–06.19°N, 116.62°–117.46°E), an area of 3,204 km2 with a population of ≈120,000 persons predominantly of Rungus and Dusun ethnicities (23) (Figure 1). The climate is tropical, with no dry season and increased rainfall during November–March; the area has both coastal and inland regions and elevations ranging from sea level to 1,000 m above sea level. Substantial environmental change is ongoing in the region because of conversion of land for oil palm plantations and other agricultural activities (24). Both districts have central referral hospitals that serve defined catchment areas where patients have access to diagnosis and treatment free of charge. All clinics refer patients to the central district hospital, where hospitalization is mandatory for malaria patients until a negative blood smear for malaria parasites has been obtained.
Geolocation of Patients and Villages
We conducted a retrospective review of malaria patients reported by Kudat and Kota Marudu district hospitals during 2008–2012. Data on diagnosis, admission date, demographics, and address of all malaria patients were obtained from hospital laboratory microscopy records. Villages and populations were identified from the 2010 Population and Housing Census in Malaysia (23), the most recent census conducted in this area, and the global positioning system coordinates of village centroids were recorded as part of a larger interdisciplinary study (MONKEYBAR ESEI project; http://malaria.lshtm.ac.uk/MONKEYBAR). Village populations were updated by using published population growth rates for Malaysia (25). All locations were confirmed by using imagery available through Google Earth (https://www.google.com/earth/) or other freely available satellite data. Patient addresses were matched to the census data or to the nearest reported village from the census data. Administrative boundaries were used to define the extent of urban areas, within which village data were combined.
Calculating the Proportion of Malaria Patients with P. knowlesi
P. knowlesi is microscopically similar to P. malariae but can also be misdiagnosed as P. falciparum or other human malaria species. In hospital microscopy records, the species of malaria is recorded, as determined by morphology, but no separate listing for P. knowlesi exists. Consequently, uncertainty in diagnosis resulted in some infections being recorded as P. malariae or P. malariae/knowlesi. To estimate the true proportion of P. knowlesi, we calculated the sensitivity and specificity of microscopy diagnosis of P. malariae for a subset of 539 malaria patients for whom both microscopy and molecularly confirmed results were available, including all patients from Kudat and Kota Marudu hospitals who were referred to a tertiary care hospital during this period and all patients recruited at these hospitals during 2013 and 2014 (22,26). The proportion of malaria cases reported as P. knowlesi per village per year was adjusted for this sensitivity and specificity by using a Bayesian estimation of true incidence from apparent incidence obtained by testing individual samples (27). This estimation is calculated as follows:
Models were fitted in R (http://www.R-project.org) through the prevalence and rjags packages interfacing with JAGS version 3.4.0 (http://mcmc-jags.sourceforge.net/) by using 2 chains containing 1,000 burn-in samples and 5,000 retained samples. Sensitivity and specificity parameters were determined by using β-PERT distributions of the minimum, maximum, and most likely values.
Environmental Data
Topography and land use data were extracted from various datasets derived from satellite-based remote-sensing data. These data were evaluated for buffer areas (i.e., areas within a certain radius of a village center) with a radius of 1, 2 and 5 km from the center of each village; these distances were chosen to explore a range of spatial scales at which environment may be relevant on the basis of the typical distribution of households, farming land, and local human and animal movements. Elevation data with a spatial resolution of 30 m were obtained from the ASTER Digital Global Elevation Model (28). The average annual normalized difference vegetation index (NDVI), which quantifies the greenness of vegetation, was calculated from Moderate Resolution Imaging Spectroradiometer 16-day composites at 250-m resolution (29). The NDVI is influenced by climatic factors (e.g., rainfall and temperature) and has been used extensively to predict malaria incidence and develop early warning systems in other contexts (30,31).
Tree cover data, derived from classified Landsat imagery at 30-m resolution, were obtained from Hansen et al. (32). Annual forest cover maps for the study districts were produced; forest was defined as >50% tree crown cover density. Although this land classification represents forested areas, it cannot distinguish types of forest or agroforestry such as rubber or oil palm. The proportion of forest coverage, proportion of forest loss during the year for which incidence was estimated, and proportion of cumulative forest loss for the previous 5 years (i.e., total forest loss for 2006–2010 was evaluated by using 2010 incidence) were calculated for each buffer radius for each village and time point. We used the Landscape Ecology Statistics plugin for Quantum GIS (33) to evaluate the effect of forest configuration as the number of forest patches per radius, a standard metric representing landscape fragmentation. Distributions of these variables were examined, and quartiles were used to categorize variables.
Because this analysis relied on passive reporting of malaria, we included travel time to the nearest clinic where patients would seek treatment for a febrile illness as a measure of access to care. Travel time to the clinic from each village was estimated by using travel times reported in community interviews and by patients recruited as part of a population-based case–control study (26).
Statistical Analysis
Annual P. knowlesi incidence for each village was mapped and smoothed incidence maps produced to visualize the data by using a kernel density estimation method, a standard method for interpolating point location data. Because the data were skewed relative to Poisson distribution, potential associations between environmental factors and reported P. knowlesi patients at the village level were assessed by using general linearized mixed models with a negative binomial distribution and an offset for population in R (34). To account for correlation between repeat measurements for the same village, we included the village variable as a random effect. Bivariable analysis was conducted for each covariate; variables for which p<0.2 were included in multivariable models. We used likelihood ratio tests to assess the significance of single variables and the Akaike Information Criterion (http://www.modelselection.org/aic/) for the final model selection. For correlated variables (e.g., mean elevation at different buffer radiuses), a single variable was selected for inclusion on the basis of marginal Akaike Information Criterion values. Potential bias from residual spatial autocorrelation in the model was explored for island and mainland areas through Moran’s I. On the basis of this statistic, the negative binomial model was fit with a spatial correlation component estimated by using a distance-based Matern correlation function.
Malaria in Kudat and Kota Marudu Districts from 2008–2012
A total of 405 villages were mapped in Kudat and Kota Marudu districts; median population per village was 168 persons (interquartile range 80–313), and median number of households was 44 (interquartile range 20–78). A total of 2,006 malaria patients were reported during 2008–2012: 833 cases in Kota Marudu district, 1,014 in Kudat district, and 159 from outside these districts. Standard reporting forms did not include age-specific data but classified patients as adults (65.7%, 1,318/2,006) or children (32.8%, 657/2,006), with a small number of records (1.5%, 31/2006) missing this information. Most malaria patients were male (66%, 1,330/2,006).
Most villages (60%, 245/405) reported at least 1 malaria patient during this period. The number of malaria patients reported varied annually, with marked seasonal variations in numbers of patients and amount of rainfall. Almost half (47%, 878/1,847) of reported malaria patients had suspected P. knowlesi or P. malariae infections diagnosed by microscopy. P. falciparum and P. vivax malaria were diagnosed in another 27% (512/1,847) and 25% (457/1,847) of patients, respectively.
Of 346 samples collected from patients for whom P. knowlesi or P. malariae was diagnosed by microscopy (including mixed infections) and sent for molecular confirmation, 90% (313/346) were confirmed as P. knowlesi by PCR. Sensitivity and specificity of microscopy diagnosis for P. knowlesi were 95% (95% CI 92%–97%) and 84% (95% CI 79%–89%), respectively (Table 1). Although studies have reported frequent misdiagnosis (1–3,5), few samples (3%, 16/539) were incorrectly identified as other species by microscopy. Because PCR results were available only for patients with confirmed malaria, we could not estimate the probability of detecting submicroscopic infections.
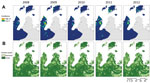
By using these values derived from collected samples, the true number of P. knowlesi patients was estimated as 739 (95% CI 664–794) for Kudat and Kota Marudu districts during 2008–2012. The range of estimated annual parasite incidence (API, expressed as cases/1,000 person/y) for P. knowlesi malaria calculated for each village was 0–102; overall mean API was 1.84. Of the 245 villages reporting malaria patients, 24% (59/245) had an estimated mean API for P. knowlesi of <l; the highest proportion, 44% (108/245), each had a mean API of 1–5; another 11% (26/245) had a mean API of 5–10; and 6% (15/245) had a mean API of 10–20. Two villages had a mean API of >20 (Figure 2, panel A).
Association with Environmental Variables
On the basis of estimates from remote sensing data (32), forest cover declined by 4.8% in Kudat and Kota Marudu during 2008–2012 (Figure 2, panel B). Although this loss was often highly localized, large tracts of forest were cleared in the interior of Pulau Banggi. Overall, substantial environmental change was observed in the districts; 39% (157/405) of villages lost >10% of forest cover within a 1-km radius during this 5-year period; 44% (179/405) lost >10% within a 2-km radius; and 51% (206/405) lost >10% within a 5-km radius.
Lower elevations, >65% of forest cover (within a 2-km radius), and higher historical forest loss were associated with greater incidence of P. knowlesi infections. Travel times to clinics, average annual NDVI, and number of forest patches were not significant at any radius in the bivariate analysis.
Spatial Patterns
Maps of interpolated P. knowlesi incidence show distinctive spatial patterns that vary from year to year, with some areas of high incidence persisting over time (Figure 2, panel A). To assess the degree of residual spatial correlation after fitting the negative binomial regression, the residuals of the best-fitting model were mapped and Moran’s I was calculated (mainland Borneo, Moran’s I 0.07, p<0.0001; islands off Borneo’s coast, Moran’s I 0.07, p = 0.07). Because Moran’s I was significant, the final model was adjusted for spatial autocorrelation (Tables 2, 3).
This study aimed to describe spatial and temporal patterns of P. knowlesi infection in northern Sabah, Malaysia, and to evaluate potential associations between village-level P. knowlesi incidence and key environmental factors. Although land use changes can affect emergence of infectious diseases, such associations have not been previously evaluated for P. knowlesi. We found links among deforestation, environmental characteristics, and reported incidence of P. knowlesi malaria in this region.
Village-level malaria data show marked spatial and temporal heterogeneity in P. knowlesi incidence in Kudat and Kota Marudu districts. After adjustment for the sensitivity and specificity of microscopy, P. knowlesi was the most common cause of human malaria, a finding consistent with other studies for this geographic area (4,5,22). Quantification of annual forest loss indicates that substantial environmental changes occurred during 2008–2012, with many villages losing substantial proportions of surrounding forest cover.
The proportion of forest surrounding a village was associated with incidence of P. knowlesi infections in the final model (Table 3); this association potentially reflects the role of forest environments as habitats of macaques and mosquito vectors. Studies report higher vectorial capacity and sporozoite rates for P. knowlesi in forest environments than in agricultural and settled lands (12,35). Long-tailed macaques have also been reported in various environments, including degraded secondary forest areas (36). The association of increased P. knowlesi incidence with both forest and forest loss likely confirms findings that transmission is occurring in forested areas undergoing substantial change, as with previously described frontier malaria (37).
Higher incidence of P. knowlesi was associated with higher proportions of forest loss surrounding villages during the 5-year period before year of reported incidence. This association could result from changes in macaque or mosquito habitats and from increased levels of human activity. Increased density of long-tailed macaques has been reported as a response to deforestation; loss of previous habitats can result in crowding within forest patches, with potential implications for disease transmission (36). Land use changes have also been shown to affect abundance and community composition of potential vectors (17,38). Deforestation and associated agricultural development are also associated with changes in human risk because of altered distribution and behavior of humans; these changes result from more employment opportunities and shifts in human movement patterns because of forest clearing and agricultural activities (19,37). Although clearing of forests may initially deplete vector populations and thereby reduce malaria transmission, this reduction may be followed by colonization of cleared areas by more efficient vector species and subsequent increases in transmission (9). Historical forest loss was more significantly associated with P. knowlesi incidence than forest loss occurring during the same year of reported incidence (Table 2), suggesting that increased transmission is related to long-term changes in vector, host, or human populations involved. Additional longitudinal studies are required to investigate this hypothesis.
The effect of habitat may also be reflected in associations with elevation. Elevation was negatively correlated with P. knowlesi incidence, although the range of elevations within the study site was limited; most villages were at elevations <100 m. Both macaques and vectors are more frequently described in low elevation areas but have been reported at higher elevations (36,39).
Neither forest configuration nor vegetation indices appeared to be strongly associated with P. knowlesi incidence. Because NDVI is a measure of vegetation greenness, this measure may not differentiate between forest and other types of agriculture (e.g., oil palm) because NDVI tends to become saturated in tropical environments. Studies of other zoonotic diseases have found that the fragmentation and configuration of other types of land cover, in addition to forest, influence disease transmission (18,19). Other fragmentation metrics could be included to evaluate the effects of forest patch size and shape on P. knowlesi transmission.
The buffer sizes that we evaluated represent a range of potential scales at which variables (e.g., mean elevation) within different radiuses may affect P. knowlesi transmission. Human infections result from many factors interacting across different spatial scales, and the strength of association of these factors likely varies by distance. Our study covers an extensive area in which behaviors, distribution of villages, and ecology vary. Villages are typically within small spatial areas, but farming practices range from small-scale swidden farming to large-scale plantations and affect the scale of human interactions with the environment. Future analyses would benefit from including more detailed spatial data on household locations and human movement patterns.
The main limitation of this study is the reliance on records of malaria patients who sought care at hospitals; these patients may represent only a portion of malaria patients in the community. Although malaria is a notifiable disease and distance to a hospital was included as a measure of access to care, asymptomatic malaria cases and symptomatic cases that resolved without treatment are unaccounted for. Previous studies of other malaria species in similar transmission settings have described a large proportion of asymptomatic carriage within communities; however, asymptomatic carriage has not been evaluated for P. knowlesi (40). A cross-sectional survey of 2,019 persons in central Vietnam identified 3 persons whose samples were positive for P. knowlesi, yet all 3 were asymptomatic at the time of the survey and for the subsequent 6 months, showing that asymptomatic carriage of P. knowlesi can occur (40).
This study was also limited by the environmental data used. Data on forest cover and forest loss were aggregated by year, and finer-scale temporal associations between land cover (i.e., forest and other land types) and incidence could not be explored. This dataset was limited by defining forest as canopy cover, a definition that does not enable differentiation between types of forest or crops or between patches of different types of land. In addition, analysis was limited by the spatial resolution of satellite-based remote-sensing data, the use of a centroid point to represent village location, and the use of circular buffers rather than buffers of actual village shape. Exploratory spatial analysis suggested spatial heterogeneity in village-level data, and these spatial effects were included in the model. Additional work with more spatially specific outcomes and environmental data are needed to investigate these spatial patterns in more detail.
Despite inherent limitations in the outcome and covariate data used in this study, results strongly suggest a link between environmental change and reported incidence of emerging P. knowlesi in northern Sabah. Spatial analysis of environmental factors affecting disease emergence can be used to target surveillance and public health activities to areas expected to have increasing disease risk. Although additional population-based studies are needed to define environmental risk factors, this study indicates that deforestation is associated with human cases of P. knowlesi within northwestern Sabah, Malaysia.
Ms. Fornace is a research fellow at the London School of Hygiene and Tropical Medicine in London, UK, and Kota Kinabalu, Malaysia. Her primary research interest is spatial epidemiology of zoonotic diseases.
Acknowledgments
We thank Albert M Lim, Lina Marlina Gulam, Beth Downe, and the MONKEYBAR team in Sabah for their help with this study. N. Alexander receives salary support from the Medical Research Council and Department for International Development (MR/K012126/1).
We acknowledge the Medical Research Council, Natural Environment Research Council, Economic and Social Research Council, and Biotechnology and Biosciences Research Council for funding received for this project through the Environmental and Social Ecology of Human Infectious Diseases Initiative, grant no. G1100796.
References
- Singh B, Kim Sung L, Matusop A, Radhakrishnan A, Shamsul SS, Cox-Singh J, A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet. 2004;363:1017–24. DOIPubMedGoogle Scholar
- Moyes CL, Henry AJ, Golding N, Huang Z, Singh B, Baird JK, Defining the geographical range of the Plasmodium knowlesi reservoir. PLoS Negl Trop Dis. 2014;8:e2780. DOIPubMedGoogle Scholar
- Singh B, Daneshvar C. Human infections and detection of Plasmodium knowlesi. Clin Microbiol Rev. 2013;26:165–84. DOIPubMedGoogle Scholar
- William T, Rahman HA, Jelip J, Ibrahim MY, Menon J, Grigg MJ, Increasing incidence of Plasmodium knowlesi malaria following control of P. falciparum and P. vivax malaria in Sabah, Malaysia. PLoS Negl Trop Dis. 2013;7:e2026. DOIPubMedGoogle Scholar
- William T, Jelip J, Menon J, Anderios F, Mohammad R, Awang Mohammad TA, Changing epidemiology of malaria in Sabah, Malaysia: increasing incidence of Plasmodium knowlesi. Malar J. 2014;13:390. DOIPubMedGoogle Scholar
- Lee KS, Divis PC, Zakaria SK, Matusop A, Julin RA, Conway DJ, Plasmodium knowlesi: reservoir hosts and tracking the emergence in humans and macaques. PLoS Pathog. 2011;7:e1002015. DOIPubMedGoogle Scholar
- Chin W, Contacos PG, Coatney GR, Kimball HR. A naturally acquited quotidian-type malaria in man transferable to monkeys. Science. 1965;149:865. DOIPubMedGoogle Scholar
- Knowles R, Das Gupta BM. A study of monkey-malaria and its experimental transmission to man. Ind Med Gaz. 1932;67:301–20.
- Guerra CA, Snow RW, Hay SI. A global assessment of closed forests, deforestation and malaria risk. Ann Trop Med Parasitol. 2006;100:189–204. DOIPubMedGoogle Scholar
- Yasuoka J, Levins R. Impact of deforestation and agricultural development on anopheline ecology and malaria epidemiology. Am J Trop Med Hyg. 2007;76:450–60.PubMedGoogle Scholar
- Tan CH, Vythilingam I, Matusop A, Chan ST, Singh B. Bionomics of Anopheles latens in Kapit, Sarawak, Malaysian Borneo in relation to the transmission of zoonotic simian malaria parasite Plasmodium knowlesi. Malar J. 2008;7:52. DOIPubMedGoogle Scholar
- Vythilingam I, Noorazian YM, Huat TC, Jiram AI, Yusri YM, Azahari AH, Plasmodium knowlesi in humans, macaques and mosquitoes in peninsular Malaysia. Parasit Vectors. 2008;1:26. DOIPubMedGoogle Scholar
- Hii J. Anopheles malaria vector in Malaysia with reference to Sabah. In: Harinasuta R, editor. Problems of malaria in endemic countries. Tokyo: Southeast Asian Medical Information Centre; 1985. p. 71–81.
- Hii J, Rueda LM. Malaria vectors in the Greater Mekong Subregion: overview of malaria vectors and remaining challenges. Southeast Asian J Trop Med Public Health. 2013;44(Suppl 1):73–165, discussion 306–7.PubMedGoogle Scholar
- Young H, Griffin RH, Wood CL, Nunn CL. Does habitat disturbance increase infectious disease risk for primates? Ecol Lett. 2013;16:656–63. DOIPubMedGoogle Scholar
- Hutchings RS, Sallum MA, Hutchings RW. Mosquito (Diptera: Culicidae) diversity of a forest-fragment mosaic in the Amazon rain forest. J Med Entomol. 2011;48:173–87. DOIPubMedGoogle Scholar
- Overgaard HJ, Ekbom B, Sukonkerd W, Takagi M. Effect of landscape structure on anopheline mosquito density and diversity in northern Thailand: implications for malaria transmission and control. Landscape Ecol. 2003;18:605–19. DOIGoogle Scholar
- Lambin EF, Tran A, Vanwambeke SO, Linard C, Soti V. Pathogenic landscapes: interactions between land, people, disease vectors, and their animal hosts. Int J Health Geogr. 2010;9:54. DOIPubMedGoogle Scholar
- Vanwambeke SO, Lambin EF, Eichhorn MP, Flasse SP, Harbach RE, Oskam L, Impact of land-use change on dengue and malaria in northern Thailand. EcoHealth. 2007;4:37–51. DOIGoogle Scholar
- Imai N, White MT, Ghani AC, Drakeley CJ. Transmission and control of Plasmodium knowlesi: a mathematical modelling study. PLoS Negl Trop Dis. 2014;8:e2978. DOIPubMedGoogle Scholar
- Alias H, Surin J, Mahmud R, Shafie A, Mohd Zin J, Mohamad Nor M, Spatial distribution of malaria in Peninsular Malaysia from 2000 to 2009. Parasit Vectors. 2014;7:186. DOIPubMedGoogle Scholar
- Barber BE, William T, Dhararaj P, Anderios F, Grigg MJ, Yeo TW, Epidemiology of Plasmodium knowlesi malaria in north-east Sabah, Malaysia: family clusters and wide age distribution. Malar J. 2012;11:401. DOIPubMedGoogle Scholar
- Department of Statistics, Malaysia. Population and housing census, Malaysia. Putrajaya (Malaysia); 2010.
- Bryan JE, Shearman PL, Asner GP, Knapp DE, Aoro G, Lokes B. Extreme differences in forest degradation in Borneo: comparing practices in Sarawak, Sabah, and Brunei. PLoS ONE. 2013;8:e69679. DOIPubMedGoogle Scholar
- United Nations Statistics Division. UNDATA. Malaysia. 2015 [cited 2015 July 1]. http://data.un.org/CountryProfile.aspx?crName=MALAYSIA
- Grigg MJ, William T, Drakeley CJ, Jelip J, von Seidlein L, Barber BE, Factors that are associated with the risk of acquiring Plasmodium knowlesi malaria in Sabah, Malaysia: a case-control study protocol. BMJ Open. 2014;4:e006004. DOIPubMedGoogle Scholar
- Speybroeck N, Devleesschauwer B, Joseph L, Berkvens D. Misclassification errors in prevalence estimation: Bayesian handling with care. Int J Public Health. 2013;58:791–5. DOIPubMedGoogle Scholar
- Japan Space Systems. Advanced spaceborne thermal emission and reflection radiometer. ASTER Global Digital Elevation Model version 2.0 (ASTER GDEM). 2014 [cited 2014 Nov 17]. http://gdem.ersdac.jspacesystems.or.jp/
- National Aeronautics and Space Administration. Moderate resolution imaging spectroradiometer. MODIS land vegetation indices. 2008–2012 [cited 2014 Dec 19]. http://modis-land.gsfc.nasa.gov/vi.html
- Liu J, Chen XP. Relationship of remote sensing normalized differential vegetation index to Anopheles density and malaria incidence rate. Biomed Environ Sci. 2006;19:130–2.PubMedGoogle Scholar
- Wayant NM, Maldonado D, Rojas de Arias A, Cousino B, Goodin DG. Correlation between normalized difference vegetation index and malaria in a subtropical rain forest undergoing rapid anthropogenic alteration. Geospat Health. 2010;4:179–90. DOIPubMedGoogle Scholar
- Hansen MC, Potapov PV, Moore R, Hancher M, Turubanova SA, Tyukavina A, High-resolution global maps of 21st-century forest cover change. Science. 2013;342:850–3. DOIPubMedGoogle Scholar
- Plugin: LecoS—landscape ecology statistics. 2012. PGIS Python Plugins Repository [cited 2015 Feb 25]. http://plugins.qgis.org/plugins/LecoS
- Hilbe JM. Negative binomial regression. Cambridge: Cambridge University Press; 2007.
- Wong ML, Chua TH, Leong CS, Khaw LT, Fornace K, Wan-Sulaiman WY, Seasonal and spatial dynamics of the primary vector of Plasmodium knowlesi within a major transmission focus in Sabah, Malaysia. PLoS Negl Trop Dis. 2015;9:e0004135. DOIPubMedGoogle Scholar
- Fooden J. Systematic review of Southeast Asian longtail macaques, Macaca fascicularis (Raffles, 1821). Fieldiana Zoology. 1995;81:1–206.
- de Castro MC, Monte-Mór RL, Sawyer DO, Singer BH. Malaria risk on the Amazon frontier. Proc Natl Acad Sci U S A. 2006;103:2452–7. DOIPubMedGoogle Scholar
- Brant HL. Changes in abundance, diversity and community composition of mosquitoes based on different land use in Sabah, Malaysia. London: Imperial College London; 2011.
- Sallum MAM, Peyton EL, Wilkerson RC. Six new species of the Anopheles leucosphyrus group, reinterpretation of An. elegans and vector implications. Med Vet Entomol. 2005;19:158–99. DOIPubMedGoogle Scholar
- Van den Eede P, Van HN, Van Overmeir C, Vythilingam I, Duc TN. Hung le X, et al. Human Plasmodium knowlesi infections in young children in central Vietnam. Malar J. 2009;8:249.
Figures
Tables
Cite This ArticleTable of Contents – Volume 22, Number 2—February 2016
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Kimberly M. Fornace, Faculty of Infectious and Tropical Diseases, London School of Hygiene and Tropical Medicine, Keppel Street, London, WC1E 7HT, UK
Top